Abstract
Introduction
Transplantation of a cell-seeded graft may improve wound healing after radiotherapy. However, the survival of the seeded cells depends on a rapid vascularization of the graft. Co-culturing of adult stem cells may be a promising strategy to accelerate the vessel formation inside the graft. Thus, we compared the in vivo angiogenic potency of mesenchymal stem cells (MSC) and endothelial progenitor cells (EPC) using dorsal skinfold chambers and intravital microscopy.
Materials and methods
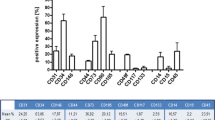
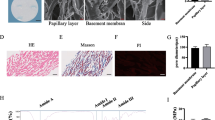
Cells were isolated from rat bone marrow and adipose tissue and characterized by immunostaining and flow cytometry. Forty-eight rats received a dorsal skinfold chamber and were divided into 2 main groups, irradiated and non-irradiated. Each of these 2 groups were further subdivided into 4 groups: unseeded matrices, matrices + fibroblasts + pericytes, matrices + fibroblasts + pericytes + MSCs and matrices + fibroblasts + pericytes + EPCs. Vessel densities were quantified semi-automatically using FIJI.
Results
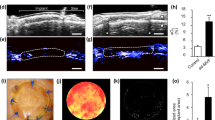
Fibroblasts + pericytes − seeded matrices showed a significantly higher vascular density in all groups with an exception of non-irradiated rats at day 12 compared to unseeded matrices. Co-seeding of MSCs increased vessel densities in both, irradiated and non-irradiated groups. Co-seeding with EPCs did not result in an increase of vascularization in none of the groups.
Discussion
We demonstrated that the pre-radiation treatment led to a significant decreased vascularization of the implanted grafts. The augmentation of the matrices with fibroblasts and pericytes in co-culture increased the vascularization compared to the non-seeded matrices. A further significant enhancement of vessel ingrowth into the matrices could be achieved by the co-seeding with MSCs in both, irradiated and non-irradiated groups.






Similar content being viewed by others
References
Abramsson A, Berlin O, Papayan H, Paulin D, Shani M, Betsholtz C (2002) Analysis of mural cell recruitment to tumor vessels. Circulation 105:112–117
Alajati A et al (2008) Spheroid-based engineering of a human vasculature in mice. Nat Methods 5:439–445. doi:10.1038/nmeth.1198
Asahara T et al (1997) Isolation of putative progenitor endothelial cells for angiogenesis. Science 275:964–967
Au P, Tam J, Fukumura D, Jain RK (2008) Bone marrow-derived mesenchymal stem cells facilitate engineering of long-lasting functional vasculature. Blood 111:4551–4558. doi:10.1182/blood-2007-10-118273
Baiguera S, Ribatti D (2013) Endothelialization approaches for viable engineered tissues. Angiogenesis 16:1–14. doi:10.1007/s10456-012-9307-8
Baiguera S, Jungebluth P, Mazzanti B, Macchiarini P (2012) Mesenchymal stromal cells for tissue-engineered tissue and organ replacements Transplant international. Transplantation 25:369–382. doi:10.1111/j.1432-2277.2011.01426.x
Baldwin J et al (2014) In vitro pre-vascularisation of tissue-engineered constructs A co-culture perspective. Vasc Cell 6:13. doi:10.1186/2045-824X-6-13
Bernardo ME, Fibbe WE (2013) Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell 13:392–402. doi:10.1016/j.stem.2013.09.006
Bianco P, Riminucci M, Gronthos S, Robey PG (2001) Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells 19:180–192. doi:10.1634/stemcells.19-3-180
Bryan BA, D’Amore PA (2008) Pericyte isolation and use in endothelial/pericyte coculture models. Methods Enzymol 443:315–331. doi:10.1016/S0076-6879(08)02016-8
