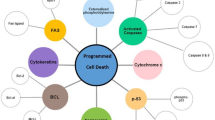
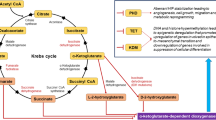
Abstract
Cellular plasticity refers to the ability of cells to change their identity or behavior, which can be advantageous in some cases (e.g., tissue regeneration) but detrimental in others (e.g., cancer metastasis). With a better understanding of cellular plasticity, the complexity of cancer cells, their heterogeneity, and their role in metastasis is being unraveled. The plasticity of the cells could also prove as a nemesis to their characterization. In this review, we have attempted to highlight the possibilities and benefits of using multiomics approach in characterizing the plastic nature of cancer cells. There is a need to integrate fragmented evidence at different levels of cellular organization (DNA, RNA, protein, metabolite, epigenetics, etc.) to facilitate the characterization of different forms of plasticity and cell types. We have discussed the role of cellular plasticity in generating intra-tumor heterogeneity. Different omics level evidence is being provided to highlight the variety of molecular determinants discovered using different techniques. Attempts have been made to integrate some of this information to provide a quantitative assessment and scoring of the plastic nature of the cells. However, there is a huge gap in our understanding of mechanisms that lead to the observed heterogeneity. Understanding of these mechanism(s) is necessary for finding targets for early detection and effective therapeutic interventions in metastasis. Targeting cellular plasticity is akin to neutralizing a moving target. Along with the advancements in precision and personalized medicine, these efforts may translate into better clinical outcomes for cancer patients, especially in metastatic stages.

Similar content being viewed by others
References
Hanahan, D. (2022). Hallmarks of cancer: New dimensions. Cancer Discovery, 12(1), 31–46. https://doi.org/10.1158/2159-8290.cd-21-1059
Hanahan, D., & Weinberg, R. A. (2011). Hallmarks of cancer: The next generation. Cell, 144(5), 646–674. https://doi.org/10.1016/j.cell.2011.02.013
Hanahan, D., & Weinberg, R. A. (2000). The hallmarks of cancer. Cell, 100(1), 57–70. https://doi.org/10.1016/s0092-8674(00)81683-9.
Mills, J. C., Stanger, B. Z., & Sander, M. (2019). Nomenclature for cellular plasticity: Are the terms as plastic as the cells themselves? The EMBO Journal, 38(19). https://doi.org/10.15252/embj.2019103148.
Gupta, P., Pastushenko, I., Skibinski, A., Blanpain, C., & Kuperwasser, C. (2019). Phenotypic plasticity: Driver of cancer initiation, progression, and therapy resistance. Cell Stem Cell, 24(1), 65–78. https://doi.org/10.1016/j.stem.2018.11.011.
Lambert, A. W., Pattabiraman, D. R., & Weinberg, R. A. (2017). Emerging biological principles of metastasis. Cell, 168(4), 670–691. https://doi.org/10.1016/j.cell.2016.11.037.
Deng, Z., Wu, S., Wang, Y., & Shi, D. (2022). Circulating tumor cell isolation for cancer diagnosis and prognosis. EBioMedicine, 83, 104237. https://doi.org/10.1016/j.ebiom.2022.104237.
Paget, S., The distribution of secondary growths in cancer of the breast. (1889). The Lancet, 133(3421), 571–573. https://doi.org/10.1016/s0140-6736(00)49915-0
Lüönd, F., Sugiyama, N., Bill, R., Bornes, L., Hager, C., Tang, F., Santacroce, N., Beisel, C., Ivánek, R., Bürglin, T. R., Tiede, S., Van Rheenen, J., & Christofori, G. (2021). Distinct contributions of partial and full EMT to breast cancer malignancy. Developmental Cell, 56(23), 3203–3221e11. https://doi.org/10.1016/j.devcel.2021.11.006.
Jehanno, C., Vulin, M., Richina, V., Richina, F., & Bentires-Alj, M. (2022). Phenotypic plasticity during metastatic colonization. Trends in Cell Biology, 32(10), 854–867. https://doi.org/10.1016/j.tcb.2022.03.007.
