Abstract
Abdominal tumors (AT) in children account for approximately 17% of all pediatric solid tumor cases, and frequently exhibit embryonal histological features that differentiate them from adult cancers. Current molecular approaches have greatly improved the understanding of the distinctive pathology of each tumor type and enabled the characterization of novel tumor biomarkers. As seen in abdominal adult tumors, microRNAs (miRNAs) have been increasingly implicated in either the initiation or progression of childhood cancer. Moreover, besides predicting patient prognosis, they represent valuable diagnostic tools that may also assist the surveillance of tumor behavior and treatment response, as well as the identification of the primary metastatic sites. Thus, the present study was undertaken to compile up-to-date information regarding the role of dysregulated miRNAs in the most common histological variants of AT, including neuroblastoma, nephroblastoma, hepatoblastoma, hepatocarcinoma, and adrenal tumors. Additionally, the clinical implications of dysregulated miRNAs as potential diagnostic tools or indicators of prognosis were evaluated.

Similar content being viewed by others
References
Yates, L. A. A., Norbury, C. J. J., & Gilbert, R. J. C. J. C. (2013). The long and short of microRNA. Cell, 153(3), 516–519. https://doi.org/10.1016/j.cell.2013.04.003
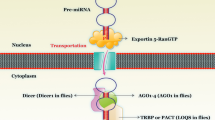
Ha, M., & Kim, V. N. (2014). Regulation of microRNA biogenesis, 15(8). https://doi.org/10.1038/nrm3838
Eulalio, A., Huntzinger, E., & Izaurralde, E. (2008). Getting to the root of miRNA-mediated gene silencing. Cell, 132(1), 9–14. https://doi.org/10.1016/j.cell.2007.12.024
Friedman, R. C., Farh, K. K.-H., Burge, C. B., & Bartel, D. P. (2009). Most mammalian mRNAs are conserved targets of microRNAs. Genome Research, 19(1), 92–105. https://doi.org/10.1101/gr.082701.108
Wu, J., Bao, J., Kim, M., Yuan, S., Tang, C., Zheng, H., … Yan, W. (2014). Two miRNA clusters, miR-34b/c and miR-449, are essential for normal brain development, motile ciliogenesis, and spermatogenesis, 111(28). https://doi.org/10.1073/pnas.1407777111
Chen, C.-Z., Schaffert, S., Fragoso, R., & Loh, C. (2013). Regulation of immune responses and tolerance: the microRNA perspective, 253(1). https://doi.org/10.1111/imr.12060
Papaioannou, G., Mirzamohammadi, F., & Kobayashi, T. (2014). MicroRNAs involved in bone formation, 71(24). https://doi.org/10.1007/s00018-014-1700-6
Follert, P., Cremer, H., & Béclin, C. (2014). MicroRNAs in brain development and function: a matter of flexibility and stability, 7, 5. https://doi.org/10.3389/fnmol.2014.00005
Hodgkinson, C. P., Kang, M. H., Dal-Pra, S., Mirotsou, M., & Dzau, V. J. (2015). MicroRNAs and cardiac regeneration, 116(10). https://doi.org/10.1161/CIRCRESAHA.116.304377
Lujambio, A., & Lowe, S. W. (2012). The microcosmos of cancer. Nature, 482(7385). https://doi.org/10.1038/nature10888
Allen-Rhoades, W., Whittle, S. B., & Rainusso, N. (2018). Pediatric solid tumors of infancy: an overview. Pediatrics in Review, 39(2), 57–67. https://doi.org/10.1542/PIR.2017-0057
Amodeo, I., Cavallaro, G., Raffaeli, G., Colombo, L., Fumagalli, M., Cavalli, R., … Mosca, F. (2017). Abdominal cystic lymphangioma in a term newborn: a case report and update of new treatments. Medicine, 96(8), e5984. https://doi.org/10.1097/MD.0000000000005984
Chiorean, L. (2015). Benign liver tumors in pediatric patients - review with emphasis on imaging features. World Journal of Gastroenterology, 21(28), 8541. https://doi.org/10.3748/wjg.v21.i28.8541
Debnath, J., Pandit, A., Roy, S., & Sahoo, S. (2013). Congenital giant hydronephrosis: a rare cause for upper abdominal mass in the newborn. Journal of Clinical Neonatology, 2(1), 33. https://doi.org/10.4103/2249-4847.109246
Brodeur, A. E., & Brodeur, G. M. (1991). Abdominal masses in children: neuroblastoma, Wilms tumor, and other considerations. Pediatrics in review, 12(7), 196–207. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/1846958
Kwok, G. T., Zhao, J. T., Weiss, J., Mugridge, N., Brahmbhatt, H., MacDiarmid, J. A., … Sidhu, S. B. (2017). Translational applications of microRNAs in cancer, and therapeutic implications. Non-coding RNA Research, 2(3–4), 143–150. https://doi.org/10.1016/j.ncrna.2017.12.002
Chen, X., Ba, Y., Ma, L., Cai, X., Yin, Y., Wang, K., … Zhang, C.-Y. (2008). Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases, 18(10), 997–1006. https://doi.org/10.1038/cr.2008.282
Mraz, M., Malinova, K., Mayer, J., & Pospisilova, S. (2009). MicroRNA isolation and stability in stored RNA samples, 390(1), 1–4. https://doi.org/10.1016/j.bbrc.2009.09.061
Lu, M., Zhang, Q., Deng, M., Miao, J., Guo, Y., Gao, W., & Cui, Q. (2008). An analysis of human microRNA and disease associations. PLoS ONE, 3(10), e3420. https://doi.org/10.1371/journal.pone.0003420
Calin, G. A., Sevignani, C., Dumitru, C. D., Hyslop, T., Noch, E., Yendamuri, S., … Croce, C. M. (2004). Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers, 101(9), 2999–3004. https://doi.org/10.1073/pnas.0307323101
Chen, C.-Z. (2005). MicroRNAs as oncogenes and tumor suppressors. New England Journal of Medicine, 353(17), 1768–1771. https://doi.org/10.1056/NEJMp058190
Wang, L. Q., & Chim, C. S. (2015). DNA methylation of tumor-suppressor miRNA genes in chronic lymphocytic leukemia. Epigenomics, 7(3), 461–473. https://doi.org/10.2217/epi.15.6
Kian, R., Moradi, S., & Ghorbian, S. (2018). Role of components of microRNA machinery in carcinogenesis. Experimental oncology, 40(1), 2–9. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/29600985
Wu, M., Sabbaghian, N., Xu, B., Addidou-Kalucki, S., Bernard, C., Zou, D., … Foulkes, W. (2013). Biallelic DICER1mutations occur in Wilms tumours. The Journal of Pathology, 230(2), 154–164. https://doi.org/10.1002/path.4196
Foulkes, W. D., Priest, J. R., & Duchaine, T. F. (2014). DICER1: mutations, microRNAs and mechanisms, 14(10). https://doi.org/10.1038/nrc3802
Solarski, M., Rotondo, F., Foulkes, W. D., Priest, J. R., Syro, L. V, Butz, H., … Kovacs, K. (2018). DICER1 gene mutations in endocrine tumors. Endocrine-Related Cancer, 25(3), R197–R208. https://doi.org/10.1530/ERC-17-0509
Mathew, P., Valentine, M. B., Bowman, L. C., Rowe, S. T., Nash, M. B., Valentine, V. A., … Look, A. T. (2001). Detection of MYCN gene amplification in neuroblastoma by fluorescence in situ hybridization: a pediatric oncology group study. Neoplasia (New York, N.Y.), 3(2), 105–9. https://doi.org/10.1038/sj/neo/7900146
Brodeur, G. M. (2003). Neuroblastoma: biological insights into a clinical enigma, 3(3). https://doi.org/10.1038/nrc1014