Chang P, Qu Y, Liu Y, Cui S, Zhu D, Wang H, Jin X (2013) Multi-therapeutic effects of human adipose-derived mesenchymal stem cells on radiation-induced intestinal injury. Cell Death Dis 4:e685. doi:10.1038/cddis.2013.178
Chen X, Aledia AS, Popson SA, Him L, Hughes CC, George SC (2010) Rapid anastomosis of endothelial progenitor cell-derived vessels with host vasculature is promoted by a high density of cotransplanted fibroblasts. Tissue Eng Part A 16:585–594. doi:10.1089/ten.TEA.2009.0491
Choi JH et al (2010) Adipose tissue engineering for soft tissue regeneration. Tissue Eng Part B Rev 16:413–426. doi:10.1089/ten.TEB.2009.0544
Costa-Almeida R, Gomez-Lazaro M, Ramalho C, Granja PL, Soares R, Guerreiro SG (2015) Fibroblast-endothelial partners for vascularization strategies in tissue engineering. Tissue Eng Part A 21:1055–1065. doi:10.1089/ten.TEA.2014.0443
da Cunha FF, Martins L, Martin PK, Stilhano RS, Paredes Gamero EJ, Han SW (2013) Comparison of treatments of peripheral arterial disease with mesenchymal stromal cells and mesenchymal stromal cells modified with granulocyte and macrophage colony-stimulating factor. Cytotherapy 15:820–829. doi:10.1016/j.jcyt.2013.02.014
Dantzer D et al (2003) Effect of radiation and cell implantation on wound healing in a rat model. J Surg Oncol 83:185–190. doi:10.1002/jso.10242
Dominici M et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8:315–317. doi:10.1080/14653240600855905
Dore-Duffy P, Cleary K (2011) Morphology and properties of pericytes. Methods Mol Biol 686:49–68. doi:10.1007/978-1-60761-938-3_2
English K (2013) Mechanisms of mesenchymal stromal cell immunomodulation. Immunol Cell Biol 91:19–26. doi:10.1038/icb.2012.56
Fadini GP, Baesso I, Albiero M, Sartore S, Agostini C, Avogaro A (2008) Technical notes on endothelial progenitor cells: ways to escape from the knowledge plateau. Atherosclerosis 197:496–503. doi:10.1016/j.atherosclerosis.2007.12.039
Falanga V et al (2007) Autologous bone marrow-derived cultured mesenchymal stem cells delivered in a fibrin spray accelerate healing in murine and human cutaneous wounds. Tissue Eng 13:1299–1312. doi:10.1089/ten.2006.0278
Farini A, Sitzia C, Erratico S, Meregalli M, Torrente Y (2014) Clinical applications of mesenchymal stem cells in chronic diseases. Stem Cells Int 2014:306573. doi:10.1155/2014/306573
Farrington-Rock C, Crofts NJ, Doherty MJ, Ashton BA, Griffin-Jones C, Canfield AE (2004) Chondrogenic and adipogenic potential of microvascular pericytes. Circulation 110:2226–2232. doi:10.1161/01.CIR.0000144457.55518.E5
Ferguson PC, Boynton EL, Wunder JS, Hill RP, O’Sullivan B, Sandhu JS, Bell RS (1999) Intradermal injection of autologous dermal fibroblasts improves wound healing in irradiated skin. J Surg Res 85:331–338. doi:10.1006/jsre.1999.5664
Golas AR, Perez JL, Fullerton N, Lekic N, Hooper RC, Spector JA (2013) Vascular smooth muscle cell optimization of vasculogenesis within naturally derived, biodegradable, hybrid hydrogel scaffolds. Plast Reconstr Surg 132:952e–963e. doi:10.1097/PRS.0b013e3182a805df
Hasegawa T et al (2010) Efficient cell-seeding into scaffolds improves bone formation. J Dent Res 89:854–859. doi:10.1177/0022034510370022
Hegen A et al (2011) Efficient in vivo vascularization of tissue-engineering scaffolds. J Tissue Eng Regen Med 5:e52–e62. doi:10.1002/term.336
Hess DC, Hill WD, Martin-Studdard A, Carroll J, Brailer J, Carothers J (2002) Bone marrow as a source of endothelial cells and NeuN-expressing cells after stroke. Stroke 33:1362–1368
Hirschi KK, D’Amore PA (1996) Pericytes in the microvasculature. Cardiovasc Res 32:687–698
