Abstract
Background
Reperfusion of the ischemic myocardium is associated with increased inflammatory processes that can exert deleterious effects and therefore contribute to cardiac dysfunction. The aim of the present study was to verify whether the administration of sTNFR-Fc, a scavenger of the pro-inflammatory cytokine TNF-α, at the time of reperfusion would protect against myocardial infarction and reduce the severity of early mechanical dysfunction.
Methods
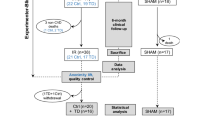
Male Wistar rats were subjected to 60 min coronary occlusion followed by reperfusion. A bolus of sTNFR-Fc (10 µg/kg, i.v.) (MI + sTNFR-Fc group) or a placebo (MI group) was injected prior to reperfusion. Cardiac geometry was assessed by echocardiography 1, 3 and 7 days after reperfusion. Eight days after reperfusion, left ventricular (LV) function was evaluated under basal conditions and during an experimental challenge of volume overload. Finally, infarct size was measured after euthanasia.
Results
sTNFR-Fc administration markedly reduced infarct size (P < 0.01) and decreased LV dilation as assessed by the echocardiographic measurement of the LV end diastolic area, 7 days post-MI (P < 0.01). Moreover, LV end-diastolic pressure was significantly preserved by sTNFR-Fc 1 week after myocardial infarction, under basal conditions (P < 0.05) as well as during cardiac overload (P < 0.05).
Conclusion
A single administration of sTNFR-Fc at the time of reperfusion after myocardial infarction is able to limit infarct size and to reduce early LV diastolic dysfunction in rats. These findings suggest that intravenous neutralization of TNF-α during surgical cardiac reperfusion might improve the outcome of myocardial infarction in humans.



Similar content being viewed by others
References
Frangogiannis NG, Smith CW, Entman ML. The inflammatory response in myocardial infarction. Cardiovasc Res 2002;53:31–47.
Jansen NJ, van Oeveren W, Gu YJ, van Vliet MH, Eijsman L, Wildevuur CR. Endotoxin release and tumor necrosis factor formation during cardiopulmonary bypass. Ann Thorac Surg 1992;54:744–7.
Maury CP, Teppo AM. Circulating tumour necrosis factor-alpha (cachectin) in myocardial infarction. J Intern Med 1989;225:333–6.
Torre-Amione G, Kapadia S, Lee J, et al. Tumor necrosis factor-alpha and tumor necrosis factor receptors in the failing human heart. Circulation 1996;93:704–11.
Gurevitch J, Frolkis I, Yuhas Y, et al. Tumor necrosis factor-alpha is released from the isolated heart undergoing ischemia and reperfusion. J Am Coll Cardiol 1996;28:247–52.
Herskowitz A, Choi S, Ansari AA, Wesselingh S. Cytokine mRNA expression in postischemic/reperfused myocardium. Am J Pathol 1995;146:419–28.
Moro C, Jouan MG, Rakotovao A, et al. Delayed expression of cytokines after reperfused myocardial infarction: possible trigger for cardiac dysfunction and ventricular remodelling. Am J Physiol Heart Circ Physiol 2007;293:H3014–9.
Schulz R, Aker S, Belosjorow S, Heusch G. TNFalpha in ischemia/reperfusion injury and heart failure. Basic Res Cardiol 2004;99:8–11.
Gurevitch J, Frolkis I, Yuhas Y, et al. Anti-tumor necrosis factor-alpha improves myocardial recovery after ischemia and reperfusion. J Am Coll Cardiol 1997;30:1554–61.
McVey M, Perrone MH, Clark KL. Does tumor necrosis factor-alpha (TNFalpha) contribute to myocardial reperfusion injury in anesthetized rats? Gen Pharmacol 1999;32:41–5.
Li D, Zhao L, Liu M, Du X, Ding W, Zhang J, et al. Kinetics of tumor necrosis factor alpha in plasma and the cardioprotective effect of a monoclonal antibody to tumor necrosis factor alpha in acute myocardial infarction. Am Heart J 1999;137:1145–52.
Gurantz D, Yndestad A, Halvorsen B, et al. Etanercept or intravenous immunoglobulin attenuates expression of genes involved in post-myocardial infarction remodeling. Cardiovasc Res 2005;67:106–15.