Cole, K. A., Attiyeh, E. F., Mosse, Y. P., Laquaglia, M. J., Diskin, S. J., Brodeur, G. M., & Maris, J. M. (2008). A functional screen identifies miR-34a as a candidate neuroblastoma tumor suppressor gene. Molecular cancer research: MCR, 6(5), 735–42. https://doi.org/10.1158/1541-7786.MCR-07-2102
Welch, C., Chen, Y., & Stallings, R. L. (2007). MicroRNA-34a functions as a potential tumor suppressor by inducing apoptosis in neuroblastoma cells. Oncogene, 26(34). https://doi.org/10.1038/sj.onc.1210293
Wei, J. S., Song, Y. K., Durinck, S., Chen, Q.-R., Cheuk, A. T. C., Tsang, P., … Khan, J. (2008). The MYCN oncogene is a direct target of miR-34a. Oncogene, 27(39). https://doi.org/10.1038/onc.2008.154
Buechner, J., Tømte, E., Haug, B. H., Henriksen, J. R., Løkke, C., Flægstad, T., & Einvik, C. (2011). Tumour-suppressor microRNAs let-7 and mir-101 target the proto-oncogene MYCN and inhibit cell proliferation in MYCN-amplified neuroblastoma. British Journal of Cancer, 105(2), 296–303. https://doi.org/10.1038/bjc.2011.220
Wang, L., Che, X.-J., Wang, N., Li, J., & Zhu, M.-H. (2014). Regulatory network analysis of microRNAs and genes in neuroblastoma. Asian Pacific journal of cancer prevention: APJCP, 15(18), 7645–52. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/25292042
Tivnan, A., Tracey, L., Buckley, P. G., Alcock, L. C., Davidoff, A. M., & Stallings, R. L. (2011). MicroRNA-34a is a potent tumor suppressor molecule in vivo in neuroblastoma, 11(1), 33. https://doi.org/10.1186/1471-2407-11-33
De Antonellis, P., Carotenuto, M., Vandenbussche, J., De Vita, G., Ferrucci, V., Medaglia, C., … Zollo, M. (2014). Early targets of miR-34a in neuroblastoma, 13(8). https://doi.org/10.1074/mcp.M113.035808
Cheng, X., Xu, Q., Zhang, Y., Shen, M., Zhang, S., Mao, F., … Zhang, Q. (2019). miR-34a inhibits progression of neuroblastoma by targeting autophagy-related gene 5. European Journal of Pharmacology, 53–63. https://doi.org/10.1016/j.ejphar.2019.01.071
He, X.-Y., Tan, Z.-L., Mou, Q., Liu, F.-J., Liu, S., Yu, C., … Zou, L. (2017). microRNA-221 enhances MYCN via targeting Nemo-like kinase and functions as an oncogene related to poor prognosis in neuroblastoma, 23(11), 2905–2918. https://doi.org/10.1158/1078-0432.CCR-16-1591
Teitz, T., Inoue, M., Valentine, M. B., Zhu, K., Rehg, J. E., Zhao, W., … Lahti, J. M. (2013). Th-MYCN mice with caspase-8 deficiency develop advanced neuroblastoma with bone marrow metastasis. Cancer Research, 73(13), 4086–4097. https://doi.org/10.1158/0008-5472.CAN-12-2681
Xin, C., Buhe, B., Hongting, L., Chuanmin, Y., Xiwei, H., Hong, Z., … Renjie, W. (2013). MicroRNA-15a promotes neuroblastoma migration by targeting reversion-inducing cysteine-rich protein with Kazal motifs (RECK) and regulating matrix metalloproteinase-9 expression, 280(3). https://doi.org/10.1111/febs.12074
Zhang, H., Qi, M., Li, S., Qi, T., Mei, H., Huang, K., … Tong, Q. (2012). microRNA-9 targets matrix metalloproteinase 14 to inhibit invasion, metastasis, and angiogenesis of neuroblastoma cells. Molecular cancer therapeutics, 11(7), 1454–66. https://doi.org/10.1158/1535-7163.MCT-12-0001
Xiang, X., Mei, H., Qu, H., Zhao, X., Li, D., Song, H., … Tong, Q. (2015). miRNA-584-5p exerts tumor suppressive functions in human neuroblastoma through repressing transcription of matrix metalloproteinase 14. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 1852(9), 1743–1754. Retrieved from https://www.sciencedirect.com/science/article/pii/S0925443915001660
Mestdagh, P., Boström, A.-K., Impens, F., Fredlund, E., Van Peer, G., De Antonellis, P., … Vandesompele, J. (2010). The miR-17-92 microRNA cluster regulates multiple components of the TGF-β pathway in neuroblastoma. Molecular Cell, 40(5), 762–773. https://doi.org/10.1016/j.molcel.2010.11.038
Fontana, L., Fiori, M. E., Albini, S., Cifaldi, L., Giovinazzi, S., Forloni, M., … Fruci, D. (2008). Antagomir-17-5p abolishes the growth of therapy-resistant neuroblastoma through p21 and BIM, 3(5), e2236. https://doi.org/10.1371/journal.pone.0002236
Schulte, J. H., Marschall, T., Martin, M., Rosenstiel, P., Mestdagh, P., Schlierf, S., … Schramm, A. (2010). Deep sequencing reveals differential expression of microRNAs in favorable versus unfavorable neuroblastoma. Nucleic Acids Research, 38(17), 5919–5928. https://doi.org/10.1093/nar/gkq342
Bray, I., Bryan, K., Prenter, S., Buckley, P. G., Foley, N. H., Murphy, D. M., … Stallings, R. L. (2009). Widespread dysregulation of miRNAs by MYCN amplification and chromosomal imbalances in neuroblastoma: association of miRNA expression with survival. PLoS ONE, 4(11), e7850. https://doi.org/10.1371/journal.pone.0007850
Ribeiro, D., Klarqvist, M. D. R., Westermark, U. K., Oliynyk, G., Dzieran, J., Kock, A., … Arsenian Henriksson, M. (2016). Regulation of nuclear hormone receptors by MYCN-driven miRNAs impacts neural differentiation and survival in neuroblastoma patients. Cell Reports, 16(4), 979–993. https://doi.org/10.1016/j.celrep.2016.06.052
De Brouwer, S., Mestdagh, P., Lambertz, I., Pattyn, F., De Paepe, A., Westermann, F., Speleman, F. (2012). Dickkopf-3 is regulated by the MYCN-induced miR-17-92 cluster in neuroblastoma, 130(11). https://doi.org/10.1002/ijc.26295
Haug, B. H., Henriksen, J. R., Buechner, J., Geerts, D., Tømte, E., Kogner, P., … Einvik, C. (2011). MYCN-regulated miRNA-92 inhibits secretion of the tumor suppressor DICKKOPF-3 (DKK3) in neuroblastoma. Carcinogenesis, 32(7), 1005–1012. https://doi.org/10.1093/carcin/bgr073
Lovén, J., Zinin, N., Wahlström, T., Müller, I., Brodin, P., Fredlund, E., … Henriksson, M. (2010). MYCN-regulated microRNAs repress estrogen receptor-alpha (ESR1) expression and neuronal differentiation in human neuroblastoma. Proceedings of the National Academy of Sciences of the United States of America, 107(4), 1553–8. https://doi.org/10.1073/pnas.0913517107
Bienertova-Vasku, J., Mazanek, P., Hezova, R., Curdova, A., Nekvindova, J., Kren, L., Slaby, O. (2013). Extension of microRNA expression pattern associated with high-risk neuroblastoma. Tumor Biology, 34(4), 2315–2319. https://doi.org/10.1007/s13277-013-0777-0
Naraparaju, K., Kolla, V., Zhuang, T., Higashi, M., Iyer, R., Kolla, S., … Brodeur, G. M. (2016). Role of microRNAs in epigenetic silencing of the CHD5 tumor suppressor gene in neuroblastomas. Oncotarget, 7(13), 15977–85. https://doi.org/10.18632/oncotarget.7434
Zhou, Y., & Sheng, B. (2016). Association of microRNA 21 with biological features and prognosis of neuroblastoma. Cancer control: journal of the Moffitt Cancer Center, 23(1), 78–84. https://doi.org/10.1177/107327481602300113
