Abstract
Purpose
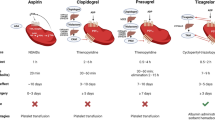
Drugs inhibiting the platelet P2Y12 receptor, such as clopidogrel and prasugrel, are potent antithrombotic agents and are widely used in cardiovascular disease. However, the adverse effects of these drugs have limited their clinical use. For example, clopidogrel resistance occurs in approximately one third of patients, while prasugrel increases the risk of major bleeding. Therefore, new generations of such drugs are of clinical interest.
Methods
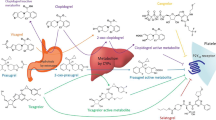
In this study, the pharmacodynamics of a new P2Y12 antagonist, CN-218, was compared with that of clopidogrel and prasugrel in rats and mice. The differences between CN-218 and clopidogrel include deuteration of the 7-position methyl carboxylate and the introduction of cinnamate in the 2-position of thiophene.
Results
CN-218 had an antiaggregatory efficacy that was at least five times more potent than that of clopidogrel but not as potent as that of prasugrel. It had a significant impact on activated partial thromboplastin time (APTT), whereby the APTT of CN-218-treated rats was approximately 9 s longer than that of the vehicle- or clopidogrel-treated group, while it had no impact on prothrombin time (PT) in rats. CN-218 had a similar potent antithrombotic effect to that of prasugrel and clopidogrel and also reduced the risk of bleeding compared to prasugrel.
Conclusion
CN-218 may be a promising antithrombotic agent, with potent antiplatelet and significant anticoagulant activity, as well as a lower risk of bleeding compared to clopidogrel and prasugrel.






Similar content being viewed by others
References
Yeung J, Li W, Holinstat M. Platelet signaling and disease: targeted therapy for thrombosis and other related diseases. Pharmacol Rev. 2018;70(3):526–48.
Cattaneo M. The platelet P2Y(1)(2) receptor for adenosine diphosphate: congenital and drug-induced defects. Blood. 2011;117(7):2102–12.
Born GV. Adenosine diphosphate as a mediator of platelet aggregation in vivo: an editorial view. Circulation. 1985;72(4):741–6.
Murugappa S, Kunapuli SP. The role of ADP receptors in platelet function. Front Biosci. 2006;11:1977–86.
Yeung J, Holinstat M. Newer agents in antiplatelet therapy: a review. J blood med. 2012;3:33–42.
Quinn MJ, Fitzgerald DJ. Ticlopidine and clopidogrel. Circulation. 1999;100(15):1667–72.
Miao J, Liu R, Li Z. Cytochrome P-450 polymorphisms and response to clopidogrel. N Engl J Med. 2009;360(21):2250–1.
Gremmel T, Panzer S. Clinical, genetic and confounding factors determine the dynamics of the in vitro response/non response to clopidogrel. Thromb Haemost. 2011;106(2):211–8.
Gachet C. P2Y(12) receptors in platelets and other hematopoietic and non-hematopoietic cells. Purinergic Signal. 2012;8(3):609–19.
Wallentin L, Becker RC, Budaj A, Cannon CP, Emanuelsson H, Held C, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361(11):1045–57.
Bhatt DL, Lincoff AM, Gibson CM, Stone GW, McNulty S, Montalescot G, et al. Intravenous platelet blockade with cangrelor during PCI. N Engl J Med. 2009;361(24):2330–41.
Shan J, Zhang B, Zhu Y, Jiao B, Zheng W, Qi X, et al. Overcoming clopidogrel resistance: discovery of vicagrel as a highly potent and orally bioavailable antiplatelet agent. J Med Chem. 2012;55(7):3342–52.
Lu N, Li L, Zheng X, Zhang S, Li Y, Yuan J et al. Synthesis of a Novel Series of Amino Acid Prodrugs Based on Thienopyridine Scaffolds and Evaluation of Their Antiplatelet Activity. Molecules (Basel, Switzerland). 2018;23(5).
Margaret Bradbury EH, Marantz Y, Baillie T, Stamler D. Site-specific deuterium substitution: A viable approach in drug discovery and development. Drug Met Pharmacokinet. 2018;34(1, Supplement):S8–9.
Xu X, Zhao X, Yang Z, Wang H, Meng X, Su C et al. Significant Improvement of Metabolic Characteristics and Bioactivities of Clopidogrel and Analogs by Selective Deuteration. Molecules (Basel, Switzerland). 2016;21(6).
Xu Z, Gu J, Gao M, Du N, Liu P, Xu X, et al. Study on antiplatelet effect of a new thiophenopyridine platelets P2Y12 receptor antagonist DV-127. Thromb Res. 2018;170:192–9.
Pontiki E, Peperidou A, Fotopoulos I, Hadjipavlou-Litina D. Cinnamate hybrids: a unique family of compounds with multiple biological activities. Curr Pharm Biotechnol. 2018;19(13):1019–48.
