Summary
Background: Ischemic preconditioning (IPC) elicits two distinct windows of cardioprotection, an early phase that lasts for 1–2 h and a delayed phase that lasts for 24–72 h. However, there is conflicting data as to how long the heart is resistant to IPC-induced cardioprotection after the initial protection wanes, leading to the demonstration of IPC-resistance. This resistance to IPC appears to be dependent on the timing of the next IPC stimulus, the species of animals used and the model studied. Furthermore, the mechanisms responsible IPC-resistance are unknown. It is also important to demonstrate therapeutic interventions that will produce cardioprotection during this period of IPC-resistance.
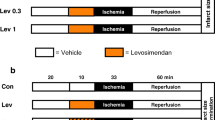
Methods and Results: To examine potential mechanisms responsible for acute IPC-induced resistance, the NHE-1 inhibitor EMD 85131 (2-methyl-5-methylsulfonyl-1-(1-pyrrollyl)-benzoylguanidine), which exerts its effects via mechanisms distinct from IPC, and the K ATP channel opener bimakalim, which bypasses the signaling mechanisms of IPC to directly open K ATP channels, were examined in a canine model of IPC-resistance. One 10 min. IPC stimulus followed by 10 min. of reperfusion produced a significant reduction in IS/AAR compared to Control (7.1 ± 2.6% versus 26.0 ± 6.2%; P < 0.05). However, IPC did not significantly protect the myocardium if a 2 h reperfusion period occurred between the initial IPC stimulus and the subsequent prolonged (60 min) ischemic challenge (IS/AAR: 22.5 ± 4.8%: P > 0.05). Furthermore, hearts treated with IPC followed by 2 h of reperfusion were resistant to an additional IPC stimulus administered just prior to the subsequent 60 min. occlusion period (IS/AAR: 22.9 ± 3.2%: P > 0.05). In contrast, administration of the NHE-1 inhibitor EMD 85131 (IS/AAR: 7.4 ± 2.5%: P < 0.05) or the K ATP channel opener bimakalim (IS/AAR: 11.8 ± 2.4%: P < 0.05) both afforded significant cardioprotection when administered at 2 h of reperfusion in previously preconditioned canine hearts resistant to IPC.
Conclusions: IPC resistance occurs in this canine model of ischemia-reperfusion injury. However, in spite of IPC resistance, hearts can still be pharmacologically protected by direct application of the K ATP channel opener bimakalim or the NHE inhibitor EMD 85131.
Similar content being viewed by others
Abbreviations
- NHE:
-
sodium/hydrogen exchanger
- LAD:
-
left anterior descending artery
- IS:
-
infarct size
- AAR:
-
area-at-risk
References
Jennings RB, Murry CE, Reimer KA. Preconditioning myocardium with ischemia. Cardiovasc Drugs Ther 1991;5(5):933–938.
Sun JZ, Tang XL, Park SW, Qiu Y, Turrens JF, Bolli R. Evidence for an essential role of reactive oxygen species in the genesis of late preconditioning against myocardial stunning in conscious pigs. J Clin Invest 1996;97(2):562–576.
Bolli R. Cardioprotective function of inducible nitric oxide synthase and role of nitric oxide in myocardial ischemia and preconditioning: An overview of a decade of research. J Mol Cell Cardiol 2001;33(11):1897–1918.
Meldrum DR, Cleveland JC, Jr, Rowland RT, Banerjee A, Harken AH, Meng X. Early and delayed preconditioning: Differential mechanisms and additive protection. Am J Physiol 1997;273(2 Pt 2):H725–H733.
Gumina RJ, Buerger E, Eickmeier C, Moore J, Daemmgen J, Gross GJ. Inhibition of the Na(+)/H(+) exchanger confers greater cardioprotection against 90 min. of myocardial ischemia than ischemic preconditioning in dogs. Circulation 1999;100(25):2519–2526; discussion 469–472.
Sack S, Mohri M, Arras M, Schwarz ER, Schaper W. Ischaemic preconditioning—time course of renewal in the pig. Cardiovasc Res 1993;27(4):551–555.
Kloner RA, Shook T, Przyklenk K, et al. Previous angina alters in-hospital outcome in TIMI 4. A clinical correlate to preconditioning? Circulation 1995;91(1):37–45.