Chen, Y., Tsai, Y.-H., Fang, Y., & Tseng, S.-H. (2012). Micro-RNA-21 regulates the sensitivity to cisplatin in human neuroblastoma cells, 47(10). https://doi.org/10.1016/j.jpedsurg.2012.05.013
Gumbiner, B. M. (2005). Regulation of cadherin-mediated adhesion in morphogenesis. Nature Reviews Molecular Cell Biology, 6(8), 622–634. https://doi.org/10.1038/nrm1699
Cheng, L., Yang, T., Kuang, Y., Kong, B., Yu, S., Shu, H., … Gu, J. (2014). MicroRNA-23a promotes neuroblastoma cell metastasis by targeting CDH1. Oncology letters, 7(3), 839–845. https://doi.org/10.3892/ol.2014.1794
Khan, F. H., Pandian, V., Ramraj, S., Aravindan, S., Herman, T. S., & Aravindan, N. (2015). Reorganization of metastamiRs in the evolution of metastatic aggressive neuroblastoma cells. BMC Genomics, 16(1), 501. https://doi.org/10.1186/s12864-015-1642-x
Maugeri, M., Barbagallo, D., Barbagallo, C., Banelli, B., Di Mauro, S., Purrello, F., … Purrello, M. (2016). Altered expression of miRNAs and methylation of their promoters are correlated in neuroblastoma. Oncotarget, 7(50), 83330–83341. https://doi.org/10.18632/oncotarget.13090
Cheung, I. Y., Farazi, T. A., Ostrovnaya, I., Xu, H., Tran, H., Mihailovic, A., … Cheung, N.-K. V. (2014). Deep microRNA sequencing reveals downregulation of miR-29a in neuroblastoma central nervous system metastasis, 53(10). https://doi.org/10.1002/gcc.22189
Parodi, F., Carosio, R., Ragusa, M., Di Pietro, C., Maugeri, M., Barbagallo, D., … Banelli, B. (2016). Epigenetic dysregulation in neuroblastoma: A tale of miRNAs and DNA methylation. Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms, 1859(12), 1502–1514. https://doi.org/10.1016/j.bbagrm.2016.10.006
Huang, T.-C., Chang, H.-Y., Chen, C.-Y., Wu, P.-Y., Lee, H., Liao, Y.-F., … Juan, H.-F. (2011). Silencing of miR-124 induces neuroblastoma SK-N-SH cell differentiation, cell cycle arrest and apoptosis through promoting AHR, 585(22). https://doi.org/10.1016/j.febslet.2011.10.025
Zhao, Z., Ma, X., Shelton, S. D., Sung, D. C., Li, M., Hernandez, D., … Du, L. (2016). A combined gene expression and functional study reveals the crosstalk between N-Myc and differentiation-inducing microRNAs in neuroblastoma cells. Oncotarget, 7(48), 79372–79387. https://doi.org/10.18632/oncotarget.12676
Ayers, D., Mestdagh, P., Van Maerken, T., & Vandesompele, J. (2015). Identification of miRNAs contributing to neuroblastoma chemoresistance, 13. https://doi.org/10.1016/j.csbj.2015.04.003
Takwi, A. A., Wang, Y.-M., Wu, J., Michaelis, M., Cinatl, J., & Chen, T. (2014). miR-137 regulates the constitutive androstane receptor and modulates doxorubicin sensitivity in parental and doxorubicin-resistant neuroblastoma cells. Oncogene, 33(28). https://doi.org/10.1038/onc.2013.330
Zhao, G., Wang, G., Bai, H., Li, T., Gong, F., Yang, H., … Wang, W. (2017). Targeted inhibition of HDAC8 increases the doxorubicin sensitivity of neuroblastoma cells via up regulation of miR-137. European journal of pharmacology, 802, 20–26. https://doi.org/10.1016/j.ejphar.2017.02.035
Chakrabarti, M., Banik, N. L., & Ray, S. K. (2013). miR-138 overexpression is more powerful than hTERT knockdown to potentiate apigenin for apoptosis in neuroblastoma in vitro and in vivo, 319(10). https://doi.org/10.1016/j.yexcr.2013.02.025
Yamagata, T., Yoshizawa, J., Ohashi, S., Yanaga, K., & Ohki, T. (2010). Expression patterns of microRNAs are altered in hypoxic human neuroblastoma cells, 26(12). https://doi.org/10.1007/s00383-010-2700-8
Zhang, H., Pu, J., Qi, T., Qi, M., Yang, C., Li, S., … Tong, Q. (2014). MicroRNA-145 inhibits the growth, invasion, metastasis and angiogenesis of neuroblastoma cells through targeting hypoxia-inducible factor 2 alpha. Oncogene, 33(3). https://doi.org/10.1038/onc.2012.574
Xu, Y., Chen, X., Lin, L., Chen, H., Yu, S., & Li, D. (2017). MicroRNA-149 is associated with clinical outcome in human neuroblastoma and modulates cancer cell proliferation through Rap1 independent of MYCN amplification. Biochimie, 139, 1–8. https://doi.org/10.1016/j.biochi.2017.04.011
Lin, R.-J., Lin, Y.-C., & Yu, A. L. (2010). miR-149* induces apoptosis by inhibiting Akt1 and E2F1 in human cancer cells, 49(8). https://doi.org/10.1002/mc.20647
Das, S., Foley, N., Bryan, K., Watters, K. M., Bray, I., Murphy, D. M., … Stallings, R. L. (2010). MicroRNA mediates DNA demethylation events triggered by retinoic acid during neuroblastoma cell differentiation. Cancer Research, 70(20), 7874–7881. https://doi.org/10.1158/0008-5472.CAN-10-1534
Gibert, B., Delloye-Bourgeois, C., Gattolliat, C.-H., Meurette, O., Le Guernevel, S., Fombonne, J., … Mehlen, P. (2014). Regulation by miR181 family of the dependence receptor CDON tumor suppressive activity in neuroblastoma. JNCI: Journal of the National Cancer Institute, 106(11). https://doi.org/10.1093/jnci/dju318
Li, Y., Wang, H., Li, J., & Yue, W. (2014). MiR-181c modulates the proliferation, migration, and invasion of neuroblastoma cells by targeting Smad7. Acta Biochimica et Biophysica Sinica, 46(1), 48–55. https://doi.org/10.1093/abbs/gmt124
Lodrini, M., Poschmann, G., Schmidt, V., Wünschel, J., Dreidax, D., Witt, O., … Deubzer, H. E. (2016). Minichromosome maintenance complex is a critical node in the miR-183 signaling network of MYCN -amplified neuroblastoma cells. Journal of Proteome Research, 15(7), 2178–2186. https://doi.org/10.1021/acs.jproteome.6b00134
Lodrini, M., Oehme, I., Schroeder, C., Milde, T., Schier, M. C., Kopp-Schneider, A., … Deubzer, H. E. (2013). MYCN and HDAC2 cooperate to repress miR-183 signaling in neuroblastoma, 41(12). https://doi.org/10.1093/nar/gkt346
Foley, N. H., Bray, I. M., Tivnan, A., Bryan, K., Murphy, D. M., Buckley, P. G., … Stallings, R. L. (2010). MicroRNA-184 inhibits neuroblastoma cell survival through targeting the serine/threonine kinase AKT2. Molecular Cancer, 9(1), 83. https://doi.org/10.1186/1476-4598-9-83
Slaby, O. (2013). MiR-190 leads to aggressive phenotype of neuroblastoma through indirect activation of TrkB pathway, 80(3). https://doi.org/10.1016/j.mehy.2012.11.033
Feinberg-Gorenshtein, G., Guedj, A., Shichrur, K., Jeison, M., Luria, D., Kodman, Y., … Avigad, S. (2013). miR-192 directly binds and regulates Dicer1 expression in neuroblastoma. PLoS ONE, 8(11), e78713. https://doi.org/10.1371/journal.pone.0078713
Roth, S. A., Hald, Ø. H., Fuchs, S., Løkke, C., Mikkola, I., Flægstad, T., … Einvik, C. (2018). MicroRNA-193b-3p represses neuroblastoma cell growth via downregulation of Cyclin D1, MCL-1 and MYCN. Oncotarget, 9(26), 18160–18179. https://doi.org/10.18632/oncotarget.24793