Pastushenko, I., Mauri, F., Song, Y., De Cock, F., Meeusen, B., Swedlund, B., Impens, F., Van Haver, D., Opitz, M., Théry, M., Barèche, Y., Lapouge, G., Vermeersch, M., Van Eycke, Y., Balsat, C., Decaestecker, C., Sokolow, Y., Hassid, S., Pérez-Bustillo, A., & Blanpain, C. (2020). Fat1 deletion promotes hybrid EMT state, tumour stemness and metastasis. Nature, 589(7842), 448–455. https://doi.org/10.1038/s41586-020-03046-1.
Massagué, J., & Ganesh, K. (2021). Metastasis-initiating cells and ecosystems. Cancer Discovery, 11(4), 971–994. https://doi.org/10.1158/2159-8290.cd-21-0010.
Kim, M. J., Huang, Y., & Park, J. (2020). Targeting WNT signaling for gastrointestinal cancer therapy: Present and evolving views. Cancers, 12(12), 3638. https://doi.org/10.3390/cancers12123638
Zhuang, X., Zhang, H., & Hu, G. (2019). Cancer and microenvironment plasticity: Double-edged swords in metastasis. Trends in Pharmacological Sciences, 40(6), 419–429. https://doi.org/10.1016/j.tips.2019.04.005
Yabo, Y. A., Niclou, S. P., & Golebiewska, A. (2021). Cancer cell heterogeneity and plasticity: A paradigm shift in glioblastoma. Neuro-oncology, 24(5), 669–682. https://doi.org/10.1093/neuonc/noab269.
Quintanal-Villalonga, Á., Chan, J. M., Yu, H. A., Pe’er, D., Sawyers, C. L., Sen, T., & Rudin, C. M. (2020). Lineage plasticity in cancer: A shared pathway of therapeutic resistance. Nature Reviews Clinical Oncology, 17(6), 360–371. https://doi.org/10.1038/s41571-020-0340-z.
Altieri, D. C. (2023). Mitochondria in cancer: Clean windmills or stressed tinkerers? Trends in Cell Biology, 33(4), 293–299. https://doi.org/10.1016/j.tcb.2022.08.001.
Yun, C. W., Jeon, J., Go, G., Lee, J. H., & Lee, S. H. (2020). The dual role of autophagy in cancer development and a therapeutic strategy for cancer by targeting autophagy. International Journal of Molecular Sciences, 22(1), 179. https://doi.org/10.3390/ijms22010179.
Luo, M., Ye, L., Chang, R., et al. (2022). Multi-omics characterization of autophagy-related molecular features for therapeutic targeting of autophagy. Nature Communications, 13, 6345. https://doi.org/10.1038/s41467-022-33946-x.
De Sousa, E., Melo, F., Vermeulen, L., Fessler, E., & Medema, J. P. (2013). Cancer heterogeneity—A multifaceted view. EMBO Reports, 14(8), 686–695. https://doi.org/10.1038/embor.2013.92
Culbert, S. J., Shuster, J. J., Land, V. J., Wharam, M. D., Thomas, P. R. M., Nitschke, R., Pinkel, D., & Vietti, T. J. (1991). Remission induction and continuation therapy in children with their first relapse of acute lymphoid leukemia. A pediatric oncology group study. Cancer, 67, 37–42. https://doi.org/10.1002/1097-0142(19910101)67:1<37::AID-CNCR2820670108>3.0.CO;2-#.
Kreso, A., & Dick, J. E. (2014). Evolution of the cancer stem cell model. Cell Stem Cell, 14(3), 275–291. https://doi.org/10.1016/j.stem.2014.02.006.
Iyer, D. N., Sin, W., & Ng, L. (2019). Linking stemness with colorectal cancer initiation, progression, and therapy. World Journal of Stem Cells, 11(8), 519–534. https://doi.org/10.4252/wjsc.v11.i8.519.
