Abstract
Background
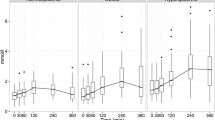
Hypertriglyceridemia subjects with metabolic syndrome exhibit variable postprandial triglyceride responses. We investigate the effects of fenofibrate therapy on postprandial triglyceride-containing lipoproteins in subjects with early (3.5 h) versus late (8 h) postprandial triglyceride responses.
Methods
Fifty-five subjects with fasting hypertriglyceridemia (≥1.7 mmol/L (150 mg/ dL) and <5.8 mmol/L (500 mg/dL)) and ≥2 Adult Treatment Panel III criteria of the metabolic syndrome were randomized to daily fenofibrate (160 mg/d) or placebo for 12 weeks in a double-blind controlled clinical trial. A standardized fat load (50 g/m2) was given orally after a 12 h fast. Blood specimens were obtained at 0 h (fasting), 3.5 h, and 8 h after the test meal. Analysis is confined to the 53 subjects with clearly identifiable early or late triglyceride peaks prior to therapy.
Results
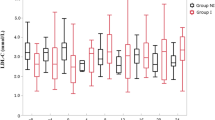
Fenofibrate was more effective in late peakers (n = 8) when compared to early peakers (n = 15) with respect to reducing postprandial triglyceride concentrations (−67% vs. −34%, p = 0.0024) and large VLDL (−76% vs. −31%, p = 0.0016), and increasing total HDL particles (20% vs. 11%, p = 0.008) and large HDL particles (185% vs. 88%, p = 0.003). On fenofibrate therapy, 100% of those initially designated as late peakers were reclassified as early peakers; 47% of late peakers assigned to placebo were reclassified as early peakers.
Conclusions
Late postprandial triglyceride responders have attenuated clearance of large VLDL particles, but they were more responsive to fenofibrate.





Similar content being viewed by others
References
Sarwar N, Danesh J, Eiriksdottir G, Sigurdsson G, Wareham N, Bingham S, et al. Triglycerides and the risk of coronary heart disease: 10.158 incident cases among 262, 525 participants in 29 Western prospective studies. Circulation. 2007;115:450–8.
Nordestgaard BG, Benn M, Schnohr P, Tybjaerg-Hansen A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA. 2007;298:299–308.
Stensvold I, Tverdal A, Urdal P, Graff-Iversen S. Non-fasting serum triglyceride concentration and mortality from coronary heart disease and any cause in middle aged Norwegian women. BMJ. 1993;307:1318–22.
Stampfer MJ, Krauss RM, Ma J, Blanche PJ, Holl LG, Sacks FM, et al. A prospective study of triglyceride level, low-density lipoprotein particle diameter, and risk of myocardial infarction. JAMA. 1996;276:882–8.
Iso H, Naito Y, Sato S, Kitamura A, Okamura T, Sankai T, et al. Serum triglycerides and risk of coronary heart disease among Japanese men and women. Am J Epidemiol. 2001;153:490–9.
Bansal S, Buring JE, Rifai N, Mora S, Sacks FM, Ridker PM. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA. 2007;298:309–16.
Triglyceride Coronary Disease Genetics Consortium and Emerging Risk Factors Collaboration. Triglyceride-mediated pathways and coronary disease: collaborative analysis of 101 studies. Lancet. 2010;375:1634–9.
Kofoed SC, Grønholdt M-LM, Bismuth J, Wilhjelm JE, Sillesen H, Nordestgaard BG. Echolucent, rupture-prone carotid plaques associated with elevated triglyceride-rich lipoproteins, particularly in women. J Vasc Surg. 2002;36:783–92.
Rosenson RS, Wolff DA, Huskin AL, Helenowski IB, Rademaker AW. Fenofibrate therapy ameliorates fasting and postprandial lipoproteinemia, oxidative stress, and the inflammatory response in subjects with hypertriglyceridemia and the metabolic syndrome. Diab Care. 2007;30:1945–51.
Executive Summary of The Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 285: 2486-2497.
Tangney CC, Hafner JM, McQuiston BD, Domas AJ, Rosenson RS. Postprandial changes in plasma and serum viscosity and plasma lipids and lipoproteins after an acute test meal. Am J Clin Nutr. 1997;65:36–40.
Jeyarajah EJ, Cromwell WC, Otvos JD. Lipoprotein particle analysis by nuclear magnetic resonance spectroscopy. Clin Lab Med. 2006;26:847–70.
Wallace T, Matthews D. The assessment of insulin resistance in man. Diabet Med. 2002;19:527–34.