Das, S., Bryan, K., Buckley, P. G., Piskareva, O., Bray, I. M., Foley, N., … Stallings, R. L. (2013). Modulation of neuroblastoma disease pathogenesis by an extensive network of epigenetically regulated microRNAs. Oncogene, 32(24). https://doi.org/10.1038/onc.2012.311
Gao, S.-L., Wang, L.-Z., Liu, H.-Y., Liu, D.-L., Xie, L.-M., & Zhang, Z.-W. (2014). miR-200a inhibits tumor proliferation by targeting AP-2γ in neuroblastoma cells. Asian Pacific journal of cancer prevention: APJCP, 15(11), 4671–6. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/24969902
Ryan, J., Tivnan, A., Fay, J., Bryan, K., Meehan, M., Creevey, L., … Stallings, R. L. (2012). MicroRNA-204 increases sensitivity of neuroblastoma cells to cisplatin and is associated with a favourable clinical outcome, 107(6). https://doi.org/10.1038/bjc.2012.356
Ooi, C. Y., Carter, D. R., Liu, B., Mayoh, C., Beckers, A., Lalwani, A., … Marshall, G. M. (2018). Network modeling of microRNA–mRNA interactions in neuroblastoma tumorigenesis identifies miR-204 as a direct inhibitor of MYCN. Cancer Research, 78(12), 3122–3134. https://doi.org/10.1158/0008-5472.CAN-17-3034
Yang, H., Li, Q., Zhao, W., Yuan, D., Zhao, H., & Zhou, Y. (2014). miR-329 suppresses the growth and motility of neuroblastoma by targeting KDM1A, 588(1). https://doi.org/10.1016/j.febslet.2013.11.036
Lynch, J., Meehan, M. H., Crean, J., Copeland, J., Stallings, R. L., & Bray, I. M. (2013). Metastasis suppressor microRNA-335 targets the formin family of actin nucleators, 8(11), e78428. https://doi.org/10.1371/journal.pone.0078428
Chen, X., Pan, M., Han, L., Lu, H., Hao, X., & Dong, Q. (2013). miR-338-3p suppresses neuroblastoma proliferation, invasion and migration through targeting PREX2a. FEBS Letters, 587(22), 3729–3737. https://doi.org/10.1016/j.febslet.2013.09.044
Wu, K., Yang, L., Chen, J., Zhao, H., Wang, J., Xu, S., & Huang, Z. (2015). miR-362-5p inhibits proliferation and migration of neuroblastoma cells by targeting phosphatidylinositol 3-kinase-C2β. FEBS Letters, 589(15), 1911–1919. https://doi.org/10.1016/J.FEBSLET.2015.05.056
Li, Y.-G., He, J.-H., YU, L., Hang, Z.-P., LI, W., Shun, W.-H., & HUANG, G. X. (2014). microRNA-202 suppresses MYCN expression under the control of E2F1 in the neuroblastoma cell line LAN-5, 9(2). https://doi.org/10.3892/mmr.2013.1845
Cui, C., Yu, J., Huang, S., Zhu, H., & Huang, Z. (2014). Transcriptional regulation of gene expression by microRNAs as endogenous decoys of transcription factors, 33(6). https://doi.org/10.1159/000362952
Swarbrick, A., Woods, S. L., Shaw, A., Balakrishnan, A., Phua, Y., Nguyen, A., … Goga, A. (2010). miR-380-5p represses p53 to control cellular survival and is associated with poor outcome in MYCN-amplified neuroblastoma, 16(10). https://doi.org/10.1038/nm.2227
Li, Y.-M. Y., Li, W., Zhang, J.-G., Li, H.-Y., & Li, Y.-M. Y. (2014). Downregulation of tumor suppressor menin by miR-421 promotes proliferation and migration of neuroblastoma. Tumor Biology, 35(10), 10011–10017. https://doi.org/10.1007/s13277-014-1921-1
Hu, H., Du, L., Nagabayashi, G., Seeger, R. C., & Gatti, R. A. (2010). ATM is down-regulated by N-Myc–regulated microRNA-421, 107(4). https://doi.org/10.1073/pnas.0907763107
Zhao, Z., Ma, X., Sung, D., Li, M., Kosti, A., Lin, G., … Du, L. (2015). microRNA-449a functions as a tumor suppressor in neuroblastoma through inducing cell differentiation and cell cycle arrest. RNA biology, 12(5), 538–54. https://doi.org/10.1080/15476286.2015.1023495
Liu, G., Xu, Z., & Hao, D. (2016). MicroRNA-451 inhibits neuroblastoma proliferation, invasion and migration by targeting macrophage migration inhibitory factor. Molecular medicine reports, 13(3), 2253–60. https://doi.org/10.3892/mmr.2016.4770
Gattolliat, C.-H., Le Teuff, G., Combaret, V., Mussard, E., Valteau-Couanet, D., Busson, P., … Douc-Rasy, S. (2014). Expression of two parental imprinted miRNAs improves the risk stratification of neuroblastoma patients, 3(4), 998–1009. https://doi.org/10.1002/cam4.264
Creevey, L., Ryan, J., Harvey, H., Bray, I. M., Meehan, M., Khan, A. R., & Stallings, R. L. (2013). MicroRNA-497 increases apoptosis in MYCN amplified neuroblastoma cells by targeting the key cell cycle regulator WEE1. Molecular Cancer, 12(1), 23. https://doi.org/10.1186/1476-4598-12-23
Soriano, A., París-Coderch, L., Jubierre, L., Martínez, A., Zhou, X., Piskareva, O., … Segura, M. F. (2016). MicroRNA-497 impairs the growth of chemoresistant neuroblastoma cells by targeting cell cycle, survival and vascular permeability genes. Oncotarget, 7(8), 9271–87. https://doi.org/10.18632/oncotarget.7005
Li, D., Cao, Y., Li, J., Xu, J., Liu, Q., & Sun, X. (2017). miR-506 suppresses neuroblastoma metastasis by targeting ROCK1. Oncology letters, 13(1), 417–422. https://doi.org/10.3892/ol.2016.5442
Harvey, H., Piskareva, O., Creevey, L., Alcock, L. C., Buckley, P. G., O’Sullivan, M. J., … Bray, I. M. (2015). Modulation of chemotherapeutic drug resistance in neuroblastoma SK-N-AS cells by the neural apoptosis inhibitory protein and miR-520f, 136(7). https://doi.org/10.1002/ijc.29144
Althoff, K., Lindner, S., Odersky, A., Mestdagh, P., Beckers, A., Karczewski, S., … Schulte, J. H. (2015). miR-542-3p exerts tumor suppressive functions in neuroblastoma by downregulating Survivin, 136(6). https://doi.org/10.1002/ijc.29091
Bray, I., Tivnan, A., Bryan, K., Foley, N. H., Watters, K. M., Tracey, L., … Stallings, R. L. (2011). MicroRNA-542-5p as a novel tumor suppressor in neuroblastoma. Cancer Letters, 303(1), 56–64. https://doi.org/10.1016/j.canlet.2011.01.016
Shohet, J. M., Ghosh, R., Coarfa, C., Ludwig, A., Benham, A. L., Chen, Z., … Gunaratne, P. H. (2011). A genome-wide search for promoters that respond to increased MYCN reveals both new oncogenic and tumor suppressor microRNAs associated with aggressive neuroblastoma, 71(11). https://doi.org/10.1158/0008-5472.CAN-10-4391
Buckley, P. G., Alcock, L., Bryan, K., Bray, I., Schulte, J. H., Schramm, A., … Stallings, R. L. (2010). Chromosomal and microRNA expression patterns reveal biologically distinct subgroups of 11q- neuroblastoma. Clinical Cancer Research, 16(11), 2971–2978. https://doi.org/10.1158/1078-0432.CCR-09-3215
Megiorni, F., Colaiacovo, M., Cialfi, S., McDowell, H. P., Guffanti, A., Camero, S., … Dominici, C. (2017). A sketch of known and novel MYCN-associated miRNA networks in neuroblastoma, 38(1), 3–20. https://doi.org/10.3892/or.2017.5701
Stigliani, S., Scaruffi, P., Lagazio, C., Persico, L., Carlini, B., Varesio, L., … Corrias, M. V. (2015). Deregulation of focal adhesion pathway mediated by miR-659-3p is implicated in bone marrow infiltration of stage M neuroblastoma patients. Oncotarget, 6(15), 13295–13308. https://doi.org/10.18632/oncotarget.3745