Pon, J. R., & Marra, M. A. (2015). Driver and passenger mutations in cancer. Annual Review of Pathology-mechanisms of Disease, 10(1), 25–50. https://doi.org/10.1146/annurev-pathol-012414-040312.
Kammenga, J. E. (2017). The background puzzle: How identical mutations in the same gene lead to different disease symptoms. FEBS Journal, 284, 3362–3373. https://doi.org/10.1111/febs.14080
Kosti, I., Jain, N., Aran, D., et al. (2016). Cross-tissue analysis of gene and protein expression in normal and cancer tissues. Scientific Reports, 6, 24799. https://doi.org/10.1038/srep24799
Shlyakhtina, Y., Moran, K. L., & Portal, M. M. (2021). Genetic and non-genetic mechanisms underlying cancer evolution. Cancers, 13(6), 1380. https://doi.org/10.3390/cancers13061380.
Dick, J. E. (2008). Stem cell concepts renew cancer research. Blood, 112(13), 4793–4807. https://doi.org/10.1182/blood-2008-08-077941.
Meacham, C. E., & Morrison, S. J. (2013). Tumour heterogeneity and cancer cell plasticity. Nature, 501(7467), 328–337. https://doi.org/10.1038/nature12624.
Nguyen, L. V., Vanner, R., Dirks, P. B., & Eaves, C. J. (2012). Cancer stem cells: An evolving concept. Nature Reviews Cancer, 12(2), 133–143. https://doi.org/10.1038/nrc3184.
Feinberg, A. P., Koldobskiy, M. A., & Göndör, A. (2016). Epigenetic modulators, modifiers and mediators in cancer aetiology and progression. Nature Reviews Genetics, 17(5), 284–299. https://doi.org/10.1038/nrg.2016.13.
Shiozawa, Y., Nie, B., Pienta, K. J., Morgan, T. M., & Taichman, R. S. (2013). Cancer stem cells and their role in metastasis. Pharmacology & Therapeutics, 138(2), 285–293. https://doi.org/10.1016/j.pharmthera.2013.01.014.
Eldai, H., Periyasamy, S., Qarni, S. A., Rodayyan, M. A., Mustafa, S., Deeb, A. M., Sheikh, E. A., Khan, M. A., Johani, M., Yousef, Z., & Aziz, M. (2013). Novel genes associated with colorectal cancer are revealed by high resolution cytogenetic analysis in a patient specific manner. PLOS ONE, 8(10), e76251. https://doi.org/10.1371/journal.pone.0076251
Pongor, L. S., Schultz, C. W., Rinaldi, L., Wangsa, D., Redon, C. E., Takahashi, N., Fialkoff, G., Desai, P., Zhang, Y., Burkett, S., Hermoni, N., Vilk, N., Gutin, J., Róna, G., Zhao, Y., Nichols, S., Vilimas, R., Sciuto, L., Graham, C., & Thomas, A. (2023). Extrachromosomal DNA amplification contributes to small cell lung cancer heterogeneity and is associated with worse outcomes. Cancer Discovery, 13(4), 928–949. https://doi.org/10.1158/2159-8290.cd-22-0796
Mealey, N. E., O’Sullivan, D. E., Pader, J., Ruan, Y., Wang, E., Quan, M. L., & Brenner, D. R. (2020). Mutational landscape differences between young-onset and older-onset breast cancer patients. Bmc Cancer, 20(1), 212. https://doi.org/10.1186/s12885-020-6684-z.
Evrony, G. D., Hinch, A. G., & Luo, C. (2021). Applications of Single-Cell DNA Sequencing. Annual Review of Genomics and Human Genetics, 22(1), 171–197.
Kong, S., Li, R., Tian, Y., Zhang, Y., Lu, Y., Ou, Q., Gao, P., Li, K., & Zhang, Y. (2023). Single-cell omics: A new direction for functional genetic research in human diseases and animal models. Frontiers in Genetics, 13, 1100016. https://doi.org/10.3389/fgene.2022.1100016.