Berry MF, Woo YJ, Pirolli TJ, et al. Administration of a tumor necrosis factor inhibitor at the time of myocardial infarction attenuates subsequent ventricular remodeling. J Heart Lung Transplant 2004;23:1061–8.
Sugano M, Hata T, Tsuchida K, et al. Local delivery of soluble TNFalpha receptor 1 gene reduces infarct size following ischemia/reperfusion injury in rats. Mol Cell Biochem 2004;266:127–32.
Berthonneche C, Sulpice T, Boucher F, et al. New insights into the pathological role of TNF-alpha in early cardiac dysfunction and subsequent heart failure after infarction in rats. Am J Physiol Heart Circ Physiol 2004;287:H340–50.
Dawn B, Guo Y, Rezazadeh A, Wang OL, Stein AB, Hunt G, et al. Tumor necrosis factor-alpha does not modulate ischemia/reperfusion injury in naïve myocardium but is essential for the development of late preconditioning. J Mol Cell Cardiol 2004;37:51–61.
Labruto F, Yang J, Vaage J, Valen G. Role of tumor necrosis factor alpha and its receptor I in preconditioning by hyperoxia. Basic Res Cardiol 2005;100:198–207.
Skyschally A, Gres P, Hoffmann S, Haude M, Erbel R, Schulz R, et al. Bidirectional role of tumor necrosis factor-alpha in coronary microembolization: progressive contractile dysfunction versus delayed protection against infarction. Circ Res 2007;100:140–6.
Lecour S, Smith RM, Woodward B, Opie LH, Rochette L, Sack MN. Identification of a novel role for sphingolipid signaling in TNF alpha and ischemic preconditioning mediated cardioprotection. J Mol Cell Cardiol 2002;34:509–18.
Tanno M, Gorog DA, Bellahcene M, Cao X, Quinlan RA, Marber MS. Tumor necrosis factor-induced protection of the murine heart is independent of p38-MAPK activation. J Mol Cell Cardiol 2003;35:1523–7.
Sack M. Tumor necrosis factor-alpha in cardiovascular biology and the potential role for anti-tumor necrosis factor-alpha therapy in heart disease. Pharmacol Ther 2002;94:123–35.
Stampfer MJ, Goldhaber SZ, Yusuf S, Peto R, Hennekens CH. Effect of intravenous streptokinase on acute myocardial infarction: pooled results from randomized trials. N Engl J Med 1982;307:1180–2.
Stone GW, Brodie BR, Griffin JJ, et al. Prospective, multicenter study of the safety and feasibility of primary stenting in acute myocardial infarction: in-hospital and 30-day results of the PAMI stent pilot trial. Primary angioplasty in myocardial infarction stent pilot trial investigators. J Am Coll Cardiol 1998;31:23–30.
Mann DL. Inflammatory mediators and the failing heart: past, present, and the foreseeable future. Circ Res 2002;91:988–98.
Shames BD, Barton HH, Reznikov LL, et al. Ischemia alone is sufficient to induce TNFl-apha mRNA and peptide in the myocardium. Shock 2002;17:114–9.
Deswal A, Bozkurt B, Seta Y, et al. Safety and efficacy of a soluble P75 tumor necrosis factor receptor (Enbrel, etanercept) in patients with advanced heart failure. Circulation 1999;99:3224–6.
Bozkurt B, Kribbs SB, Clubb FJ Jr, et al. Pathophysiologically relevant concentrations of tumor necrosis factor-alpha promote progressive left ventricular dysfunction and remodeling in rats. Circulation 1998;97:1382–91.
Sivasubramanian N, Coker ML, Kurrelmeyer KM, et al. Left ventricular remodeling in transgenic mice with cardiac restricted overexpression of tumor necrosis factor. Circulation 2001;104:826–31.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Toufektsian, MC., Robbez-Masson, V., Sanou, D. et al. A Single Intravenous sTNFR-Fc Administration at the Time of Reperfusion Limits Infarct Size—Implications in Reperfusion Strategies in Man. Cardiovasc Drugs Ther 22, 437–442 (2008). https://doi.org/10.1007/s10557-008-6130-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-008-6130-y