Afanasyeva, E. A., Mestdagh, P., Kumps, C., Vandesompele, J., Ehemann, V., Theissen, J., … Westermann, F. (2011). MicroRNA miR-885-5p targets CDK2 and MCM5, activates p53 and inhibits proliferation and survival, 18(6). https://doi.org/10.1038/cdd.2010.164
Wu, T., Lin, Y., & Xie, Z. (2018). MicroRNA-1247 inhibits cell proliferation by directly targeting ZNF346 in childhood neuroblastoma. Biological research, 51(1), 13. https://doi.org/10.1186/s40659-018-0162-y
Beveridge, N. J., Tooney, P. A., Carroll, A. P., Tran, N., & Cairns, M. J. (2009). Down-regulation of miR-17 family expression in response to retinoic acid induced neuronal differentiation, 21(12). https://doi.org/10.1016/j.cellsig.2009.07.019
Das, E., & Bhattacharyya, N. P. (2014). MicroRNA-432 contributes to dopamine cocktail and retinoic acid induced differentiation of human neuroblastoma cells by targeting NESTIN and RCOR1 genes, 588(9). https://doi.org/10.1016/j.febslet.2014.03.015
Watanabe, K., Yamaji, R., & Ohtsuki, T. (2018). MicroRNA-664a-5p promotes neuronal differentiation of SH-SY5Y cells. Genes to cells: devoted to molecular & cellular mechanisms, 23(3), 225–233. https://doi.org/10.1111/gtc.12559
Zhao, Z., Ma, X., Hsiao, T.-H., Lin, G., Kosti, A., Yu, X., … Du, L. (2014). A high-content morphological screen identifies novel microRNAs that regulate neuroblastoma cell differentiation. Oncotarget, 5(9), 2499–512. https://doi.org/10.18632/oncotarget.1703
Foley, N. H., Bray, I., Watters, K. M., Das, S., Bryan, K., Bernas, T., … Stallings, R. L. (2011). MicroRNAs 10a and 10b are potent inducers of neuroblastoma cell differentiation through targeting of nuclear receptor corepressor 2. Cell death and differentiation, 18(7), 1089–98. https://doi.org/10.1038/cdd.2010.172
Chen, H., Shalom-Feuerstein, R., Riley, J., Zhang, S.-D., Tucci, P., Agostini, M., … Vasa-Nicotera, M. (2010). miR-7 and miR-214 are specifically expressed during neuroblastoma differentiation, cortical development and embryonic stem cells differentiation, and control neurite outgrowth in vitro, 394(4). https://doi.org/10.1016/j.bbrc.2010.03.076
Adhikary, S., & Eilers, M. (2005). Transcriptional regulation and transformation by Myc proteins. Nature Reviews Molecular Cell Biology, 6(8), 635–645. https://doi.org/10.1038/nrm1703
Mestdagh, P., Fredlund, E., Pattyn, F., Schulte, J. H., Muth, D., Vermeulen, J., … Vandesompele, J. (2010). MYCN/c-MYC-induced microRNAs repress coding gene networks associated with poor outcome in MYCN/c-MYC-activated tumors. Oncogene, 29(9). https://doi.org/10.1038/onc.2009.429
Tanzer, A., & Stadler, P. F. (2004). Molecular evolution of a microRNA cluster. Journal of molecular biology, 339(2), 327–35. https://doi.org/10.1016/j.jmb.2004.03.065
Li, Z., Xu, Z., Xie, Q., Gao, W., Xie, J., & Zhou, L. (2016). miR-1303 promotes the proliferation of neuroblastoma cell SH-SY5Y by targeting GSK3β and SFRP1. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 83, 508–513. https://doi.org/10.1016/j.biopha.2016.07.010
Qu, H., Zheng, L., Pu, J., Mei, H., Xiang, X., Zhao, X., … Tong, Q. (2015). miRNA-558 promotes tumorigenesis and aggressiveness of neuroblastoma cells through activating the transcription of heparanase, 24(9). https://doi.org/10.1093/hmg/ddv018
Soriano, A., Masanas, M., Boloix, A., Masiá, N., París-Coderch, L., Piskareva, O., … Segura, M. F. (2019). Functional high-throughput screening reveals miR-323a-5p and miR-342-5p as new tumor-suppressive microRNA for neuroblastoma. Cellular and Molecular Life Sciences, 76(11), 2231–2243. https://doi.org/10.1007/s00018-019-03041-4
Zhu, K., Su, Y., Xu, B., Wang, Z., Sun, H., Wang, L., … He, X. (2019). MicroRNA-186-5p represses neuroblastoma cell growth via downregulation of Eg5. American journal of translational research, 11(4), 2245–2256. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/31105832
Zhao, Z., Partridge, V., Sousares, M., Shelton, S. D., Holland, C. L., Pertsemlidis, A., & Du, L. (2018). MicroRNA-2110 functions as an onco-suppressor in neuroblastoma by directly targeting Tsukushi. PLoS ONE, 13(12). https://doi.org/10.1371/journal.pone.0208777
Cao, X. Y., Sun, Z. Y., Zhang, L. J., Chen, M. K., & Yuan, B. (2019). MicroRNA-144-3p suppresses human neuroblastoma cell proliferation by targeting HOXA7. European Review for Medical and Pharmacological Sciences, 23(2), 716–723. https://doi.org/10.26355/eurrev_201901_16885
Schulte, J. H., Schowe, B., Mestdagh, P., Kaderali, L., Kalaghatgi, P., Schlierf, S., … Schramm, A. (2010). Accurate prediction of neuroblastoma outcome based on miRNA expression profiles, 127(10). https://doi.org/10.1002/ijc.25436
Chen, Y., & Stallings, R. L. (2007). Differential patterns of microRNA expression in neuroblastoma are correlated with prognosis, differentiation, and apoptosis, 67(3). https://doi.org/10.1158/0008-5472.CAN-06-3667
Zhao, L.-L., Jin, F., Ye, X., Zhu, L., Yang, J.-S., & Yang, W.-J. (2015). Expression profiles of miRNAs and involvement of miR-100 and miR-34 in regulation of cell cycle arrest in Artemia. The Biochemical journal, 470(2), 223–31. https://doi.org/10.1042/BJ20150116
Stigliani, S., Scaruffi, P., Lagazio, C., Persico, L., Carlini, B., Varesio, L., … Corrias, M. V. (2015). Deregulation of focal adhesion pathway mediated by miR-659-3p is implicated in bone marrow infiltration of stage M neuroblastoma patients. Oncotarget, 6(15), 13295–308. https://doi.org/10.18632/oncotarget.3745
Fabbri, E., Montagner, G., Bianchi, N., Finotti, A., Borgatti, M., Lampronti, I., … Gambari, R. (2016). MicroRNA miR-93-5p regulates expression of IL-8 and VEGF in neuroblastoma SK-N-AS cells. Oncology reports, 35(5), 2866–72. https://doi.org/10.3892/or.2016.4676
Chakrabarti, M., Khandkar, M., Banik, N. L., & Ray, S. K. (2012). Alterations in expression of specific microRNAs by combination of 4-HPR and EGCG inhibited growth of human malignant neuroblastoma cells, 1454, 1–13. https://doi.org/10.1016/j.brainres.2012.03.017
Chakrabarti, M., Ai, W., Banik, N. L., & Ray, S. K. (2013). Overexpression of miR-7-1 increases efficacy of green tea polyphenols for induction of apoptosis in human malignant neuroblastoma SH-SY5Y and SK-N-DZ cells. Neurochemical research, 38(2), 420–32. https://doi.org/10.1007/s11064-012-0936-5
Breslow, N., Olshan, A., Beckwith, J. B., & Green, D. M. (1993). Epidemiology of Wilms tumor. Medical and pediatric oncology, 21(3), 172–81. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/7680412
Green, D. M., Beckwith, J. B., Breslow, N. E., Faria, P., Moksness, J., Finklestein, J. Z., … Shochat, S. (1994). Treatment of children with stages II to IV anaplastic Wilms’ tumor: a report from the National Wilms’ Tumor Study Group., 12(10). https://doi.org/10.1200/JCO.1994.12.10.2126