Hoffmann J, Glassford AJ, Doyle TC, Robbins RC, Schrepfer S, Pelletier MP (2010) Angiogenic effects despite limited cell survival of bone marrow-derived mesenchymal stem cells under ischemia. Thoracic Cardiovasc Surg 58:136–142. doi:10.1055/s-0029-1240758
Honmou O, Onodera R, Sasaki M, Waxman SG, Kocsis JD (2012) Mesenchymal stem cells: therapeutic outlook for stroke. Trends Mol Med 18:292–297. doi:10.1016/j.molmed.2012.02.003
Hu KX, Sun QY, Guo M, Ai HS (2010) The radiation protection and therapy effects of mesenchymal stem cells in mice with acute radiation injury. Br J Radiol 83:52–58. doi:10.1259/bjr/61042310
Hur J et al (2004) Characterization of two types of endothelial progenitor cells and their different contributions to neovasculogenesis. Arterioscler Thromb Vasc Biol 24:288–293. doi:10.1161/01.ATV.0000114236.77009.06
Ingram DA et al (2004) Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood 104:2752–2760. doi:10.1182/blood-2004-04-1396
Jain RK (2003) Molecular regulation of vessel maturation. Nat Med 9:685–693. doi:10.1038/nm0603-685
Kahler CM et al (2007) Peripheral infusion of rat bone marrow derived endothelial progenitor cells leads to homing in acute lung injury. Respir Res 8:50. doi:10.1186/1465-9921-8-50
Kampmann A et al (2013) Additive effect of mesenchymal stem cells and VEGF to vascularization of PLGA scaffolds. Microvasc Res 90:71–79. doi:10.1016/j.mvr.2013.07.006
Kesler CT et al (2013) Vascular endothelial growth factor-C enhances radiosensitivity of lymphatic endothelial cells. Angiogenesis. doi:10.1007/s10456-013-9400-7
Khan ZA, Melero-Martin JM, Wu X, Paruchuri S, Boscolo E, Mulliken JB, Bischoff J (2006) Endothelial progenitor cells from infantile hemangioma and umbilical cord blood display unique cellular responses to endostatin. Blood 108:915–921. doi:10.1182/blood-2006-03-006478
Kirkpatrick CJ, Fuchs S, Unger RE (2011) Co-culture systems for vascularization–learning from nature. Adv Drug Deliv Rev 63:291–299. doi:10.1016/j.addr.2011.01.009
Kloc M, Kubiak JZ, Li XC, Ghobrial RM (2015) Pericytes, microvasular dysfunction, and chronic rejection. Transplantation 99:658–667. doi:10.1097/TP.0000000000000648
Kocher AA et al (2001) Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 7:430–436. doi:10.1038/86498
Krauss MC (1999) Recent advances in soft tissue augmentation. Semin Cutan Med Surg 18:119–128
Kwon DS et al (2008) Treatment with bone marrow-derived stromal cells accelerates wound healing in diabetic rats. Int Wound J 5:453–463. doi:10.1111/j.1742-481X.2007.00408.x
Lamagna C, Bergers G (2006) The bone marrow constitutes a reservoir of pericyte progenitors. J Leukoc Biol 80:677–681. doi:10.1189/jlb.0506309
Lange C et al (2011) Radiation rescue: mesenchymal stromal cells protect from lethal irradiation. PLOS ONE 6:e14486. doi:10.1371/journal.pone.0014486
Laschke MW et al (2006) Angiogenesis in tissue engineering: breathing life into constructed tissue substitutes. Tissue Eng 12:2093–2104. doi:10.1089/ten.2006.12.2093
Laschke MW et al (2013) Three-dimensional spheroids of adipose-derived mesenchymal stem cells are potent initiators of blood vessel formation in porous polyurethane scaffolds. Acta Biomater 9:6876–6884. doi:10.1016/j.actbio.2013.02.013
Liu Y, Dulchavsky DS, Gao X, Kwon D, Chopp M, Dulchavsky S, Gautam SC (2006) Wound repair by bone marrow stromal cells through growth factor production. J Surg Res 136:336–341. doi:10.1016/j.jss.2006.07.037
Liu G et al (2014) Isolation, purification, and cultivation of primary retinal microvascular pericytes: a novel model using rats. Microcirculation 21:478–489. doi:10.1111/micc.12121
