Abstract
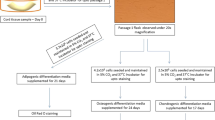
Mesenchymal stem cells (MSCs) though multipotent exhibit limited lifespan in vitro, with progressive reduction in capacity for self-renewal leading to irreversible arrest of cell division, which limits their use for therapeutic purposes. Human umbilical cord wall MSCs are easy to process and proliferate rapidly in culture, but variability of individual samples and impact upon in vitro expansion and aging processes is unknown. We compared isolation protocols to determine which one yields the highest number of viable cells with the best proliferation capacity. Three different protocols were tested: two were enzymatic procedures and one explant method. Isolated cells were evaluated in terms of proliferation, differentiation capacity, and phenotype. All samples were processed using one or more protocols. After passage 2 adherent cells displayed standard phenotypic and differentiation characteristics of MSCs, but our results show that isolating cells directly from Wharton’s jelly is more advantageous. Cells obtained from explants presented similar characteristics to those from enzymatic protocols, but always reached proliferation arrest earlier, irrespective of initial population doubling times. From the same sample, cells obtained with enzymatic protocol ii reached later passages while exhibiting shorter doubling times in culture than cells from other protocols, that is, took longer to reach senescence. More important, each individual MSC sample exhibited different population doubling rates and reached senescence at different passages, irrespective of protocol. Thus, even when in strict conformity with procedures and quality control, each cord sample shows a unique behavior, a finding that should be taken into account when planning for therapeutic approaches.





Similar content being viewed by others
References
Al-Nbaheen M, Vishnubalaji R, Ali D, Bouslimi A, Al-Jassir F, Megges M, Prigione A, Adjaye J, Kassem M, Aldahmash A (2013) Human stromal (mesenchymal) stem cells from bone marrow, adipose tissue and skin exhibit differences in molecular phenotype and differentiation potential. Stem Cell Rev 9:32–43
Badraiq H, Devito L, Ilic D (2014) Isolation and expansion of mesenchymal stromal/stem cells from umbilical cord under chemically defined conditions. Methods Mol Biol 1283:65–71
Can A, Karahuseyinoglu S (2007) Concise review: human umbilical cord stroma with regard to the source of fetus-derived stem cells. Stem Cells 25:2886–2895
Carrade DD, Borjesson DL (2013) Immunomodulation by mesenchymal stem cells in veterinary species. Comp Med 63:207–217
Chatzistamatiou TK, Papassavas AC, Michalopoulos E, Gamaloutsos C, Mallis P, Gontika I, Panagouli E, Koussoulakos SL, Stavropoulos-Giokas C (2014) Optimizing isolation culture and freezing methods to preserve Wharton’s jelly’s mesenchymal stem cell (MSC) properties: an MSC banking protocol validation for the hellenic cord blood bank. Transfusion 54(12):3108–3120
Conget PA, Minguell JJ (1999) Phenotypical and functional properties of human bone marrow mesenchymal progenitor cells. J Cell Physiol 181:67–73
Covas DT, Siufi JL, Silva AR, Orellana MD (2003) Isolation and culture of umbilical vein mesenchymal stem cells. Braz J Med Biol Res Revista brasileira de pesquisas medicas e biologicas/Sociedade Brasileira de Biofisica [et al] 36:1179–1183
Cristofalo VJ, Allen RG, Pignolo RJ, Martin BG, Beck JC (1998) Relationship between donor age and the replicative lifespan of human cells in culture: a reevaluation. Proc Natl Acad Sci USA 95:10614–10619
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8:315–317
Fong CY, Chak LL, Biswas A, Tan JH, Gauthaman K, Chan WK, Bongso A (2011) Human Wharton’s jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev 7:1–16
Fu YS, Cheng YC, Lin MY, Cheng H, Chu PM, Chou SC, Shih YH, Ko MH, Sung MS (2006) Conversion of human umbilical cord mesenchymal stem cells in Wharton’s jelly to dopaminergic neurons in vitro: potential therapeutic application for Parkinsonism. Stem Cells 24:115–124
Gazit R, Weissman IL, Rossi DJ (2008) Hematopoietic stem cells and the aging hematopoietic system. Semin Hematol 45:218–224
Gonzalez R, Griparic L, Umana M, Burgee K, Vargas V, Nasrallah R, Silva F, Patel A (2010) An efficient approach to isolation and characterization of pre- and postnatal umbilical cord lining stem cells for clinical applications. Cell Transplant 19:1439–1449