Malkin, D., Sexsmith, E., Yeger, H., Williams, B. R., & Coppes, M. J. (1994). Mutations of the p53 tumor suppressor gene occur infrequently in Wilms’ tumor. Cancer research, 54(8), 2077–9. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/8174107
Hawthorn, L., & Cowell, J. K. (2011). Analysis of Wilms tumors using SNP mapping array-based comparative genomic hybridization, 6(4), e18941. https://doi.org/10.1371/journal.pone.0018941
Major, M. B., Camp, N. D., Berndt, J. D., Yi, X., Goldenberg, S. J., Hubbert, C., … Moon, R. T. (2007). Wilms tumor suppressor WTX negatively regulates WNT/beta-catenin signaling. Science (New York, N.Y.), 316(5827), 1043–6. https://doi.org/10.1126/science/1141515
Kort, E. J., Farber, L., Tretiakova, M., Petillo, D., Furge, K. A., Yang, X. J., … Teh, B. T. (2008). The E2F3-oncomir-1 axis is activated in Wilms’ tumor, 68(11). https://doi.org/10.1158/0008-5472.CAN-08-0592
Hayashita, Y., Osada, H., Tatematsu, Y., Yamada, H., Yanagisawa, K., Tomida, S., … Takahashi, T. (2005). A polycistronic microRNA cluster, miR-17-92, is overexpressed in human lung cancers and enhances cell proliferation, 65(21). https://doi.org/10.1158/0008-5472.CAN-05-2352
Yu, H., & Jove, R. (2004). The STATs of cancer — new molecular targets come of age. Nature Reviews Cancer, 4(2), 97–105. https://doi.org/10.1038/nrc1275
Veronese, A., Lupini, L., Consiglio, J., Visone, R., Ferracin, M., Fornari, F., … Negrini, M. (2010). Oncogenic role of miR-483-3p at the IGF2/483 locus, 70(8). https://doi.org/10.1158/0008-5472.CAN-09-4456
Hong, L., Zhao, X., Shao, X., & Zhu, H. (2017). miR-590 regulates WT1 during proliferation of G401 cells. Molecular medicine reports, 16(1), 247–253. https://doi.org/10.3892/mmr.2017.6561
Cui, M., Liu, W., Zhang, L., Guo, F., Liu, Y., Chen, F., … Wu, R. (2017). Clinicopathological parameters and prognostic relevance of miR-21 and PTEN expression in Wilms’ tumor. Journal of pediatric surgery, 52(8), 1348–1354. https://doi.org/10.1016/j.jpedsurg.2016.12.005
Cui, M., Liu, W., Zhang, L., Guo, F., Liu, Y., Chen, F., … Wu, R. (2017). Over-expression of miR-21 and lower PTEN levels in Wilms’ tumor with aggressive behavior. The Tohoku journal of experimental medicine, 242(1), 43–52. https://doi.org/10.1620/tjem.242.43
Liu, G.-L., Yang, H.-J., Liu, B., & Liu, T. (2017). Effects of microRNA-19b on the proliferation, apoptosis, and migration of Wilms’ tumor cells via the PTEN/PI3K/AKT signaling pathway. Journal of cellular biochemistry, 118(10), 3424–3434. https://doi.org/10.1002/jcb.25999
Zhang, C., Lv, G. Q., Cui, L. F., Guo, C. C., & Liu, Q. E. (2019). MicroRNA-572 targets CDH1 to promote metastasis of Wilms’ tumor. European Review for Medical and Pharmacological Sciences, 23(9), 3709–3717. https://doi.org/10.26355/eurrev_201905_17794
Gong, Y., Zou, B., Chen, J., Ding, L., Li, P., Chen, J., … Li, J. (2019). Potential five-microRNA signature model for the prediction of prognosis in patients with Wilms tumor. Medical Science Monitor, 25, 5435–5444. https://doi.org/10.12659/msm.916230
Wang, H.-F., Zhang, Y.-Y., Zhuang, H.-W., & Xu, M. (2017). MicroRNA-613 attenuates the proliferation, migration and invasion of Wilms’ tumor via targeting FRS2. European review for medical and pharmacological sciences, 21(15), 3360–3369. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/28829507
Liu, Z., He, F., Ouyang, S., Li, Y., Ma, F., Chang, H., … Wu, J. (2019). MiR-140-5p could suppress tumor proliferation and progression by targeting TGFBRI/SMAD2/3 and IGF-1R/AKT signaling pathways in Wilms’ tumor. BMC Cancer, 19(1). https://doi.org/10.1186/s12885-019-5609-1
Imam, J. S., Buddavarapu, K., Lee-Chang, J. S., Ganapathy, S., Camosy, C., Chen, Y., & Rao, M. K. (2010). MicroRNA-185 suppresses tumor growth and progression by targeting the Six1 oncogene in human cancers. Oncogene, 29(35), 4971–9. https://doi.org/10.1038/onc.2010.233
Newman, M. A., Thomson, J. M., & Hammond, S. M. (2008). Lin-28 interaction with the Let-7 precursor loop mediates regulated microRNA processing. RNA (New York, N.Y.), 14(8), 1539–49. https://doi.org/10.1261/rna.1155108
Rakheja, D., Chen, K. S., Liu, Y., Shukla, A. A., Schmid, V., Chang, T.-C., … Amatruda, J. F. (2014). Somatic mutations in DROSHA and DICER1 impair microRNA biogenesis through distinct mechanisms in Wilms tumours, (1), 4802. https://doi.org/10.1038/ncomms5802
Viswanathan, S. R., Powers, J. T., Einhorn, W., Hoshida, Y., Ng, T. L., Toffanin, S., … Daley, G. Q. (2009). Lin28 promotes transformation and is associated with advanced human malignancies. Nature genetics, 41(7), 843–8. https://doi.org/10.1038/ng.392
Urbach, A., Yermalovich, A., Zhang, J., Spina, C. S., Zhu, H., Perez-Atayde, A. R., … Daley, G. Q. (2014). Lin28 sustains early renal progenitors and induces Wilms tumor. Genes & Development, 28(9), 971–982. https://doi.org/10.1101/gad.237149.113
Koller, K., Das, S., Leuschner, I., Korbelius, M., Hoefler, G., & Guertl, B. (2013). Identification of the transcription factor HOXB4 as a novel target of miR-23a. Genes, chromosomes & cancer, 52(8), 709–15. https://doi.org/10.1002/gcc.22066
Koller, K., Pichler, M., Koch, K., Zandl, M., Stiegelbauer, V., Leuschner, I., … Guertl, B. (2014). Nephroblastomas show low expression of microR-204 and high expression of its target, the oncogenic transcription factor MEIS1. Pediatric and developmental pathology: the official journal of the Society for Pediatric Pathology and the Paediatric Pathology Society, 17(3), 169–75. https://doi.org/10.2350/13-01-1288-OA.1
Jiang, X., & Li, H. (2018). MiR-1180-5p regulates apoptosis of Wilms’ tumor by targeting<em> p</em>73. OncoTargets and Therapy, Volume 11, 823–831. https://doi.org/10.2147/OTT.S148684
Senanayake, U., Das, S., Vesely, P., Alzoughbi, W., Frohlich, L. F., Chowdhury, P., … Guertl, B. (2012). miR-192, miR-194, miR-215, miR-200c and miR-141 are downregulated and their common target ACVR2B is strongly expressed in renal childhood neoplasms. Carcinogenesis, 33(5), 1014–1021. https://doi.org/10.1093/carcin/bgs126
Liu, K., He, B., Xu, J., Li, Y., Guo, C., Cai, Q., & Wang, S. (2019). MiR-483-5p targets MKNK1 to suppress Wilms’ tumor cell proliferation and apoptosis in vitro and in vivo. Medical Science Monitor, 25, 1459–1468. https://doi.org/10.12659/MSM.913005
Allen, K. E., & Weiss, G. J. (2010). Resistance may not be futile: microRNA biomarkers for chemoresistance and potential therapeutics, 9(12). https://doi.org/10.1158/1535-7163.MCT-10-0397