Fan, X., Yang, C., Li, W., Bai, X., Zhou, X., Xie, H., et al. (2021). SMOOTH-Seq: Single-cell genome sequencing of human cells on a third-generation sequencing platform. Genome Biology, 22(1), 195. https://doi.org/10.1186/s13059-021-02406-y.
Lynch, A. R., Bradford, S. V., Zhou, A. S., Oxendine, K., Henderson, L., Horner, V. L., Weaver, B. A., & Burkard, M. E. (2023). A survey of CIN measures across mechanistic models. bioRxiv (Cold Spring Harbor Laboratory). https://doi.org/10.1101/2023.06.15.544840.
Aziz, M. A., Periyasamy, S., Yousef, Z., AlAbdulkarim, I., Otaibi, M. A., Alfahed, A., & Alasiri, G. (2014). Integrated EXON level expression analysis of driver genes explain their role in colorectal cancer. PLOS ONE, 9(10), e110134. https://doi.org/10.1371/journal.pone.0110134.
Pöhl, S., & Myant, K. (2022). Alternative RNA splicing in tumour heterogeneity, plasticity and therapy. Disease Models & Mechanisms, 15(1). https://doi.org/10.1242/dmm.049233.
Althenayyan, S., AlMuhanna, M. H., AlAbdulrahman, A., Alghanem, B., Alsagaby, S. A., Alfahed, A., Alasiri, G., & Aziz, M. (2023). Alternatively spliced isoforms of MUC4 and ADAM12 as biomarkers for colorectal cancer metastasis. Journal of Personalized Medicine, 13(1), 135. https://doi.org/10.3390/jpm13010135.
Kröger, C., Afeyan, A., Mraz, J., Eaton, E. N., Reinhardt, F., Khodor, Y. L., Thiru, P., Bierie, B., Ye, X., Burge, C. B., & Weinberg, R. A. (2019). Acquisition of a hybrid E/M state is essential for tumorigenicity of basal breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America, 116(15), 7353–7362. https://doi.org/10.1073/pnas.1812876116.
Panchy, N., Azeredo-Tseng, C., Luo, M., Randall, N., & Hong, T. (2020). Integrative transcriptomic analysis reveals a multiphasic epithelial–mesenchymal spectrum in cancer and non-tumorigenic cells. Frontiers in Oncology, 9. https://doi.org/10.3389/fonc.2019.01479.
Aiello, N. M., & Kang, Y. (2019). Context-dependent EMT programs in cancer metastasis. Journal of Experimental Medicine, 216(5), 1016–1026. https://doi.org/10.1084/jem.20181827.
Grigore, A. D., Jolly, M. K., Jia, D., Farach-Carson, M. C., & Levine, H. (2016). Tumor budding: The name is EMT. Partial EMT. Journal of Clinical Medicine, 5(5), 51. https://doi.org/10.3390/jcm5050051.
Saitoh, M. (2018). Involvement of partial EMT in cancer progression. Journal of Biochemistry, 164(4), 257–264. https://doi.org/10.1093/jb/mvy047.
Chan, J. M., Quintanal-Villalonga, Á., Gao, V. R., Xie, Y., Allaj, V., Chaudhary, O., Masilionis, I., Egger, J. V., Chow, A., Walle, T., Mattar, M. S., Yarlagadda, D. V., Wang, J. L., Uddin, F., Offin, M., Ciampricotti, M., Qeriqi, B., Bahr, A., De Stanchina, E., & Rudin, C. M. (2021). Signatures of plasticity, metastasis, and immunosuppression in an atlas of human small cell lung cancer. Cancer Cell, 39(11), 1479–1496e18. https://doi.org/10.1016/j.ccell.2021.09.008.