Ishige I, Nagamura-Inoue T, Honda MJ, Harnprasopwat R, Kido M, Sugimoto M, Nakauchi H, Tojo A (2009) Comparison of mesenchymal stem cells derived from arterial, venous, and Wharton’s jelly explants of human umbilical cord. Int J Hematol 90:261–269
Itahana K, Dimri G, Campisi J (2001) Regulation of cellular senescence by p53. Eur J Biochem FEBS 268:2784–2791
Jaffe EA, Nachman RL, Becker CG, Minick CR (1973) Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Investig 52:2745–2756
Lu LL, Liu YJ, Yang SG, Zhao QJ, Wang X, Gong W, Han ZB, Xu ZS, Lu YX, Liu D et al (2006) Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials. Haematologica 91:1017–1026
Mori Y, Ohshimo J, Shimazu T, He H, Takahashi A, Yamamoto Y, Tsunoda H, Tojo A, Nagamura-Inoue T (2014) Improved explant method to isolate umbilical cord-derived mesenchymal stem cells and their immunosuppressive properties. Tissue Eng Part C Methods 21(4):367–372
Panepucci RA, Siufi JL, Silva WA Jr et al (2004) Comparison of gene expression of umbilical cord vein and bone marrow-derived mesenchymal stem cells. Stem Cells 22:1263–1278
Patel AN, Genovese J (2011) Potential clinical applications of adult human mesenchymal stem cell (Prochymal(R)) therapy. Stem Cells Cloning Adv Appl 4:61–72
Reinisch A, Strunk D (2009) Isolation and animal serum free expansion of human umbilical cord derived mesenchymal stromal cells (MSCs) and endothelial colony forming progenitor cells (ECFCs). J Vis Exp (32):1525
Romanov YA, Svintsitskaya VA, Smirnov VN (2003) Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord. Stem Cells 21:105–110
Rossant J (2001) Stem cells in the mammalian blastocyst. Harvey Lect 97:17–40
Sarugaser R, Hanoun L, Keating A, Stanford WL, Davies JE (2009) Human mesenchymal stem cells self-renew and differentiate according to a deterministic hierarchy. PLoS One 4:e6498
Seshareddy K, Troyer D, Weiss ML (2008) Method to isolate mesenchymal-like cells from Wharton’s Jelly of umbilical cord. Methods Cell Biol 86:101–119
Sobolewski K, Malkowski A, Bankowski E, Jaworski S (2005) Wharton’s jelly as a reservoir of peptide growth factors. Placenta 26:747–752
Swamynathan P, Venugopal P, Kannan S, Thej C, Kolkundar U, Bhagwat S, Ta M, Majumdar AS, Balasubramanian S (2014) Are serum-free and xeno-free culture conditions ideal for large scale clinical grade expansion of Wharton’s jelly derived mesenchymal stem cells? A comparative study. Stem Cell Res Therapy 5:88
Tong CK, Vellasamy S, Tan BC, Abdullah M, Vidyadaran S, Seow HF, Ramasamy R (2011) Generation of mesenchymal stem cell from human umbilical cord tissue using a combination enzymatic and mechanical disassociation method. Cell Biol Int 35:221–226
Troyer DL, Weiss ML (2008) Wharton’s jelly-derived cells are a primitive stromal cell population. Stem Cells 26:591–599
Wagner W, Horn P, Castoldi M, Diehlmann A, Bork S, Saffrich R, Benes V, Blake J, Pfister S, Eckstein V et al (2008) Replicative senescence of mesenchymal stem cells: a continuous and organized process. PLoS One 3:e2213
Wagner J, Kean T, Young R, Dennis JE, Caplan AI (2009) Optimizing mesenchymal stem cell-based therapeutics. Curr Opin Biotechnol 20:531–536
Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ, Fu YS, Lai MC, Chen CC (2004) Mesenchymal stem cells in the Wharton’s jelly of the human umbilical cord. Stem Cells 22:1330–1337
Acknowledgments
We would like to acknowledge the contribution of Maria Alves, Isis Mozetic, and Érica Moreira that have helped make this study possible. We thank Andreia Kondo and the team of nurses at our local Public Umbilical Cord Blood Bank for their help to obtain the samples. We thank Dr. Luiz Sardinha, Dr. Luciana Marti, and Dr. Andrea Sertié for expert technical assistance and insightful suggestions. UNIEMP and CAPES have financed graduate fellowships. Anna Carla Goldberg is a recipient of personal fellowship from CNPq. We are indebted to UC donors and acknowledge the generous support by the Ruhman family.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Paladino, F.V., Peixoto-Cruz, J.S., Santacruz-Perez, C. et al. Comparison between isolation protocols highlights intrinsic variability of human umbilical cord mesenchymal cells. Cell Tissue Bank 17, 123–136 (2016). https://doi.org/10.1007/s10561-015-9525-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-015-9525-6