Mizumura T, Nithipatikom K, Gross GJ. Bimakalim, an ATP-sensitive potassium channel opener, mimics the effects of ischemic preconditioning to reduce infarct size, adenosine release, and neutrophil function in dogs. Circulation 1995;92(5):1236–1245.
Yao Z, Gross GJ. Effects of the KATP channel opener bimakalim on coronary blood flow, monophasic action potential duration, and infarct size in dogs. Circulation 1994;89(4):1769–1975.
Gross GJ, Auchampach JA. Blockade of ATP-sensitive potassium channels prevents myocardial preconditioning in dogs. Circ Res 1992;70(2):223–233.
Gumina RJ, Mizumura T, Beier N, Schelling P, Schultz JJ, Gross GJ. A new sodium/hydrogen exchange inhibitor, EMD 85131, limits infarct size in dogs when administered before or after coronary artery occlusion. J Pharmacol Exp Ther 1998;286(1):175–183.
Gumina RJ, Beier N, Schelling P, Gross GJ. Inhibitors of ischemic preconditioning do not attenuate Na+/H+ exchange inhibitor mediated cardioprotection. J Cardiovasc Pharmacol 2000;35(6):949–953.
Sun JZ, Tang XL, Knowlton AA, Park SW, Qiu Y, Bolli R. Late preconditioning against myocardial stunning. An endogenous protective mechanism that confers resistance to postischemic dysfunction 24 h after brief ischemia in conscious pigs. J Clin Invest 1995;95(1):388–403.
Schwartz LM, Sebbag L, Jennings RB, Reimer KA. Duration and reinstatement of myocardial protection against infarction by ischemic preconditioning in open chest dogs. J Mol Cell Cardiol 2001;33(9):1561–1570.
Ytrehus K, Liu Y, Downey JM. Preconditioning protects ischemic rabbit heart by protein kinase C activation. Am J Physiol 1994;266(3 Pt 2):H1145–H1152.
Vahlhaus C, Schulz R, Post H, Rose J, Heusch G. Prevention of ischemic preconditioning only by combined inhibition of protein kinase C and protein tyrosine kinase in pigs. J Mol Cell Cardiol 1998;30(2):197–209.
Baines CP, Wang L, Cohen MV, Downey JM. Protein tyrosine kinase is downstream of protein kinase C for ischemic preconditioning's anti-infarct effect in the rabbit heart. J Mol Cell Cardiol 1998;30(2):383–392.
Maulik N, Yoshida T, Engelman RM, et al. Ischemic preconditioning attenuates apoptotic cell death associated with ischemia/reperfusion. Mol Cell Biochem 1998;186(1–2):139–145.
Ping P, Zhang J, Qiu Y, et al. Ischemic preconditioning induces selective translocation of protein kinase C isoforms epsilon and eta in the heart of conscious rabbits without subcellular redistribution of total protein kinase C activity. Circ Res 1997;81(3):404–414.
Ping P, Song C, Zhang J, et al. Formation of protein kinase C(epsilon)-Lck signaling modules confers cardioprotection. J Clin Invest 2002;109(4):499–507.
Kitakaze M, Funaya H, Minamino T, et al. Role of protein kinase C-alpha in activation of ecto-5′-nucleotidase in the preconditioned canine myocardium. Biochem Biophys Res Commun 1997;239(1):171–175.
Yoshida K, Kawamura S, Mizukami Y, Kitakaze M. Implication of protein kinase C-alpha, delta, and epsilon isoforms in ischemic preconditioning in perfused rat hearts. J Biochem (Tokyo) 1997;122(3):506–511.
Jin ZQ, Zhou HZ, Zhu P, et al. Cardioprotection mediated by sphingosine-1-phosphate and ganglioside GM-1 in wild-type and PKC epsilon knockout mouse hearts. Am J Physiol Heart Circ Physiol 2002;282(6):H1970–H1977.
Gumina RJ, Pucar D, Bast P, et al. Knockout of Kir6.2 negates ischemic preconditioning-induced protection of myocardial energetics. Am J Physiol Heart Circ Physiol 2003;284(6):H2106–H2113.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gumina, R.J., Schultz, J., Moore, J. et al. Cardioprotective-Mimetics Reduce Myocardial Infarct Size in Animals Resistant to Ischemic Preconditioning. Cardiovasc Drugs Ther 19, 315–322 (2005). https://doi.org/10.1007/s10557-005-3693-8
Issue Date:
DOI: https://doi.org/10.1007/s10557-005-3693-8