Aceto, N., Bardia, A., Miyamoto, D. T., Donaldson, M. C., Wittner, B. S., Spencer, J. A., Yu, M., Pely, A., Engstrom, A., Zhu, H., Brannigan, B. W., Kapur, R., Stott, S. L., Shioda, T., Ramaswamy, S., Ting, D. T., Lin, C. P., Toner, M., Haber, D. A., & Maheswaran, S. (2014). Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell, 158(5), 1110–1122. https://doi.org/10.1016/j.cell.2014.07.013.
Gkountela, S., Castro-Giner, F., Szczerba, B. M., Vetter, M., Landin, J., Scherrer, R., Krol, I., Scheidmann, M. C., Beisel, C., Stirnimann, C. U., Kurzeder, C., Heinzelmann-Schwarz, V., Rochlitz, C., Weber, W., & Aceto, N. (2019). Circulating tumor cell clustering shapes DNA methylation to enable metastasis seeding. Cell, 176(1–2), 98–112e14. https://doi.org/10.1016/j.cell.2018.11.046.
Vincent, A., Ouelkdite-Oumouchal, A., Souidi, M., Leclerc, J., Neve, B., & Van Seuningen, I. (2019). Colon cancer stemness as a reversible epigenetic state: Implications for anticancer therapies. World Journal of Stem Cells, 11(11), 920–936. https://doi.org/10.4252/wjsc.v11.i11.920.
Chaffer, F., Brueckmann, C. L., Scheel, I., Kaestli, C., Wiggins, A. J., Rodrigues, P. A., Brooks, L. O., Reinhardt, M., Su, F., Polyak, Y., Arendt, K., Kuperwasser, L. M., Bierie, C., & Weinberg, B. (2011). Normal and neoplastic nonstem cells can spontaneously convert to a stem-like state. Proc Natl Acad Sci U S A, 108(19), 7950–7955. https://doi.org/10.1073/pnas.1102454108.
Zhang, C. C., Aldrees, M., Arif, M., Li, X., Mardinoğlu, A., & Aziz, M. A. (2019). Elucidating the reprograming of colorectal cancer metabolism using genome-scale metabolic modeling. Frontiers in Oncology, 9. https://doi.org/10.3389/fonc.2019.00681.
Wang, Z., Wu, X., Chen, H., & Wang, K. (2023). Amino acid metabolic reprogramming in tumor metastatic colonization. Frontiers in Oncology, 13. https://doi.org/10.3389/fonc.2023.1123192.
Bergers, G., & Fendt, S. (2021). The metabolism of cancer cells during metastasis. Nature Reviews Cancer, 21(3), 162–180. https://doi.org/10.1038/s41568-020-00320-2.
Gaude, E., & Frezza, C. (2016). Tissue-specific and convergent metabolic transformation of cancer correlates with metastatic potential and patient survival. Nature Communications, 7(1). https://doi.org/10.1038/ncomms13041.
Christen, S., Lorendeau, D., Schmieder, R., Broekaert, D., Metzger, K. L., Veys, K., Elia, I., Buescher, J. M., Orth, M. F., Davidson, S. M., Grünewald, T., De Bock, K., & Fendt, S. (2016). Breast cancer-derived lung metastases show increased pyruvate carboxylase-dependent anaplerosis. Cell Reports, 17(3), 837–848. https://doi.org/10.1016/j.celrep.2016.09.042
Delaunay, S., Pascual, G., Feng, B., Klann, K., Behm, M., Hotz-Wagenblatt, A., Richter, K., Zaoui, K., Herpel, E., Münch, C., Dietmann, S., Heß, J., Benitah, S. A., & Frye, M. (2022). Mitochondrial RNA modifications shape metabolic plasticity in metastasis. Nature, 607(7919), 593–603. https://doi.org/10.1038/s41586-022-04898-5.
Chen, A., Luo, Y., Yang, Y., Fu, J., Geng, X., Shi, J., & Yang, J. (2021). Lactylation, a novel Metabolic Reprogramming Code: Current status and prospects. Frontiers in Immunology, 12. https://doi.org/10.3389/fimmu.2021.688910.
