Abstract
Purpose
The glucose-lowering drug metformin has recently been shown to reduce myocardial oxygen consumption and increase myocardial efficiency in chronic heart failure (HF) patients without diabetes. However, it remains to be established whether these beneficial myocardial effects are associated with metformin-induced alterations in whole-body insulin sensitivity and substrate metabolism.
Methods
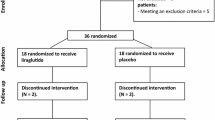
Eighteen HF patients with reduced ejection fraction and without diabetes (median age, 65 (interquartile range 55–68); ejection fraction 39 ± 6%; HbA1c 5.5 to 6.4%) were randomized to receive metformin (n = 10) or placebo (n = 8) for 3 months. We studied the effects of metformin on whole-body insulin sensitivity using a two-step hyperinsulinemic euglycemic clamp incorporating isotope-labeled tracers of glucose, palmitate, and urea. Substrate metabolism and skeletal muscle mitochondrial respiratory capacity were determined by indirect calorimetry and high-resolution respirometry, and body composition was assessed by bioelectrical impedance analysis. The primary outcome measure was change in insulin sensitivity.
Results
Compared with placebo, metformin treatment lowered mean glycated hemoglobin levels (absolute mean difference, − 0.2%; 95% CI − 0.3 to 0.0; p = 0.03), reduced body weight (− 2.8 kg; 95% CI − 5.0 to − 0.6; p = 0.02), and increased fasting glucagon levels (3.2 pmol L−1; 95% CI 0.4 to 6.0; p = 0.03). No changes were observed in whole-body insulin sensitivity, endogenous glucose production, and peripheral glucose disposal or oxidation with metformin. Equally, resting energy expenditure, lipid and urea turnover, and skeletal muscle mitochondrial respiratory capacity remained unaltered.
Conclusion
Increased myocardial efficiency during metformin treatment is not mediated through improvements in insulin action in HF patients without diabetes.
Clinical Trial Registration
URL: https://clinicaltrials.gov. Unique identifier: NCT02810132. Date of registration: June 22, 2016.



Similar content being viewed by others
Availability of Data and Material
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Bertero E, Maack C. Metabolic remodelling in heart failure. Nat Rev Cardiol. 2018;15:457–70. https://doi.org/10.1038/s41569-018-0044-6.
Larsen AH, Jessen N, Nørrelund H, Tolbod LP, Harms HJ, Feddersen S, et al. A randomized, double-blind, placebo-controlled trial of metformin on myocardial efficiency in insulin resistant chronic heart failure patients without diabetes. Eur J Heart Fail. 2019;(in press). https://doi.org/10.1002/EJHF.1656.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HAW. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577–89.
Gormsen LC, Sondergaard E, Christensen NL, Brosen K, Jessen N, Nielsen S. Metformin increases endogenous glucose production in non-diabetic individuals and individuals with recent-onset type 2 diabetes. Diabetologia. 2019;62:1251–6. https://doi.org/10.1007/s00125-019-4872-7.
Johnson AB, Webster JM, Sum CF, Heseltine L, Argyraki M, Cooper BG, et al. The impact of metformin therapy on hepatic glucose production and skeletal muscle glycogen synthase activity in overweight type II diabetic patients. Metabolism. 1993;42(9):1217–22.
Duca FA, Côté CD, Rasmussen BA, Zadeh-Tahmasebi M, Rutter GA, Filippi BM, et al. Metformin activates a duodenal Ampk-dependent pathway to lower hepatic glucose production in rats. Nat Med. 2015;21(5):506–11.
Madiraju AK, Erion DM, Rahimi Y, Zhang XM, Braddock DT, Albright RA, et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature. 2014;510(7506):542–6. https://doi.org/10.1038/nature13270.
Shaw RJ, Lamia KA, Vasquez D, Koo S-H, Bardeesy N, DePinho RA, et al. The kinase LKB1 mediates glucose homeostasis in liver and therapeutic effects of metformin. Science. 2005;310(5754):1642–6.
Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest. 2001;108(8):1167–74. https://doi.org/10.1172/jci13505.
Owen MR, Doran E, Halestrap AP. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. The Biochemical journal. 2000;348(Pt 3):607–14.