Madonna R, Taylor DA, Geng YJ, De Caterina R, Shelat H, Perin EC, Willerson JT (2013) Transplantation of mesenchymal cells rejuvenated by the overexpression of telomerase and myocardin promotes revascularization and tissue repair in a murine model of hindlimb ischemia. Circ Res 113:902–914. doi:10.1161/CIRCRESAHA.113.301690
Mao XW (2006) A quantitative study of the effects of ionizing radiation on endothelial cells and capillary-like network formation. Technol Cancer Res Treat 5:127–134
Martin BJ (2013) Inhibiting vasculogenesis after radiation: a new paradigm to improve local control by radiotherapy. Semin Radiat Oncol 23:281–287. doi:10.1016/j.semradonc.2013.05.002
Martins L, Martin PK, Han SW (2014) Angiogenic properties of mesenchymal stem cells in a mouse model of limb ischemia. Methods Mol Biol 1213:147–169. doi:10.1007/978-1-4939-1453-1_13
Maxson S, Lopez EA, Yoo D, Danilkovitch-Miagkova A, Leroux MA (2012) Concise review: role of mesenchymal stem cells in wound repair. Stem Cells Transl Med 1:142–149. doi:10.5966/sctm.2011-0018
McFadden TM et al (2013) The delayed addition of human mesenchymal stem cells to pre-formed endothelial cell networks results in functional vascularization of a collagen-glycosaminoglycan scaffold in vivo. Acta Biomater 9:9303–9316. doi:10.1016/j.actbio.2013.08.014
McFarlin K et al (2006) Bone marrow-derived mesenchymal stromal cells accelerate wound healing in the rat. Wound Repair Regen 14:471–478. doi:10.1111/j.1743-6109.2006.00153.x
Melero-Martin JM, Khan ZA, Picard A, Wu X, Paruchuri S, Bischoff J (2007) In vivo vasculogenic potential of human blood-derived endothelial progenitor cells. Blood 109:4761–4768. doi:10.1182/blood-2006-12-062471
Mukai N et al (2008) A comparison of the tube forming potentials of early and late endothelial progenitor cells. Exp Cell Res 314:430–440. doi:10.1016/j.yexcr.2007.11.016
Murray IR et al (2014) Natural history of mesenchymal stem cells, from vessel walls to culture vessels. CMLS 71:1353–1374. doi:10.1007/s00018-013-1462-6
Novaes AB Jr, Marchesan JT, Macedo GO, Palioto DB (2007) Effect of in vitro gingival fibroblast seeding on the in vivo incorporation of acellular dermal matrix allografts in dogs. J Periodontol 78:296–303. doi:10.1902/jop.2007.060060
Parekkadan B, Milwid JM (2010) Mesenchymal stem cells as therapeutics. Annu Rev Biomed Eng 12:87–117. doi:10.1146/annurev-bioeng-070909-105309
Pearson JD (2010) Endothelial progenitor cells–an evolving story. Microvasc Res 79:162–168. doi:10.1016/j.mvr.2009.12.004
Phelps EA, Garcia AJ (2010) Engineering more than a cell: vascularization strategies in tissue engineering. Curr Opin Biotechnol 21:704–709. doi:10.1016/j.copbio.2010.06.005
Phinney DG, Prockop DJ (2007) Concise review: mesenchymal stem/multipotent stromal cells: the state of trans differentiation and modes of tissue repair–current views. Stem Cells 25:2896–2902. doi:10.1634/stemcells.2007-0637
Reinhold HS, Calvo W, Hopewell JW, van der Berg AP (1990) Development of blood vessel-related radiation damage in the fimbria of the central nervous system. Int J Radiat Oncol Biol Phys 18:37–42
Roessner ED, Thier S, Hohenberger P, Schwarz M, Pott P, Dinter D, Smith M (2011) Acellular dermal matrix seeded with autologous fibroblasts improves wound breaking strength in a rodent soft tissue damage model in neoadjuvant settings. J Biomater Appl 25:413–427. doi:10.1177/0885328209347961
Roessner ED, Vitacolonna M, Hohenberger P (2012) Confocal laser scanning microscopy evaluation of an acellular dermis tissue transplant (Epiflex(R)). PLOS ONE 7:e45991. doi:10.1371/journal.pone.0045991
