Abstract
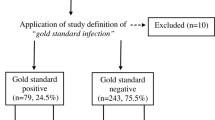
The bioburden screening process of allograft musculoskeletal tissue samples received at the South Eastern Area Laboratory Services includes the routine use of solid agar and cooked meat (CM) broth media. CM has been routinely sub-cultured onto solid agar plates after aerobic incubation at 35 °C. This study will evaluate whether a visual assessment of CM can replace sub-culture by an in vitro inoculation and a prospective study. Eight challenge organisms were serially diluted and inoculated into CM. The average inoculum of 0.5–5.5 CFU produced visible turbidity of CM after 24-h incubation for 7 of the challenge organisms with one organism producing turbidity after 48-h incubation. The prospective study evaluated 222 CM of which 213 were visually clear and no-growth on sub-culture and 9 turbid CM which were culture positive. Broth cultures are an integral part of the bioburden screening process of allograft musculoskeletal tissue and swab samples and visual assessment of CM can replace sub-culture.
Similar content being viewed by others
References
British Pharmacopoeia Commission (2016) British pharmacopoeia volume 5 appendix XVI a testing for sterility. London, United Kingdom
Derby P, Davies R, Oliver S (1997) The value of including broth cultures as part of a routine culture protocol. J Clin Micro 35:1101–1102
Reinhold CE, Nickolai DJ, Picciini TE, Byford BA, York MK, Brooks GF (1988) Evaluation of broth media for routine culture of cerebrospinal and joint fluid specimens. Am J Pathol 89(5):671–674
SaBTO: The Advisory Committee on the Safety of Blood, Tissues and Organs (2011) Guidance on the microbiological safety of human organs, tissues and cells used in transplantation. Department of Health, UK
Saegeman VSM, Lismont D, Verduyckt B, Ectors NL, Verhaegen J (2007) Comparison of microbiological culture methods in screening allograft tissue. Swab versus nutrient broth. J Microbiol Methods 10:374–378
Scythes KD, Louie M, Simor AE (1996) Evaluation of nutritive capacities of 10 broth media. J Clin Micro 34(7):1804–1807
Therapeutic Goods Administration (2013) Australian code of good manufacturing practice for human blood and blood components, human tissues and human cellular therapy products. Department of Health and Ageing, Canberra
Varettas K (2013a) Broth versus solid agar culture of swab samples of cadaveric musculoskeletal tissue. Cell Tiss Bank 14:627–631
Varettas K (2013b) Micro-organisms isolated from cadaveric samples of allograft musculoskeletal tissue. Cell Tissue Bank 14:621–625
Varettas K, Taylor P (2011) Bioburden assessment of banked bone used for allografts. Cell Tissue Bank 12:37–43
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varettas, K. The visual assessment of broth cultures for tissue bank samples. Cell Tissue Bank 18, 343–345 (2017). https://doi.org/10.1007/s10561-016-9608-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-016-9608-z