Hunter RW, Hughey CC, Lantier L, Sundelin EI, Peggie M, Zeqiraj E, et al. Metformin reduces liver glucose production by inhibition of fructose-1-6-bisphosphatase. Nat Med. 2018;24(9):1395–406. https://doi.org/10.1038/s41591-018-0159-7.
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403. https://doi.org/10.1056/NEJMoa012512.
Preiss D, Lloyd SM, Ford I, McMurray JJ, Holman RR, Welsh P, et al. Metformin for non-diabetic patients with coronary heart disease (the CAMERA study): a randomised controlled trial. Lancet Diabetes Endocrinol. 2014;2(2):116–24. https://doi.org/10.1016/s2213-8587(13)70152-9.
Wang XF, Zhang JY, Li L, Zhao XY, Tao HL, Zhang L. Metformin improves cardiac function in rats via activation of AMP-activated protein kinase. Clin Exp Pharmacol Physiol. 2011;38(2):94–101.
Cittadini A, Napoli R, Monti MG, Rea D, Longobardi S, Netti PA, et al. Metformin prevents the development of chronic heart failure in the SHHF rat model. Diabetes. 2012;61(4):944–53.
Wong AK, Symon R, AlZadjali MA, Ang DS, Ogston S, Choy A, et al. The effect of metformin on insulin resistance and exercise parameters in patients with heart failure. Eur J Heart Fail. 2012;14(11):1303–10.
Natali A, Ferrannini E. Effects of metformin and thiazolidinediones on suppression of hepatic glucose production and stimulation of glucose uptake in type 2 diabetes: a systematic review. Diabetologia. 2006;49(3):434–41. https://doi.org/10.1007/s00125-006-0141-7.
International Conference on Harmonisation Harmonised Tripartite Guideline for Good Clinical Practice (ICH-GCP), E6(R2), European Medicines Agency. 2016. https://www.ema.europa.eu/en/documents/scientific-guideline/ich-e-6-r2-guideline-good-clinical-practice-step-5_en.pdf. Accessed 8 July 2020.
DeFronzo RA, Tobin JD, Andres R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Phys. 1979;237(3):E214–23. https://doi.org/10.1152/ajpendo.1979.237.3.E214.
Krentz AJ, Heinemann L, Hompesch M. Translational research methods for diabetes, obesity and cardiometabolic drug development: a focus on early phase clinical studies. Springer; 2014.
Dollerup OL, Christensen B, Svart M, Schmidt MS, Sulek K, Ringgaard S, et al. A randomized placebo-controlled clinical trial of nicotinamide riboside in obese men: safety, insulin-sensitivity, and lipid-mobilizing effects. Am J Clin Nutr. 2018;108(2):343–53. https://doi.org/10.1093/ajcn/nqy132.
Pedersen MH, Svart MV, Lebeck J, Bidlingmaier M, Stodkilde-Jorgensen H, Pedersen SB, et al. Substrate metabolism and insulin sensitivity during fasting in obese human subjects: impact of GH blockade. J Clin Endocrinol Metab. 2017;102(4):1340–9. https://doi.org/10.1210/jc.2016-3835.
Moller N, Jorgensen JO, Schmitz O, Moller J, Christiansen J, Alberti KG, et al. Effects of a growth hormone pulse on total and forearm substrate fluxes in humans. Am J Phys. 1990;258(1 Pt 1):E86–91. https://doi.org/10.1152/ajpendo.1990.258.1.E86.
Steele R. Influences of glucose loading and of injected insulin on hepatic glucose output. Ann N Y Acad Sci. 1959;82:420–30.
Jahoor F, Wolfe RR. Reassessment of primed constant-infusion tracer method to measure urea kinetics. Am J Phys. 1987;252(4 Pt 1):E557–64. https://doi.org/10.1152/ajpendo.1987.252.4.E557.
Poulsen MM, Vestergaard PF, Clasen BF, Radko Y, Christensen LP, Stødkilde-Jørgensen H, et al. High-dose resveratrol supplementation in obese men. Diabetes. 2013;62(4):1186–95.
