Abstract
Purpose
Chronic kidney disease (CKD) associates with inflammatory and prothrombotic phenotypes, resulting in higher cardiovascular risk. Factor Xa displays functions beyond coagulation, exhibiting proinflammatory effects. The aim of the present study was to investigate whether a direct FXa inhibitor protects from the endothelial dysfunction (ED) caused by uremia.
Methods
Macro (HUVEC) and microvascular (HMEC) endothelial cells (ECs) were exposed to serum from uremic patients or healthy donors, in absence and presence of apixaban (60 ng/ml). We evaluated changes in surface VCAM-1 and ICAM-1, intracellular eNOS, reactive oxygen species (ROS), and von Willebrand Factor (VWF) production by immunofluorescence, reactivity of the extracellular matrix (ECM) towards platelets, and intracellular signaling.
Results
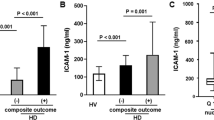
ECs exposed to uremic serum triggered dysregulation of all the parameters. Presence of apixaban resulted in decreased expression of VCAM-1 (178 ± 14 to 89 ± 2% on HMEC and 324 ± 71 to 142 ± 25% on HUVEC) and ICAM-1 (388 ± 60 to 111 ± 10% on HMEC and 148 ± 9% to 90 ± 7% on HUVEC); increased eNOS (72 ± 8% to 95 ± 10% on HMEC); normalization of ROS levels (173 ± 21 to 114 ± 13% on HMEC and 165 ± 14 to 127 ± 7% on HUVEC); lower production of VWF (168 ± 14 to 92 ± 4% on HMEC and 151 ± 22 to 99 ± 11% on HUVEC); and decreased platelet adhesion onto ECM (134 ± 22 to 93 ± 23% on HMEC and 161 ± 14 to 117 ± 7% on HUVEC). Apixaban inhibited p38MAPK and p42/44 activation in HUVEC (139 ± 15 to 48 ± 15% and 411 ± 66 to 177 ± 57%, respectively) (p < 0.05 vs control for all parameters).
Conclusion
Anti-FXa strategies, such as apixaban, prevented ED caused by the uremic milieu, exhibiting anti-inflammatory and antioxidant properties and modulating the reactivity of the ECM.





Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author, M.D-R, upon reasonable request.
Abbreviations
- FXa:
-
Activated factor X
- DOACs:
-
Direct oral anticoagulants
- PARs:
-
Protease-activated receptors
- ECs:
-
Endothelial cells
- ED:
-
Endothelial dysfunction
- CKD:
-
Chronic kidney disease
- ESRD:
-
End-stage renal disease
- ECM:
-
Extracellular matrix
- HUVECs:
-
Human umbilical vein endothelial cells
- HMEC-1:
-
Human dermal microvascular endothelial cells
- C:
-
Control condition
- U:
-
Uremic condition
- A:
-
Apixaban treatment
References
Aird WC. Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ Res. 2007;100:158–73.
Vanhoutte PM, Shimokawa H, Feletou M, Tang EHC. Endothelial dysfunction and vascular disease – a 30th anniversary update. Acta Physiol. 2017;219:22–96.
Cines DB, Pollak ES, Buck CA, Loscalzo J, Zimmerman GA, McEver RP, et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood. 1998;91:3527–61.
Shi Y, Vanhoutte PM. Macro- and microvascular endothelial dysfunction in diabetes. J Diabetes. 2017;9:434–49.
Huxley VH, Kemp SS, Schramm C, Sieveking S, Bingaman S, Yu Y, et al. Sex differences influencing micro- and macrovascular endothelial phenotype in vitro. J Physiol. 2018;596:3929–49.
Serradell M, Díaz-Ricart M, Cases A, Zurbano MJ, López-Pedret J, Arranz O, et al. Uremic medium causes expression, redistribution and shedding of adhesion molecules in cultured endothelial cells. Haematologica. 2002;87:1053–61.
Hanzu FA, Palomo M, Kalko SG, Parrizas M, Garaulet M, Escolar G, et al. Translational evidence of endothelial damage in obese individuals: inflammatory and prothrombotic responses. J Thromb Haemost. 2011;9:1236–45.