Born GV. Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature. 1962;194:927–9.
Bekemeier H, Hirschelmann R, Giessler AJ. Carrageenin-induced thrombosis in rats and mice: a model for testing antithrombotic substances? Agents actions. 1985;16(5):446–51.
Sanderson K. Big interest in heavy drugs. Nature. 2009;458(7236):269.
Hagihara K, Kazui M, Ikenaga H, Nanba T, Fusegawa K, Takahashi M, et al. Comparison of formation of thiolactones and active metabolites of prasugrel and clopidogrel in rats and dogs. Xenobiotica. 2009;39(3):218–26.
Qiu Z, Li N, Wang X, Tian F, Liu Q, Song L, et al. Pharmacokinetics of vicagrel, a promising analog of clopidogrel, in rats and beagle dogs. J Pharm Sci. 2013;102(2):741–9.
Sugidachi A, Asai F, Ogawa T, Inoue T, Koike H. The in vivo pharmacological profile of CS-747, a novel antiplatelet agent with platelet ADP receptor antagonist properties. Br J Pharmacol. 2000;129(7):1439–46.
Sugidachi A, Ogawa T, Kurihara A, Hagihara K, Jakubowski JA, Hashimoto M, et al. The greater in vivo antiplatelet effects of prasugrel as compared to clopidogrel reflect more efficient generation of its active metabolite with similar antiplatelet activity to that of clopidogrel's active metabolite. J Thromb Haemost. 2007;5(7):1545–51.
Wang KH, Li SF, Zhao Y, Li HX, Zhang LW. In Vitro Anticoagulant Activity and Active Components of Safflower Injection. Molecules (Basel, Switzerland). 2018;23(1).
Arrieta-Baez D, Dorantes-Alvarez L, Martinez-Torres R, Zepeda-Vallejo G, Jaramillo-Flores ME, Ortiz-Moreno A, et al. Effect of thermal sterilization on ferulic, coumaric and cinnamic acids: dimerization and antioxidant activity. J Sci Food Agric. 2012;92(13):2715–20.
Hadjipavlou-Litina D, Pontiki E. Aryl-acetic and cinnamic acids as lipoxygenase inhibitors with antioxidant, anti-inflammatory, and anticancer activity. Methods mol biology (Clifton, NJ). 2015;1208:361–77.
Guzman JD. Natural cinnamic acids, synthetic derivatives and hybrids with antimicrobial activity. Molecules (Basel, Switzerland). 2014;19(12):19292–349.
Lapeyre C, Delomenede M, Bedos-Belval F, Duran H, Negre-Salvayre A, Baltas M. Design, synthesis, and evaluation of pharmacological properties of cinnamic derivatives as antiatherogenic agents. J Med Chem. 2005;48(26):8115–24.
Kurup A, Kumar AV, Rao MN. Anti-inflammatory activity of cinnamic acids. Die Pharmazie. 1989;44(12):870.
Song F, Li H, Sun J, Wang S. Protective effects of cinnamic acid and cinnamic aldehyde on isoproterenol-induced acute myocardial ischemia in rats. J Ethnopharmacol. 2013;150(1):125–30.
Hagimori M, Kamiya S, Yamaguchi Y, Arakawa M. Improving frequency of thrombosis by altering blood flow in the carrageenan-induced rat tail thrombosis model. Pharmacol Res. 2009;60(4):320–3.
Acknowledgments
We thank Conn Hastings, PhD, from Liwen Bianji, Edanz Editing China (www.liwenbianji.cn/ac), for editing the English text of a draft of this manuscript.
Funding
This work was supported by the Health and Family Planning Commission of Jilin Province (No. 2017 J055) and the Education Department of Jilin Province (No. JJKH20190009KJ).
Author information
Authors and Affiliations
Contributions
Conceptualization of study, Xia Cao and Jinkai Gu; experimental design, Xiaohua Li, Peng Liu, and Zhiping Xu; performed the experiments, Xiaohua Li, Peng Liu, Zhiping Xu, Dong Sun, Yujia Miao, and Jujin Zhang; data analysis and interpretation, Xiaohua Li, Peng Liu, and Xia Cao; funding acquisition and supervision, Xia Cao; original manuscript draft preparation, Xiaohua Li; reviewing and editing the manuscript, Xiaohua Li. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This study was approved by the Animal Experiment Committee of Jilin University. Experimental mice and rats were housed with 12/12 h dark/light cycles and standard food and water under specific sterile (SPF) conditions. All mice and rats were provided with anesthesia and postoperative care.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, X., Liu, P., Xu, Z. et al. A Pharmacodynamic Study of CN-218, a Novel Antiplatelet and Antithrombotic Agent Primarily Targeting the P2Y12 Receptor. Cardiovasc Drugs Ther 34, 15–23 (2020). https://doi.org/10.1007/s10557-019-06930-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-019-06930-9