Abstract
Introduction
Irisin is a newly identified 112 amino acid hormone, derived as a product of fibronectin type III domain containing 5 (FNDC5), which is highly related to metabolic activity in skeletal muscle and brown fat. The effects of irisin on cardiovascular functions are unknown.
Purpose
To explore the effects of central and peripheral irisin on cardiovascular functions.
Methods
Irisin was either administrated into 3rd ventricle of rats or intravenously, and its effects on blood pressure and cardiac contractibility measured.
Results
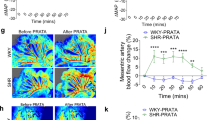
Administration of recombinant human irisin into the 3rd brain ventricle of rats activated neurons in the paraventricular nuclei of the hypothalamus. Central administration of irisin increased blood pressure and cardiac contractibility. Exogenous irisin reversed atenolol-induced inhibition of cardiac contractibility. In contrast, peripheral administration of irisin reduced blood pressure in both control and spontaneously hypertensive rats. Irisin dilated mesenteric artery rings through ATP-sensitive potassium channels.
Conclusion
Our studies indicate that central and peripheral irisin may differentially regulate cardiovascular activities.





Similar content being viewed by others
References
Bostrom P, Wu J, Jedrychowski MP, et al. A pgc1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8.
Brenmoehl J, Albrecht E, Komolka K, et al. Irisin is elevated in skeletal muscle and serum of mice immediately after acute exercise. Int J Biol Sci. 2014;10:338–49.
Norheim F, Langleite TM, Hjorth M, et al. The effects of acute and chronic exercise on pgc-1alpha, irisin and browning of subcutaneous adipose tissue in humans. FEBS J. 2014;281:739–49.
Hecksteden A, Wegmann M, Steffen A, et al. Irisin and exercise training in humans - results from a randomized controlled training trial. BMC Med. 2013;11:235.
Kraemer RR, Shockett P, Webb ND, Shah U, Castracane VD. A transient elevated irisin blood concentration in response to prolonged, moderate aerobic exercise in young men and women. Horm Metab Res. 2014;46:150–4.
Lee P, Linderman JD, Smith S, et al. Irisin and fgf21 are cold-induced endocrine activators of brown fat function in humans. Cell Metab. 2014;19:302–9.
Zhang Y, Li R, Meng Y, et al. Irisin stimulates browning of white adipocytes through mitogen-activated protein kinase p38 map kinase and erk map kinase signaling. Diabetes. 2014;63:514–25.
Lopez-Legarrea P, de la Iglesia R, Crujeiras AB, et al. Higher baseline irisin concentrations are associated with greater reductions in glycemia and insulinemia after weight loss in obese subjects. Nutr Diabetes. 2014;4:e110.
Sesti G, Andreozzi F, Fiorentino TV, et al. High circulating irisin levels are associated with insulin resistance and vascular atherosclerosis in a cohort of nondiabetic adult subjects. Acta Diabetol. 2014. doi:10.1007/s00592-014-0576-0.
Teufel A, Malik N, Mukhopadhyay M, Westphal H. Frcp1 and frcp2, two novel fibronectin type iii repeat containing genes. Gene. 2002;297:79–83.
Ferrer-Martinez A, Ruiz-Lozano P, Chien KR. Mouse pep: a novel peroxisomal protein linked to myoblast differentiation and development. Dev Dyn. 2002;224:154–67.
Dun SL, Lyu RM, Chen YH, Chang JK, Luo JJ, Dun NJ. Irisin-immunoreactivity in neural and non-neural cells of the rodent. Neuroscience. 2013;240:155–62.
Aydin S, Kuloglu T, Aydin S, et al. A comprehensive immunohistochemical examination of the distribution of the fat-burning protein irisin in biological tissues. Peptides. 2014;61C:130–6.
Hashemi MS, Ghaedi K, Salamian A, et al. Fndc5 knockdown significantly decreased neural differentiation rate of mouse embryonic stem cells. Neuroscience. 2013;231:296–304.
Moon HS, Dincer F, Mantzoros CS. Pharmacological concentrations of irisin increase cell proliferation without influencing markers of neurite outgrowth and synaptogenesis in mouse h19-7 hippocampal cell lines. Metabolism. 2013;62:1131–6.
Piya MK, Harte AL, Sivakumar K, et al. The identification of irisin in human cerebrospinal fluid: Influence of adiposity, metabolic markers, and gestational diabetes. Am J Physiol Endocrinol Metab. 2014;306:E512–8.
Walls EK, Wishart TB. Reliable method for cannulation of the third ventricle of the rat. Physiol Behav. 1977;19:171–3.
Li JY, Chai BX, Zhang W, Wang H, Mulholland MW. Expression of ankyrin repeat and suppressor of cytokine signaling box protein 4 (asb-4) in proopiomelanocortin neurons of the arcuate nucleus of mice produces a hyperphagic, lean phenotype. Endocrinology. 2010;151:134–42.
Zhang J, Villacorta L, Chang L, et al. Nitro-oleic acid inhibits angiotensin ii-induced hypertension. Circ Res. 2010;107:540–8.20.
Chang L, Villacorta L, Li R, et al. Loss of perivascular adipose tissue on peroxisome proliferator-activated receptor-gamma deletion in smooth muscle cells impairs intravascular thermoregulation and enhances atherosclerosis. Circulation. 2012;126:1067–78.
Rahmouni K, Morgan DA. Hypothalamic arcuate nucleus mediates the sympathetic and arterial pressure responses to leptin. Hypertension. 2007;49:647–52.
Mark AL, Rahmouni K, Correia M, Haynes WG. A leptin-sympathetic-leptin feedback loop: potential implications for regulation of arterial pressure and body fat. Acta Physiol Scand. 2003;177:345–9.
Correia ML, Morgan DA, Mitchell JL, Sivitz WI, Mark AL, Haynes WG. Role of corticotrophin-releasing factor in effects of leptin on sympathetic nerve activity and arterial pressure. Hypertension. 2001;38:384–8.
Pardridge WM. Recent developments in peptide drug delivery to the brain. Pharmacol Toxicol. 1992;71:3–10.25.
Timmons JA, Baar K, Davidsen PK, Atherton PJ. Is irisin a human exercise gene? Nature. 2012;488:E9–10. discussion E10-11.
Kurdiova T, Balaz M, Vician M, et al. Effects of obesity, diabetes and exercise on fndc5 gene expression and irisin release in human skeletal muscle and adipose tissue: in vivo and in vitro studies. J Physiol. 2014;592:1091–107.
Erickson HP. Irisin and fndc5 in retrospect: an exercise hormone or a transmembrane receptor? Adipocyte. 2012;2:289–93.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (81330010 and 81390354 to W.Z.), American Diabetes Association grant #1-13-BS-225 (to W.Z.), and the National Institute of Health grants 5R37DK043225 (to M. M.), HL105114 (to E. C.) and HL122664 (to L. C.).
Disclosures
None.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
(DOCX 365 kb)
Rights and permissions
About this article
Cite this article
Zhang, W., Chang, L., Zhang, C. et al. Central and Peripheral Irisin Differentially Regulate Blood Pressure. Cardiovasc Drugs Ther 29, 121–127 (2015). https://doi.org/10.1007/s10557-015-6580-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-015-6580-y