Martin-Rodriguez S, Caballo C, Gutierrez G, Vera M, Cruzado JM, Cases A, et al. TLR4 and NALP3 inflammasome in the development of endothelial dysfunction in uraemia. Eur J Clin Invest. 2015;45:160–9.
Carbó C, Arderiu G, Escolar G, Fusté B, Cases A, Carrascal M, et al. Differential expression of proteins from cultured endothelial cells exposed to uremic versus normal serum. Am J Kidney Dis. 2008;51:603–12.
Gómez-Outes A, Suárez-Gea ML, Lecumberri R, Terleira-Fernández AI, Vargas-Castrillón E. Direct-acting oral anticoagulants: pharmacology, indications, management, and future perspectives. Eur J Haematol. 2015;95:389–404.
Busch G, Seitz I, Steppich B, Hess S, Eckl R, Schömig A, et al. Coagulation factor Xa stimulates interleukin-8 release in endothelial cells and mononuclear leukocytes: implications in acute myocardial infarction. Arterioscler Thromb Vasc Biol. 2005;25:461–6.
Borensztajn K, Peppelenbosch MP, Spek CA. Coagulation factor Xa signaling: the link between coagulation and inflammatory bowel disease? Trends Pharmacol Sci. 2009;30:8–16.
Coughlin SR. Thrombin signalling and protease-activated receptors. Nature. 2000;407:258–64.
Esmon CT. Targeting factor Xa and thrombin: impact on coagulation and beyond. Thromb Haemost. 2013;111:625–33.
Granger CB, Alexander JH, McMurray JJV, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981–92.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883–91.
Hart RG, Diener HC, Yang S, Connolly SJ, Wallentin L, Reilly PA, et al. Intracranial hemorrhage in atrial fibrillation patients during anticoagulation with warfarin or dabigatran: The RE-LY trial. Stroke. 2012;43:1511–7.
Borissoff JI, Otten JJT, Heeneman S, Leenders P, van Oerle R, Soehnlein O, et al. Genetic and pharmacological modifications of thrombin formation in apolipoprotein E-deficient mice determine atherosclerosis severity and atherothrombosis onset in a neutrophil-dependent manner. PLoS One. 2013;8:e55784.
Ellinghaus P, Perzborn E, Hauenschild P, Gerdes C, Heitmeier S, Visser M, et al. Expression of pro-inflammatory genes in human endothelial cells: Comparison of rivaroxaban and dabigatran. Thromb Res. 2016;142:44–51.
Spronk HMH, De Jong AM, Crijns HJ, Schotten U, Van Gelder IC, Ten Cate H. Pleiotropic effects of factor Xa and thrombin: what to expect from novel anticoagulants. Cardiovasc Res. 2014;101:344–51.
Lutz J, Menke J, Sollinger D, Schinzel H, Thürmel K. Haemostasis in chronic kidney disease. Nephrol Dial Transplant. 2014;29:29–40.
Siontis KC, Zhang X, Eckard A, Bhave N, Schaubel DE, He K, et al. Outcomes associated with apixaban use in patients with end-stage kidney disease and atrial fibrillation in the United States. Circulation. 2018;138:1519–29.
Frost C, Nepal S, Wang J, Schuster A, Byon W, Boyd RA, et al. Safety, pharmacokinetics and pharmacodynamics of multiple oral doses of apixaban, a factor Xa inhibitor, in healthy subjects. Br J Clin Pharmacol. 2013;76:776–86.
Pujadas-Mestres L, Lopez-Vilchez I, Arellano-Rodrigo E, Reverter JC, Lopez-Farre A, Diaz-Ricart M, et al. Differential inhibitory action of apixaban on platelet and fibrin components of forming thrombi: studies with circulating blood and in a platelet-based model of thrombin generation. PLoS One. 2017;12:e0171486.
Serradell M, Díaz-Ricart M, Cases A, Zurbano MJ, Aznar-Salatti J, López-Pedret J, et al. Uremic medium disturbs the hemostatic balance of cultured human endothelial cells. Thromb Haemost. 2001;86:1099–105.