Jespersen NR, Yokota T, Stottrup NB, Bergdahl A, Paelestik KB, Povlsen JA, et al. Pre-ischaemic mitochondrial substrate constraint by inhibition of malate-aspartate shuttle preserves mitochondrial function after ischaemia–reperfusion. J Physiol. 2017;595(12):3765–80. https://doi.org/10.1113/jp273408.
Christiansen LB, Dela F, Koch J, Hansen CN, Leifsson PS, Yokota T. Impaired cardiac mitochondrial oxidative phosphorylation and enhanced mitochondrial oxidative stress in feline hypertrophic cardiomyopathy. Am J Physiol Heart Circ Physiol. 2015;308(10):H1237–47. https://doi.org/10.1152/ajpheart.00727.2014.
Ferrannini E. The theoretical bases of indirect calorimetry: a review. Metabolism. 1988;37(3):287–301.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9.
Inzucchi SE, Maggs DG, Spollett GR, Page SL, Rife FS, Walton V, et al. Efficacy and metabolic effects of metformin and troglitazone in type II diabetes mellitus. N Engl J Med. 1998;338(13):867–72. https://doi.org/10.1056/nejm199803263381303.
Kim YB, Ciaraldi TP, Kong A, Kim D, Chu N, Mohideen P, et al. Troglitazone but not metformin restores insulin-stimulated phosphoinositide 3-kinase activity and increases p110beta protein levels in skeletal muscle of type 2 diabetic subjects. Diabetes. 2002;51(2):443–8.
Rudvik A, Mansson M. Evaluation of surrogate measures of insulin sensitivity—correlation with gold standard is not enough. BMC Med Res Methodol. 2018;18(1):64. https://doi.org/10.1186/s12874-018-0521-y.
Konopka AR, Esponda RR, Robinson MM, Johnson ML, Carter RE, Schiavon M, et al. Hyperglucagonemia mitigates the effect of metformin on glucose production in prediabetes. Cell Rep. 2016;15(7):1394–400. https://doi.org/10.1016/j.celrep.2016.04.024.
Christensen MM, Hojlund K, Hother-Nielsen O, Stage TB, Damkier P, Beck-Nielsen H, et al. Endogenous glucose production increases in response to metformin treatment in the glycogen-depleted state in humans: a randomised trial. Diabetologia. 2015;58(11):2494–502. https://doi.org/10.1007/s00125-015-3733-2.
Hother-Nielsen O, Schmitz O, Andersen PH, Beck-Nielsen H, Pedersen O. Metformin improves peripheral but not hepatic insulin action in obese patients with type II diabetes. Acta Endocrinol. 1989;120(3):257–65.
Prager R, Schernthaner G, Graf H. Effect of metformin on peripheral insulin sensitivity in non insulin dependent diabetes mellitus. Diabetes Metab. 1986;12(6):346–50.
Hallsten K, Virtanen KA, Lonnqvist F, Sipila H, Oksanen A, Viljanen T, et al. Rosiglitazone but not metformin enhances insulin- and exercise-stimulated skeletal muscle glucose uptake in patients with newly diagnosed type 2 diabetes. Diabetes. 2002;51(12):3479–85.
Karlsson HK, Hallsten K, Bjornholm M, Tsuchida H, Chibalin AV, Virtanen KA, et al. Effects of metformin and rosiglitazone treatment on insulin signaling and glucose uptake in patients with newly diagnosed type 2 diabetes: a randomized controlled study. Diabetes. 2005;54(5):1459–67.
Stumvoll M, Nurjhan N, Perriello G, Dailey G, Gerich JE. Metabolic effects of metformin in non-insulin-dependent diabetes mellitus. N Engl J Med. 1995;333(9):550–4. https://doi.org/10.1056/nejm199508313330903.
Zierath JR, Galuska D, Nolte LA, Thorne A, Kristensen JS, Wallberg-Henriksson H. Effects of glycaemia on glucose transport in isolated skeletal muscle from patients with NIDDM: in vitro reversal of muscular insulin resistance. Diabetologia. 1994;37(3):270–7.
Kelley DE, Mandarino LJ. Fuel selection in human skeletal muscle in insulin resistance: a reexamination. Diabetes. 2000;49(5):677–83.