Thakur, C., & Chen, F. (2019). Connections between metabolism and epigenetics in cancers. Seminars in Cancer Biology, 57, 52–58. https://doi.org/10.1016/j.semcancer.2019.06.006.
Dar, M. A., Arafah, A., Bhat, K. A., Khan, A., Khan, M. S., Ali, A., Ahmad, S. M., Rashid, S. M., & Rehman, M. U. (2023). Multiomics technologies: Role in disease biomarker discoveries and therapeutics. Briefings in Functional Genomics, 22(2), 76–96. https://doi.org/10.1093/bfgp/elac017.
Althenayyan, S., AlMuhanna, M. H., AlAbdulrahman, A., Alghanem, B., Alsagaby, S. A., Alfahed, A., Alasiri, G., & Aziz, M. A. (2023). Alternatively spliced isoforms of MUC4 and ADAM12 as biomarkers for colorectal cancer metastasis. J Pers Med, 13(1), 135. https://doi.org/10.3390/jpm13010135
Liu, F., Ke, J., & Song, Y. (2020). Application of biomarkers for the prediction and diagnosis of bone metastasis in breast cancer. J Breast Cancer, 23(6), 588–598. https://doi.org/10.4048/jbc.2020.23.e65
Aziz, M., Yousef, Z., Saleh, A. M., Mohammad, S., & Knawy, B. A. (2017). Towards personalized medicine of colorectal cancer. Critical Reviews in Oncology/Hematology, 118, 70–78. https://doi.org/10.1016/j.critrevonc.2017.08.007.
Castro-Giner, F., & Aceto, N. (2020). Tracking cancer progression: From circulating tumor cells to metastasis. Genome Medicine, 12(1). https://doi.org/10.1186/s13073-020-00728-3.
Loreth, D., Schuette, M., Zinke, J., Mohme, M., Piffko, A., Schneegans, S., Stadler, J., Janning, M., Loges, S., Joosse, S. A., Lamszus, K., Westphal, M., Müller, V., Glatzel, M., Matschke, J., Gebhardt, C., Schneider, S. W., Belczacka, I., Volkmer, B., & Wikman, H. (2021). CD74 and CD44 expression on CTCs in cancer patients with brain metastasis. International Journal of Molecular Sciences, 22(13), 6993. https://doi.org/10.3390/ijms22136993
Sperger, J. M., Emamekhoo, H., McKay, R. R., Stahlfeld, C. N., Singh, A., Chen, X. E., Kwak, L., Gilsdorf, C. S., Wolfe, S. K., Wei, X. X., Silver, R., Zhang, Z., Morris, M. J., Bubley, G. J., Feng, F. Y., Scher, H. I., Rathkopf, D. E., Dehm, S. M., Choueiri, T. K., & Lang, J. M. (2021). Prospective evaluation of clinical outcomes using a multiplex liquid biopsy targeting diverse resistance mechanisms in metastatic prostate cancer. Journal of Clinical Oncology, 39(26), 2926–2937. https://doi.org/10.1200/jco.21.00169.
Zhang, J., Chen, J., Wo, D., Yan, H., Liu, P., Eglitis, M. A., Li, L., Zheng, L., Chen, D., Yu, Z., Liang, C., Peng, J., Ren, D., & Zhu, W. (2019). LRP6 ectodomain prevents SDF-1/CXCR4-induced breast cancer metastasis to lung. Clinical Cancer Research, 25(15), 4832–4845. https://doi.org/10.1158/1078-0432.ccr-18-3557
Yao, Q., An, Y., Hou, W., Cao, Y., Yao, M., Ma, N., Hou, L., Zhang, H., Liu, H., & Zhang, B. (2017). LRP6 promotes invasion and metastasis of colorectal cancer through cytoskeleton dynamics. Oncotarget, 8(65), 109632–109645. https://doi.org/10.18632/oncotarget.22759.