Lassen MR, Davidson BL, Gallus A, Pineo G, Ansell J, Deitchman D. The efficacy and safety of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement. J Thromb Haemost. 2007;5:2368–75.
Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS, Johnson M, et al. Apixaban for extended treatment of venous thromboembolism. N Engl J Med. 2013;368:699–708.
Duffy A, Liew A, O’Sullivan J, Avalos G, Samali A, O’Brien T. Distinct effects of high-glucose conditions on endothelial cells of macrovascular and microvascular origins. Endothelium. 2006;13:9–16.
Yannoutsos A, Levy BI, Safar ME, Slama G, Blacher J. Pathophysiology of hypertension: interactions between macro and microvascular alterations through endothelial dysfunction. J Hypertens. 2014;32:216–24.
Sandoo A, Carroll D, Metsios GS, Kitas GD, Veldhuijzen van Zanten JJCS. The association between microvascular and macrovascular endothelial function in patients with rheumatoid arthritis: a cross-sectional study. Arthritis Res Ther. 2011;13:R99.
Himmelfarb J. Uremic toxicity, oxidative stress, and hemodialysis as renal replacement therapy. Semin Dial. 2009;22:636–43.
Vera M, Torramade-Moix S, Martin-Rodriguez S, Cases A, Cruzado JM, Rivera J, et al. Antioxidant and anti-inflammatory strategies based on the potentiation of glutathione peroxidase activity prevent endothelial dysfunction in chronic kidney disease. Cell Physiol Biochem. 2018;51:1287–300.
Poulianiti KP, Kaltsatou A, Mitrou GI, Jamurtas AZ, Koutedakis Y, Maridaki M, et al. Systemic redox imbalance in chronic kidney disease: a systematic review. Oxid Med Cell Longev. 2016;2016:1–19.
Ancedy Y, Berthelot E, Lang S, Ederhy S, Boyer-Chatenet L, Di Angelantonio E, et al. Is von Willebrand factor associated with stroke and death at mid-term in patients with non-valvular atrial fibrillation? Arch Cardiovasc Dis. 2018;111:357–69.
Borgel D, Bianchini E, Lasne D, Pascreau T, Saller F. Inflammation in deep vein thrombosis: a therapeutic target? Hematology. 2019;24:742–50.
Puech C, Delavenne X, He Z, Forest V, Mismetti P, Perek N. Direct oral anticoagulants are associated with limited damage of endothelial cells of the blood-brain barrier mediated by the thrombin/PAR-1 pathway. Brain Res. 1719;2019:57–63.
Borensztajn K, Spek CA. Blood coagulation factor Xa as an emerging drug target. Expert Opin Ther Targets. 2011;15:341–9.
Schurgers LJ, Spronk HMH. Differential cellular effects of old and new oral anticoagulants: consequences to the genesis and progression of atherosclerosis. Thromb Haemost. 2014;112:909–17.
Papadaki S, Tselepis AD. Nonhemostatic activities of factor Xa: are there pleiotropic effects of anti-FXa direct oral anticoagulants? Angiology. 2019;70:896–907.
Lerman A, Zeiher AM. Endothelial function: cardiac events. Circulation. 2005;111:363–8.
Mabley J, Patel JP, Sayed A, Arya R, Scutt G. Direct oral anticoagulant (DOAC)-mediated vasodilation: role of nitric oxide. Thromb Res. 2019;176:36–8.
Villari A, Giurdanella G, Bucolo C, Drago F, Salomone S. Apixaban enhances vasodilatation mediated by protease-activated receptor 2 in isolated rat arteries. Front Pharmacol. 2017;8:480.
Álvarez E, Paradela-Dobarro B, Raposeiras-Roubín S, González-Juanatey JR. Protective, repairing and fibrinolytic effects of rivaroxaban on vascular endothelium. Br J Clin Pharmacol. 2018;84:280–91.
Pham PT, Fukuda D, Yagi S, Kusunose K, Yamada H, Soeki T, et al. Rivaroxaban, a specific FXa inhibitor, improved endothelium-dependent relaxation of aortic segments in diabetic mice. Sci Rep. 2019;9:11206.