Roden M, Perseghin G, Petersen KF, Hwang JH, Cline GW, Gerow K, et al. The roles of insulin and glucagon in the regulation of hepatic glycogen synthesis and turnover in humans. J Clin Invest. 1996;97(3):642–8. https://doi.org/10.1172/jci118460.
Miller RA, Chu Q, Xie J, Foretz M, Viollet B, Birnbaum MJ. Biguanides suppress hepatic glucagon signalling by decreasing production of cyclic AMP. Nature. 2013;494(7436):256–60. https://doi.org/10.1038/nature11808.
Hernandez-Cascales J. Does glucagon have a positive inotropic effect in the human heart? Cardiovasc Diabetol. 2018;17(1):148. https://doi.org/10.1186/s12933-018-0791-z.
Aroda VR, Knowler WC, Crandall JP, Perreault L, Edelstein SL, Jeffries SL, et al. Metformin for diabetes prevention: insights gained from the diabetes prevention program/diabetes prevention program outcomes study. Diabetologia. 2017;60(9):1601–11. https://doi.org/10.1007/s00125-017-4361-9.
Malin SK, Kashyap SR. Effects of metformin on weight loss: potential mechanisms. Curr Opin Endocrinol Diabetes Obes. 2014;21(5):323–9. https://doi.org/10.1097/med.0000000000000095.
DeFronzo RA, Barzilai N, Simonson DC. Mechanism of metformin action in obese and lean noninsulin-dependent diabetic subjects. J Clin Endocrinol Metab. 1991;73(6):1294–301. https://doi.org/10.1210/jcem-73-6-1294.
Tschritter O, Fritsche A, Thamer C, Haap M, Shirkavand F, Rahe S, et al. Plasma adiponectin concentrations predict insulin sensitivity of both glucose and lipid metabolism. Diabetes. 2003;52(2):239–43. https://doi.org/10.2337/diabetes.52.2.239.
Acknowledgments
We are grateful for the excellent assistance provided by the medical laboratory technicians at the Medical Research Laboratory and at the Department of Cardiology, Aarhus University Hospital, Denmark.
Funding
This work was supported by the Danish Diabetes Academy supported by the Novo Nordisk Foundation (OL8201 to A.H.L. and H.W.), the Danish Council for Independent Research (4183-00384 to N.J.), NNF-Diabetes Excellence Project (NNF13OC0003882 to N.J.), the Arvid Nilsson Foundation (Dok 1780631 to A.H.L.), the Faculty of Health at Aarhus University (phd0115 to A.H.L.), the Danish Heart Foundation (15-R99-A5947-22932; 15-R99-A5947-22933 to A.H.L. and H.W.), Kirsten Anthonius Memorial Fund (to A.H.L.), the Health Research Fund of Central Denmark Region (to A.H.L.), the Augustinus Foundation (15-4919 to A.H.L.), the Aase and Ejnar Danielsen Foundation (10-001657 to A.H.L.), the Helga and Peter Korning Foundation (55027-100 to A.H.L.), and the Hede Nielsen Foundation (to A.H.L.).
Author information
Authors and Affiliations
Contributions
N.J., H.W., H.N., K.B., J.F., N.M., and A.H.L. contributed to the study conception and/or design. Material preparation, data collection, and analysis were performed by A.H.L. The first draft of the manuscript was written by A.H.L. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
Merck KGaA (Darmstadt, Germany) provided the metformin (Glucophage XR®) and placebo tablets free of charge. Merck KGaA reviewed the manuscript for medical accuracy only before journal submission. The authors are fully responsible for the content of this manuscript, and the views and opinions described in the publication reflect solely those of the authors. H.W. has been the principal or a sub-investigator in studies involving the following pharmaceutical companies: MSD, Bayer, Daiichi-Sankyo, Novartis, Novo Nordisk, Sanofi-Aventis, and Pfizer. The other authors report no conflicts.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 119 kb)
Rights and permissions
About this article
Cite this article
Larsen, A.H., Wiggers, H., Dollerup, O.L. et al. Metformin Lowers Body Weight But Fails to Increase Insulin Sensitivity in Chronic Heart Failure Patients without Diabetes: a Randomized, Double-Blind, Placebo-Controlled Study. Cardiovasc Drugs Ther 35, 491–503 (2021). https://doi.org/10.1007/s10557-020-07050-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-07050-5