Abstract
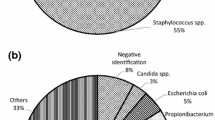
To assess the effectiveness of antimicrobial treatment by using cool decontamination protocol with low concentration of antibiotics during processing of cardiovascular allografts, 948 allografts processed during a 2-year period were analysed. Five hundred and fourty one donors aged <62 years were classified in: multiorgan donors (MOD) with non-transplantable hearts; recipients of cardiac transplantation (RHT); and non-beating heart cadavers with a warm ischemic time of less than 6 h (NBHD). During processing three samples for bacteriology testing were taken A (sampling before decontamination); B (sampling after decontamination); C (sampling on the final product). Samples A were positive in 348 cases (36.4%), respectively 36% for MOD, 21.6% for RHT and 78.1% for NBHD. All the allografts were immersed in a cocktail of four antibiotics at 4 °C. After exposure to antibiotics the rate of decontamination of those with A positive was 90.4, 92.5, 82.5% respectively for MOD, RHT, NBHD. At the end of processing, 57 allografts (6%) were positive in B and/or C, 15 allografts remained contaminated with the same bacteria as in A, 42 were contaminated during processing. The overall rate of sterility in the end of processing is 94% and for each group this is: 95.4% for MOD, 96.8% for RHT and 86.3% for NBHD. Analysis shows that there is no influence of time of exposure in AB in the rate of decontamination for MOD and RHT. The most predominant germ in contamination is Coagulase Negative Staphylococcus (CNS) (53.4% alone, 8.9% with other bacteria). 83.3% of MOD; 88.5% of RHT were contaminated with one germ, while 40.4% of NBHD were contaminated with more than one.
Similar content being viewed by others
References
Ault A 2004. US FDA increases scrutiny of tissue banks. Lancet 363(9406): 377.
Barratt-Boyes B. G. and Roche A. H. 1969. A review of aortic valve homografts over a six and one-half year period. Ann Surg. 170(3): 483–492.
Barratt-Boyes B. G., Roche A. H., Subramanyan R., Pemberton J. R. and Whitlock R. M. 1987. Long-term follow-up of patients with the antibiotic-sterilized aortic homograft valve inserted freehand in the aortic position. Circulation 75(4): 768–777.
Brockbank K. G. and Dawson P. E. 1993. Citotoxicity of amphotericin B for broblasts in human heart valve lea. ets. Cryobiology 30(1): 19–24.
Deijkers R. L. M., Bloem R. M., Petit P. L. C., Brand R., Veh-meyer S. B. W. and Veen M. R. 1994. Contamination of bone allografts: analysis of incidence and predominant factors. J. Bone Joint Surg. Br. 79: 161–166.
Gall K., Smith S., Willmette C., Wong M. and O' Brien M. 1995. Allograft heart valve sterilization: a six-year in-depth analysis of a twenty-ve-year experience with low-dose anti-biotics. J. Thorac. Cardiovasc. Surg. 110(3): 680–787.
Go. n Y., Grandmougin D. and Van Hoeck B. 1996. Banking cryopreserved heart valves in Europe: assessment of a 5-year operation in an international tissue bank in Brussels. Eur. J. Cardiothorac. Surg. 10(7): 505–512.
Go. n Y. A., Van Hoeck B., Jashari R., Soots G. and Kalmar P. 2000. Banking of cryopreserved heart valves in Europe: assessment of a 10-year operation in the European Homo-graft Bank (EHB). J. Heart Valve Dis. 9(2): 207–214.
Hu J. F., Gilmer L., Hopkins R. and Wol nbarger L. Jr. 1989. Effects of antibiotics on cellular viability in porcine heart valve tissue. Cardiovasc. Res. 23(11): 960–964.
Khanna S. K., Ross J. K. and Monro J. L. 1981. Homograft aortic valve replacement: seven years' experience with anti-biotic-treated valves. Thorax. May 36(5): 330–337.
Kirklin J. K., Smith D., Novick W., Naftel D. C., Kirklin J. W., Paci co A. D., Nanda N. C., Helmcke F. R. and Bourge R. C. 1993. Long-term function of cryopreserved aortic homografts. A ten-year study. J. Thorac. Cardiovasc. Surg. 106(1): 154–165.
Martinez O. V., Malinin T. I., Valla P. H. and Flores A. 1985. Postmortem bacteriology of cadaver tissue donors: an evaluation of blood cultures as an index of tissue sterility. Diagn. Microbiol. Infect. Dis. 3(3): 193–200.
Mulligan M. S., Tsai T. T., Kneebone J. M., Ward P. A. and Lupinetti F. M. 1994. E. ects of preservation techniques on in vivo expression of adhesion molecules by aortic valve allografts. J. Thorac. Cardiovasc. Surg. 107(3): 717–23.
O' Brien M. F., Sta. ord E. G., Gardner M. A., Pohlner P. G., Tesar P. J., Cochrane A. D., Mau T. K., Gall K. L. and Smith S. E. 1995. Allograft aortic valve replacement: long-term follow-up. Ann Thorac Surg. 60 (Suppl): S65–70.
Schmehl M. K., Bank H. L. and Brockbank K. G. 1993. E. ects of antibiotics on the endothelium of fresh and cryopreserved canine saphenous veins. Cryobiology 30(2): 164–171.
Strickett M. G., Barratt-Boyes B. G. and MacCulloch D. 1983. Disinfection of human heart valve allografts with antibiotics in low concentration. Pathology 15(4): 457–462.
Wain W. H., Pearce H. M., Riddell R. W. and Ross D. N. 1977. A re-evaluation of antibiotic sterilisation of heart valve allografts. Thorax 32(6): 740–742.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tabaku, M., Jashari, R., Carton, H. et al. Processing of cardiovascular allografts: effectiveness of European Homograft Bank (EHB) antimicrobial treatment (cool decontamination protocol with low concentration of antibiotics). Cell Tissue Banking 5, 261–266 (2004). https://doi.org/10.1007/s10561-004-1440-1
Issue Date:
DOI: https://doi.org/10.1007/s10561-004-1440-1