Wu TC, Chan JS, Lee CY, Leu HB, Huang PH, Chen JS, et al. Rivaroxaban, a factor Xa inhibitor, improves neovascularization in the ischemic hindlimb of streptozotocin-induced diabetic mice. Cardiovasc Diabetol. 2015;14:81.
Bonaca MP, Bauersachs RM, Anand SS, Debus ES, Nehler MR, Patel MR, et al. Rivaroxaban in peripheral artery disease after revascularization. N Engl J Med. 2020;NEJMoa2000052.
Acknowledgments
We would like to thank the Primary Hemostasis laboratory group from Hospital Clínic de Barcelona for their technical support. We would also like to thank the collaboration of the nurses from Hospital de la Maternitat from Barcelona for collecting and providing us with human umbilical cords from which we obtained primary endothelial cell cultures.
Funding
This study was partially supported by Bristol Myers-Squibb (ERISTA 15), German José Carreras Leukaemia Foundation (grant 11R/2016), Integrated Project in Health Institutes (PIE15/00027), Technology Development Projects in Health 2016 (DTS/00133) and FIS PI19/00888 from the Instituto de Salud Carlos III, Spanish Government; 2017-SGR671 from Generalitat de Catalunya, and CERCA Programme of the Generalitat de Catalunya. CIBEREHD is funded by Instituto de Salud Carlos III.
Author information
Authors and Affiliations
Contributions
S Torramade-Moix and M Palomo designed the research, performed the experiments, analyzed the results, and wrote the paper. M Vera, D Jerez-Dolz, and AB Moreno performed the experiments and analyzed the results. MU Zafar, J Rovira, and F Diekmann revised the manuscript. JC Garcia-Pagan, G Escolar, A Cases, and M Diaz-Ricart designed the research and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
S.T-M., M.P., M.V., D.J-D., A.B.M-C, M.U.Z., and J.R. have nothing to disclose. F.D. has received lecture fees from Alexion, Chiesi, Novartis, Mallinckrodt, Pfizer, Transplant Biomedical, and Neovii; travel support from Chiesi and Novartis; and grants from TEVA, Chiesi, Pfizer, Novartis, Astellas, and Transplant Biomedical. J.C.G-P. has received speaker fees from GORE, grants from Novartis and Theravance, and has been part of advisory board for Shionogi and Cook. G.E. has received honoraria/consultant fees from Bayer, Bristol Myers-Squibb, Boehringer Ingelheim, CSL Behring, Novo Nordisk, and Pfizer. A.C. has been part of the advisory board for Daiichi Sankyo, has received grants from Bristol Myers-Squibb, and has had lecture fees from Bayer, Bristol Myers-Squibb, and Boehringer. M.D-R. has received grants from Bristol Myers-Squibb and Jazz Pharmaceuticals, and lecture fees from JAZZ pharmaceuticals and Siemens Healthineers.
Ethics Approval
All patients gave their written informed consent to participate in the study, and the study was approved by the Hospital Clinic Ethical Committee of Clinical Investigation.
Consent to Participate
Written informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Code Availability
Density of labeling was calculated by computerized morphometric analysis using image processing and analysis in Java (ImageJ, National Institutes of Health, Bethesda, MD, USA).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Summary Table
What is known on this topic:
- Targetable factor Xa is involved in thrombogenicity, inflammation, and cellular remodeling.
- Endothelial cells in culture exposed to uremic sera extracted from chronic kidney disease patients exhibit a proinflammatory and prothrombotic phenotype
What this paper adds:
- Apixaban exhibits anti-inflammatory and antioxidant properties on the endothelium under uremic conditions.
- Apixaban modulates uremia-induced endothelial damage.
Rights and permissions
About this article
Cite this article
Torramade-Moix, S., Palomo, M., Vera, M. et al. Apixaban Downregulates Endothelial Inflammatory and Prothrombotic Phenotype in an In Vitro Model of Endothelial Dysfunction in Uremia. Cardiovasc Drugs Ther 35, 521–532 (2021). https://doi.org/10.1007/s10557-020-07010-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-07010-z