Roessner E, Vitacolonna M, Schulmeister A, Pilz L, Tsagogiorgas C, Brockmann M, Hohenberger P (2013) Human acellular dermis seeded with autologous fibroblasts enhances bronchial anastomotic healing in an irradiated rodent sleeve resection model. Ann Surg Oncol 20(Suppl 3):S709–S715. doi:10.1245/s10434-013-3209-x
Rossner E et al (2011) Epiflex((R)) a new decellularised human skin tissue transplant: manufacture and properties. Cell Tissue Bank 12:209–217. doi:10.1007/s10561-010-9187-3
Rouwkema J, de Boer J, Van Blitterswijk CA (2006) Endothelial cells assemble into a 3-dimensional prevascular network in a bone tissue engineering construct. Tissue Eng 12:2685–2693. doi:10.1089/ten.2006.12.2685
Rouwkema J, Rivron NC, van Blitterswijk CA (2008) Vascularization in tissue engineering. Trends Biotechnol 26:434–441. doi:10.1016/j.tibtech.2008.04.009
Salazar AB, McAlister VC, Gupta R, MacDonald AS (2001) Circulating endothelial cells after transplantation. Lancet 357:1450
Salem HK, Thiemermann C (2010) Mesenchymal stromal cells: current understanding and clinical status. Stem Cells 28:585–596. doi:10.1002/stem.269
Sanz L et al (2008) Long-term in vivo imaging of human angiogenesis: critical role of bone marrow-derived mesenchymal stem cells for the generation of durable blood vessels. Microvasc Res 75:308–314. doi:10.1016/j.mvr.2007.11.007
Schindelin J et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. doi:10.1038/nmeth.2019
Schumann P et al (2009) Consequences of seeded cell type on vascularization of tissue engineering constructs in vivo. Microvasc Res 78:180–190. doi:10.1016/j.mvr.2009.06.003
Schumann P et al (2014) Accelerating the early angiogenesis of tissue engineering constructs in vivo by the use of stem cells cultured in matrigel. J Biomed Mater Res Part A 102:1652–1662. doi:10.1002/jbm.a.34826
Semont A et al (2006) Mesenchymal stem cells increase self-renewal of small intestinal epithelium and accelerate structural recovery after radiation injury. Adv Exp Med Biol 585:19–30
Shepherd BR, Enis DR, Wang F, Suarez Y, Pober JS, Schechner JS (2006) Vascularization and engraftment of a human skin substitute using circulating progenitor cell-derived endothelial cells. FASEB J 20:1739–1741. doi:10.1096/fj.05-5682fje
Shin L, Peterson DA (2013) Human mesenchymal stem cell grafts enhance normal and impaired wound healing by recruiting existing endogenous tissue stem/progenitor cells. Stem Cells Transl Med 2:33–42. doi:10.5966/sctm.2012-0041
Singh S, Wu BM, Dunn JC (2011) Accelerating vascularization in polycaprolactone scaffolds by endothelial progenitor cells. Tissue Eng Part A 17:1819–1830. doi:10.1089/ten.TEA.2010.0708
Stratman AN, Davis GE (2012) Endothelial cell-pericyte interactions stimulate basement membrane matrix assembly: influence on vascular tube remodeling, maturation, and stabilization. Microsc Microanal 18:68–80. doi:10.1017/S1431927611012402
Suh W et al (2005) Transplantation of endothelial progenitor cells accelerates dermal wound healing with increased recruitment of monocytes/macrophages and neovascularization. Stem Cells 23:1571–1578. doi:10.1634/stemcells.2004-0340
Takahashi T et al (1999) Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat Med 5:434–438. doi:10.1038/7434
Tigges U, Welser-Alves JV, Boroujerdi A, Milner R (2012) A novel and simple method for culturing pericytes from mouse brain. Microvasc Res 84:74–80. doi:10.1016/j.mvr.2012.03.008
Vater C, Kasten P, Stiehler M (2011) Culture media for the differentiation of mesenchymal stromal cells. Acta Biomater 7:463–477. doi:10.1016/j.actbio.2010.07.037