Schmitt, J., Backes, C., Nourkami-Tutdibi, N., Leidinger, P., Deutscher, S., Beier, M., … Meese, E. (2012). Treatment-independent miRNA signature in blood of wilms tumor patients, 13(1), 379. https://doi.org/10.1186/1471-2164-13-379
Watson, J. A., Bryan, K., Williams, R., Popov, S., Vujanic, G., Coulomb, A., … O’Sullivan, M. (2013). miRNA profiles as a predictor of chemoresponsiveness in Wilms’ tumor blastema. PLoS ONE, 8(1), e53417. https://doi.org/10.1371/journal.pone.0053417
Torrezan, G. T., Ferreira, E. N., Nakahata, A. M., Barros, B. D. F., Castro, M. T. M., Correa, B. R., … Carraro, D. M. (2014). Recurrent somatic mutation in DROSHA induces microRNA profile changes in Wilms tumour, 5(1), 4039. https://doi.org/10.1038/ncomms5039
Wegert, J., Ishaque, N., Vardapour, R., Geörg, C., Gu, Z., Bieg, M., … Gessler, M. (2015). Mutations in the SIX1/2 pathway and the DROSHA/DGCR8 miRNA microprocessor complex underlie high-risk blastemal type Wilms tumors. Cancer cell, 27(2), 298–311. https://doi.org/10.1016/j.ccell.2015.01.002
Spreafico, F., Ciceri, S., Gamba, B., Torri, F., Terenziani, M., Collini, P., … Perotti, D. (2016). Chromosomal anomalies at 1q, 3, 16q, and mutations of SIX1 and DROSHA genes underlie Wilms tumor recurrences. Oncotarget, 7(8), 8908–15. https://doi.org/10.18632/oncotarget.6950
Lam, A. K.-Y. (2017). Update on adrenal tumours in 2017 World Health Organization (WHO) of endocrine tumours, 28(3), 213–227. https://doi.org/10.1007/s12022-017-9484-5
Khanna, S., Priya, R., Bhartiya, S. K., Basu, S., & Shukla, V. K. (2015). Adrenal tumors: an experience of 10 years in a single surgical unit, 52(3). https://doi.org/10.4103/0019-509X.176749
Igaz, P., Igaz, I., Nagy, Z., Nyírő, G., Szabó, P. M., Falus, A., … Rácz, K. (2015). MicroRNAs in adrenal tumors: relevance for pathogenesis, diagnosis, and therapy. Cellular and Molecular Life Sciences, 72(3), 417–428. https://doi.org/10.1007/s00018-014-1752-7
Feinmesser, M., Benbassat, C., Meiri, E., Benjamin, H., Lebanony, D., Lebenthal, Y., … Spector, Y. (2015). Specific microRNAs differentiate adrenocortical adenomas from carcinomas and correlate with weiss histopathologic system. Applied Immunohistochemistry & Molecular Morphology, 23(7), 522–531. https://doi.org/10.1097/PAI.0000000000000117
Bimpaki, E. I., Iliopoulos, D., Moraitis, A., & Stratakis, C. A. (2009). MicroRNA signature in massive macronodular adrenocortical disease and implications for adrenocortical tumourigenesis. Clinical Endocrinology, 72(6), 744–751. https://doi.org/10.1111/j.1365-2265.2009.03725.x
Koduru, S. V., Leberfinger, A. N., & Ravnic, D. J. (2017). Small non-coding RNA abundance in adrenocortical carcinoma: a footprint of a rare cancer. Journal of Genomics, 5, 99–118. https://doi.org/10.7150/jgen.22060
Agosta, C., Laugier, J., Guyon, L., Denis, J., Bertherat, J., Libé, R., … Cherradi, N. (2018). MiR-483-5p and miR-139-5p promote aggressiveness by targeting N-myc downstream-regulated gene family members in adrenocortical cancer. International Journal of Cancer, 143(4), 944–957. https://doi.org/10.1002/ijc.31363
Robertson, S., MacKenzie, S. M., Alvarez-Madrazo, S., Diver, L. A., Lin, J., Stewart, P. M., … Davies, E. (2013). MicroRNA-24 is a novel regulator of aldosterone and cortisol production in the human adrenal cortex. Hypertension, 62(3), 572–578. https://doi.org/10.1161/HYPERTENSIONAHA.113.01102
Robertson, S., Diver, L. A., Alvarez-Madrazo, S., Livie, C., Ejaz, A., Fraser, R., … Davies, E. (2017). Regulation of corticosteroidogenic genes by microRNAs, 2017. https://doi.org/10.1155/2017/2021903
Nusrin, S., Tong, S. K. H. H., Chaturvedi, G., Wu, R. S. S. S., Giesy, J. P., & Kong, R. Y. C. C. (2014). Regulation of CYP11B1 and CYP11B2 steroidogenic genes by hypoxia-inducible miR-10b in H295R cells, 85(2). https://doi.org/10.1016/j.marpolbul.2014.04.002
Kwok, G. T. Y., Zhao, J. T., Glover, A. R., Gill, A. J., Clifton-Bligh, R., Robinson, B. G., … Sidhu, S. B. (2019). microRNA-431 as a chemosensitizer and potentiator of drug activity in adrenocortical carcinoma. The Oncologist, 24(6), e241–e250. https://doi.org/10.1634/theoncologist.2018-0849
El Wakil, A., Doghman, M., Latre De Late, P., Zambetti, G. P., Figueiredo, B. C., & Lalli, E. (2011). Genetics and genomics of childhood adrenocortical tumors. Molecular and Cellular Endocrinology, 336(1–2), 169–173. https://doi.org/10.1016/j.mce.2010.11.008
Faria, A. M., & Almeida, M. Q. (2012). Differences in the molecular mechanisms of adrenocortical tumorigenesis between children and adults. Molecular and Cellular Endocrinology, 351(1), 52–57. https://doi.org/10.1016/j.mce.2011.09.040
Doghman, M., Wakil, A. El, Cardinaud, B., Thomas, E., Wang, J., Zhao, W., … Lalli, E. (2010). Regulation of insulin-like growth factor–mammalian target of rapamycin signaling by microRNA in childhood adrenocortical tumors, 70(11). https://doi.org/10.1158/0008-5472.CAN-09-3970
Almeida, M. Q., Fragoso, M. C. B. V., Lotfi, C. F. P., Santos, M. G., Nishi, M. Y., Costa, M. H. S., … Latronico, A. C. (2008). Expression of insulin-like growth factor-II and its receptor in pediatric and adult adrenocortical tumors, 93(9). https://doi.org/10.1210/jc.2008-0065
Ribeiro, T. C., Jorge, A. A., Almeida, M. Q., Mariani, B. M. de P., Nishi, M. Y., Mendonca, B. B., … Latronico, A. C. (2014). Amplification of the insulin-like growth factor 1 receptor gene is a rare event in adrenocortical adenocarcinomas: searching for potential mechanisms of overexpression, 2014. https://doi.org/10.1155/2014/936031
Finegold, M. J., Lopez-Terrada, D. H., Bowen, J., Washington, M. K., Qualman, S. J., & College of American Pathologists. (2007). Protocol for the examination of specimens from pediatric patients with hepatoblastoma. Archives of pathology & laboratory medicine, 131(4), 520–9. https://doi.org/10.1043/1543-2165(2007)131[520:PFTEOS]2.0.CO;2
Spector, L. G., & Birch, J. (2012). The epidemiology of hepatoblastoma. Pediatric blood & cancer, 59(5), 776–9. https://doi.org/10.1002/pbc.24215
Purcell, R., Childs, M., Maibach, R., Miles, C., Turner, C., Zimmermann, A., … Sullivan, M. (2012). Potential biomarkers for hepatoblastoma: results from the SIOPEL-3 study. European journal of cancer (Oxford, England: 1990), 48(12), 1853–9. https://doi.org/10.1016/j.ejca.2011.10.019
Rodriguez-Galindo, C., Krailo, M., Frazier, L., Chintagumpala, M., Amatruda, J., Katzenstein, H., … COG Rare Tumors Disease Committee. (2013). Children’s Oncology Group’s 2013 blueprint for research: rare tumors. Pediatric blood & cancer, 60(6), 1016–21. https://doi.org/10.1002/pbc.24428
Linabery, A. M., & Ross, J. A. (2008). Trends in childhood cancer incidence in the U.S. (1992–2004). Cancer, 112(2), 416–32. https://doi.org/10.1002/cncr.23169