Lai C-Q, Arnett DK, Corella D, Straka RJ, Tsai MY, Peacock JM, et al. Fenofibrate effect on triglyceride and postprandial response of apolipoprotein A5 Variants: The GOLDN Study. Arterioscler Thromb Vasc Biol. 2007;27:1417–25.
Lai C-Q, Corella D, Demissie S, Cupples LA, Adiconis X, Zhu Y, et al. Dietary intake of n-6 fatty acids modulates effect of apolipoprotein A5 gene on plasma fasting triglycerides, remnant lipoprotein concentrations, and lipoprotein particle size: The Framingham Heart Study. Circulation. 2006;113:2062–70.
Baass A, Dubuc G, Tremblay M, Delvin EE, O’Loughlin J, Levy E, et al. Plasma PCSK9 is associated with age, sex, and multiple metabolic markers in a population-based sample of children and adolescents. Clin Chem. 2009;55:1637–45.
Lambert G, Jarnoux A-L, Pineau T, Pape O, Chetiveaux M, Laboisse C, et al. Fasting induces hyperlipidemia in mice overexpressing proprotein convertase subtilisin kexin type 9: lack of modulation of very-low density lipoprotein hepatic output by the low-density lipoprotein receptor. Endocrinology. 2006;147:4985–95.
Dubuc G, Tremblay M, Pare G, Jacques H, Hamelin J, Benjannet S, et al. A new method for measurement of total plasma PCSK9: clinical applications. J Lipid Res. 2010;51:140–9.
Le May C, Kourimate S, Langhi C, Chetiveaux M, Jarry A, Comera C, et al. Proprotein convertase subtilisin kexin type 9 null mice are protected from postprandial triglyceridemia. Arterioscler Thromb Vasc Biol. 2009;29:684–90.
Kourimate S, Le May C, Langhi C, Jarnoux AL, Ouguerram K, Zair Y, et al. Dual mechanism for the fibrate-mediated repression of proprotein convertase subtilisn/kexin type 9. J Biol Chem. 2008;283:9666–73.
Lewis GF, Uffelman KD, Szeto LW, Weller B, Steiner G. Interaction between free fatty acids and insulin in the acute control of very low density lipoprotein production in humans. J Clin Invest. 1995;95:158–66.
Baldeweg SE, Golay A, Natali A, Balkau B, Del Prato S, Coppack SW. Insulin resistance, lipid and fatty acid concentrations in 867 healthy Europeans. European Group for the Study of Insulin Resistance (EGIR). Eur J Clin Invest. 2000;30:45–52.
McKeigue PM, Laws A, Chen YD, Marmot MG, Reaven GM. Relation of plasma triglyceride and apoB levels to insulin-mediated suppression of nonesterified fatty acids. Possible explanation for sex differences in lipoprotein pattern. Arterioscler Thromb. 1993;13:1187–92.
Laws A, Hoen HM, Selby JV, Saad MF, Haffner SM, Howard BV. Differences in insulin suppression of free fatty acid levels by gender and glucose tolerance status: relation to plasma triglyceride and apolipoprotein B concentrations. Arterioscler Thromb Vasc Biol. 1997;17:64–71.
Arii K, Suehiro T, Yamamoto M, Ito H, Hashimoto K. Suppression of plasma cholesteryl ester transfer protein activity in acute hyperinsulinemia and effect of plasma nonesterified fatty acid. Metabolism. 1997;46:1166–70.
Lagrost L, Florentin E, Guyard-Dangremont V, Athias A, Gandjini H, Lallemant C, et al. Evidence for nonesterified fatty acids as modulators of neutral lipid transfers in normolipidemic human plasma. Arterioscler Thromb Vasc Biol. 1995;15:1388–96.
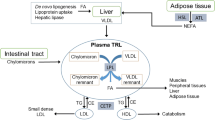
Grundy SM, Mok HY. Chylomicron clearance in normal and hyperlipidemic man. Metabolism. 1976;25:1225–39.
Acknowledgments
The author acknowledges Michael Ranella for manuscript preparation.
Funding Sources
This study was supported through a research grant to the University of Michigan and Northwestern University from Abbott Laboratories, Inc.
Disclosures
Dr. Rosenson has received funds from Abbott Laboratories, Inc. (honoraria payment, and consultant/advisory board) and LipoScience, Inc. (honoraria payment, ownership interest, and consultant/advisory board).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rosenson, R.S., Helenowski, I.B. & Tangney, C.C. Heterogeneous Postprandial Lipoprotein Responses in the Metabolic Syndrome, and Response to Fenofibrate Therapy. Cardiovasc Drugs Ther 24, 439–447 (2010). https://doi.org/10.1007/s10557-010-6264-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-010-6264-6