Vitacolonna M, Belharazem D, Hohenberger P, Roessner ED (2013) Effect of static seeding methods on the distribution of fibroblasts within human acellular dermis. Biomed Eng Online 12:55. doi:10.1186/1475-925X-12-55
Vitacolonna M et al (2014) Effect on the tensile strength of human acellular dermis (Epiflex(R)) of in vitro incubation simulating an open abdomen setting. BMC Surg 14:7. doi:10.1186/1471-2482-14-7
Vitacolonna M, Belharazem D, Hohenberger P, Roessner ED (2015a) Effect of dynamic seeding methods on the distribution of fibroblasts within human acellular dermis. Cell Tissue Bank. doi:10.1007/s10561-015-9508-7
Vitacolonna M, Belharazem D, Maier P, Hohenberger P, Roessner ED (2015b) In vivo quantification of the effects of radiation and presence of hair follicle pores on the proliferation of fibroblasts in an acellular human dermis in a dorsal skinfold chamber: relevance for tissue reconstruction following neoadjuvant therapy. PLOS ONE 10:e0125689. doi:10.1371/journal.pone.0125689
Watt SM, Gullo F, van der Garde M, Markeson D, Camicia R, Khoo CP, Zwaginga JJ (2013) The angiogenic properties of mesenchymal stem/stromal cells and their therapeutic potential. Br Med Bull 108:25–53. doi:10.1093/bmb/ldt031
Wenger A, Kowalewski N, Stahl A, Mehlhorn AT, Schmal H, Stark GB, Finkenzeller G (2005) Development and characterization of a spheroidal coculture model of endothelial cells and fibroblasts for improving angiogenesis in tissue engineering. Cells Tissues Organs 181:80–88. doi:10.1159/000091097
Wu Y, Chen L, Scott PG, Tredget EE (2007) Mesenchymal stem cells enhance wound healing through differentiation and angiogenesis. Stem Cells 25:2648–2659. doi:10.1634/stemcells.2007-0226
Yagi H, Soto-Gutierrez A, Parekkadan B, Kitagawa Y, Tompkins RG, Kobayashi N, Yarmush ML (2010) Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cell Transpl 19:667–679. doi:10.3727/096368910X508762
Yamamoto M, James D, Li H, Butler J, Rafii S, Rabbany S (2010) Generation of stable co-cultures of vascular cells in a honeycomb alginate scaffold. Tissue Eng Part A 16:299–308. doi:10.1089/ten.TEA.2009.0010
Yang M, Chen CZ, Shu YS, Shi WP, Cheng SF, Gu YJ (2012) Preseeding of human vascular cells in decellularized bovine pericardium scaffold for tissue-engineered heart valve: an in vitro and in vivo feasibility study. J Biomed Mater Res Part B Appl Biomater 100:1654–1661. doi:10.1002/jbm.b.32734
Yoshikawa T et al (2008) Wound therapy by marrow mesenchymal cell transplantation. Plast Reconstr Surg 121:860–877. doi:10.1097/01.prs.0000299922.96006.24
Zammaretti P, Zisch AH (2005) Adult ‘endothelial progenitor cells’. Renewing vasculature. Int J Biochem Cell Biol 37:493–503. doi:10.1016/j.biocel.2004.06.018
Zhang L, Chan C (2010) Isolation and enrichment of rat mesenchymal stem cells (MSCs) and separation of single-colony derived MSCs. J Vis Exp. doi:10.3791/1852
Acknowledgements
The authors thank Dr. Jan C. Brune and Dr. Mark D. Smith from the German Institute for Cell and Tissue Replacement (DIZG) for advice relating to use of Epiflex® and for assistance with data analysis and editing of the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Mario Vitacolonna, Dr. Djeda Belharazem, Prof. Dr. Peter Hohenberger and Dr. Eric Roessner declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Vitacolonna, M., Belharazem, D., Hohenberger, P. et al. In-vivo quantification of the revascularization of a human acellular dermis seeded with EPCs and MSCs in co-culture with fibroblasts and pericytes in the dorsal chamber model in pre-irradiated tissue. Cell Tissue Bank 18, 27–43 (2017). https://doi.org/10.1007/s10561-016-9606-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-016-9606-1