Nalejska, E., Mączyńska, E., & Lewandowska, M. A. (2014). Prognostic and predictive biomarkers: Tools in personalized oncology. Molecular Diagnosis & Therapy, 18, 273–284. https://doi.org/10.1007/s40291-013-0077-9
Wood, S. L., & Brown, J. E. (2020). Personal medicine and bone metastases: Biomarkers, micro-RNAs and bone metastases. Cancers, 12(8), 2109. https://doi.org/10.3390/cancers12082109
Kong, D., Hughes, C. J., & Ford, H. L. (2020). Cellular plasticity in breast cancer progression and therapy. Frontiers in Molecular Biosciences, 7. https://doi.org/10.3389/fmolb.2020.00072.
Fillmore, C. M., Gupta, P. B., Rudnick, J. A., Caballero, S., Keller, P. J., & Lander, E. S. (2010). Estrogen expands breast cancer stem-like cells through paracrine FGF/Tbx3 signaling. Proceedings of the National Academy of Sciences U S A, 107, 21737–21742. https://doi.org/10.1073/pnas.1007863107
Schwitalla, S. (2014). Tumor cell plasticity: The challenge to catch a moving target. Journal of Gastroenterology, 49(4), 618–627. https://doi.org/10.1007/s00535-014-0943-1.
He, X., Liu, X., Zuo, F., Shi, H., & Jing, J. (2023). Artificial intelligence-based multi-omics analysis fuels cancer precision medicine. Seminars in Cancer Biology, 88, 187–200. https://doi.org/10.1016/j.semcancer.2022.12.009.
Naulaerts, S., Datsi, A., Borrás, D., Antoranz, A., Messiaen, J., Vanmeerbeek, I., Sprooten, J., Laureano, R. S., Govaerts, J., Panovska, D., Derweduwe, M., Sabel, M., Rapp, M., Ni, W., Mackay, S., Van Herck, Y., Gelens, L., Venken, T., More, S., & Garg, A. D. (2023). Multiomics and spatial mapping characterizes human CD8 + T cell states in cancer. Science Translational Medicine, 15(691). https://doi.org/10.1126/scitranslmed.add1016.
Popa, M., Albulescu, R., Neagu, M., Hinescu, M. E., & Tănase, C. (2019). Multiplex assay for multiomics advances in personalized-precision medicine. Journal of Immunoassay & Immunochemistry, 40(1), 3–25. https://doi.org/10.1080/15321819.2018.156294.
Sell, S. (2008). Alpha-fetoprotein, stem cells and cancer: How study of the production of alpha-fetoprotein during chemical hepatocarcinogenesis led to reaffirmation of the stem cell theory of cancer. Tumor Biology, 29(3), 161–180. https://doi.org/10.1159/000143402
Walcher, L., Kistenmacher, A., Suo, H., Kitte, R., Dluczek, S., Strauß, A., Blaudszun, A., Yevsa, T., Stephan, F., & Kossatz-Boehlert, U. (2020). Cancer stem cells—Origins and biomarkers: Perspectives for targeted personalized therapies. Frontiers in Immunology, 11. https://doi.org/10.3389/fimmu.2020.01280.
Alhumaid, A., Alyousef, Z., Bakhsh, H. A., Alghamdi, S., & Aziz, M. (2018). Emerging paradigms in the treatment of liver metastases in colorectal cancer. Critical Reviews in Oncology/Hematology, 132, 39–50. https://doi.org/10.1016/j.critrevonc.2018.09.011.
Acknowledgements
The encouragement of Cancer Nanomedicine Consortium members is acknowledged in the preparation of this review. Help extended by Professor Afzal Anees, Rashid Ali, Mohammad Jaseem Hasan, Mohammad Akram, Hamid Ashraf, and Hifzurrahman Siddique is duly acknowledged.
Author information
Authors and Affiliations
Contributions
MAA is responsible for conceiving, writing, and proofreading the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aziz, M.A. Multiomics approach towards characterization of tumor cell plasticity and its significance in precision and personalized medicine. Cancer Metastasis Rev (2024). https://doi.org/10.1007/s10555-024-10190-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10555-024-10190-x