Anna, C. H., Sills, R. C., Foley, J. F., Stockton, P. S., Ton, T.-V., & Devereux, T. R. (2000). Catenin mutations and protein accumulation in all hepatoblastomas examined from B6C3F1 mice treated with anthraquinone or oxazepam. CANCER RESEARCH (Vol. 60). Retrieved from http://cancerres.aacrjournals.org/content/canres/60/11/2864.full.pdf
Armengol, C., Cairo, S., Fabre, M., & Buendia, M. A. (2011). Wnt signaling and hepatocarcinogenesis: the hepatoblastoma model. The international journal of biochemistry & cell biology, 43(2), 265–70. https://doi.org/10.1016/j.biocel.2009.07.012
Czauderna, P., Lopez-Terrada, D., Hiyama, E., Häberle, B., Malogolowkin, M. H., & Meyers, R. L. (2014). Hepatoblastoma state of the art. Current Opinion in Pediatrics, 26(1), 19–28. https://doi.org/10.1097/MOP.0000000000000046
López-Terrada, D., Alaggio, R., de Dávila, M. T., Czauderna, P., Hiyama, E., Katzenstein, H., … Children’s Oncology Group Liver Tumor Committee. (2014). Towards an international pediatric liver tumor consensus classification: proceedings of the Los Angeles COG liver tumors symposium. Modern pathology: an official journal of the United States and Canadian Academy of Pathology, Inc, 27(3), 472–91. https://doi.org/10.1038/modpathol.2013.80
Maibach, R., Roebuck, D., Brugieres, L., Capra, M., Brock, P., Dall’Igna, P., … Perilongo, G. (2012). Prognostic stratification for children with hepatoblastoma: the SIOPEL experience. European journal of cancer (Oxford, England: 1990), 48(10), 1543–9. https://doi.org/10.1016/j.ejca.2011.12.011
Darbari, A., Sabin, K. M., Shapiro, C. N., & Schwarz, K. B. (2003). Epidemiology of primary hepatic malignancies in U.S. children. https://doi.org/10.1053/jhep.2003.50375
Heck, J. E., Meyers, T. J., Lombardi, C., Park, A. S., Cockburn, M., Reynolds, P., & Ritz, B. (2013). Case-control study of birth characteristics and the risk of hepatoblastoma. Cancer epidemiology, 37(4), 390–5. https://doi.org/10.1016/j.canep.2013.03.004
Ries, L., Harkins, D., Krapcho, M., Mariotto, A., Miller, B., Feuer, E., … Edwards, B. (2006). SEER Cancer Statistics Review, 1975–2003. Public Health Faculty Publications. Retrieved from https://scholarworks.gsu.edu/iph_facpub/132
Hughes, L. J., & Michels, V. V. (1992). Risk of hepatoblastoma in familial adenomatous polyposis. American Journal of Medical Genetics, 43(6), 1023–1025. https://doi.org/10.1002/ajmg.1320430621
Johnson, K. J., Williams, K. S., Ross, J. A., Krailo, M. D., Tomlinson, G. E., Malogolowkin, M. H., … Spector, L. G. (2013). Parental tobacco and alcohol use and risk of hepatoblastoma in offspring: a report from the Children’s Oncology Group. Cancer Epidemiology Biomarkers & Prevention, 22(10), 1837–1843. https://doi.org/10.1158/1055-9965.EPI-13-0432
DeBaun, M. R., & Tucker, M. A. (1998). Risk of cancer during the first four years of life in children from The Beckwith-Wiedemann Syndrome Registry. The Journal of Pediatrics, 132(3), 398–400. https://doi.org/10.1016/S0022-3476(98)70008-3
Ansell, P., Mitchell, C. D., Roman, E., Simpson, J., Birch, J. M., & Eden, T. O. B. (2005). Relationships between perinatal and maternal characteristics and hepatoblastoma: a report from the UKCCS. European journal of cancer (Oxford, England: 1990), 41(5), 741–8. https://doi.org/10.1016/j.ejca.2004.10.024
Williams, C. L., Bunch, K. J., Stiller, C. A., Murphy, M. F. G., Botting, B. J., Wallace, W. H., … Sutcliffe, A. G. (2013). Cancer risk among children born after assisted conception. New England Journal of Medicine, 369(19), 1819–1827. https://doi.org/10.1056/NEJMoa1301675
Ortega, J. A., Douglass, E. C., Feusner, J. H., Reynolds, M., Quinn, J. J., Finegold, M. J., … Krailo, M. D. (2000). Randomized comparison of cisplatin/vincristine/fluorouracil and cisplatin/continuous infusion doxorubicin for treatment of pediatric hepatoblastoma: a report from the Children’s Cancer Group and the Pediatric Oncology Group. Journal of clinical oncology: official journal of the American Society of Clinical Oncology, 18(14), 2665–75. https://doi.org/10.1200/JCO.2000.18.14.2665
Kremer, N., Walther, A. E., & Tiao, G. M. (2014). Management of hepatoblastoma. Current Opinion in Pediatrics, 26(3), 362–369. https://doi.org/10.1097/MOP.0000000000000081
Czauderna, P., & Garnier, H. (2018). Hepatoblastoma: current understanding, recent advances, and controversies. F1000Research, 7, 53. https://doi.org/10.12688/f1000research.12239.1
Haeberle, B., & Schweinitz, D. von. (2012). Treatment of hepatoblastoma in the German cooperative pediatric liver tumor studies. Frontiers in bioscience (Elite edition), 4, 493–8. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/22201890
Magrelli, A., Azzalin, G., Salvatore, M., Viganotti, M., Tosto, F., Colombo, T., … Taruscio, D. (2009). Altered microRNA expression patterns in hepatoblastoma patients. Translational oncology, 2(3), 157–63. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/19701500
Cairo, S., Wang, Y., de Reynies, A., Duroure, K., Dahan, J., Redon, M.-J., … Buendia, M.-A. (2010). Stem cell-like micro-RNA signature driven by Myc in aggressive liver cancer. Proceedings of the National Academy of Sciences, 107(47), 20471–20476. https://doi.org/10.1073/pnas.1009009107
He, J., Guo, X., Sun, L., Wang, N., & Bao, J. (2016). Regulatory network analysis of genes and microRNAs in human hepatoblastoma. Oncology Letters, 12(5), 4099–4106. https://doi.org/10.3892/ol.2016.5196
von Frowein, J., Pagel, P., Kappler, R., von Schweinitz, D., Roscher, A., & Schmid, I. (2011). MicroRNA-492 is processed from the keratin 19 gene and up-regulated in metastatic hepatoblastoma. Hepatology, 53(3), 833–842. https://doi.org/10.1002/hep.24125
von Frowein, J., Hauck, S. M., Kappler, R., Pagel, P., Fleischmann, K. K., Magg, T., … Schmid, I. (2018). MiR-492 regulates metastatic properties of hepatoblastoma via CD44. Liver International, 38(7), 1280–1291. https://doi.org/10.1111/liv.13687
Indersie, E., Lesjean, S., Hooks, K. B., Sagliocco, F., Ernault, T., Cairo, S., … Grosset, C. F. (2017). MicroRNA therapy inhibits hepatoblastoma growth in vivo by targeting β-catenin and Wnt signaling. Hepatology communications, 1(2), 168–183. https://doi.org/10.1002/hep4.1029
Ecevit, Ç. O., Aktaş, S., Tosun Yildirim, H., Demiraǧ, B., Erbay, A., Karaca, I., … Olgun, N. (2019). MicroRNA-17, microRNA-19b, microRNA-146a, microRNA-302d expressions in hepatoblastoma and clinical importance. Journal of Pediatric Hematology/Oncology, 41(1), 7–12. https://doi.org/10.1097/MPH.0000000000001234
Gyugos, M., Lendvai, G., Kenessey, I., Schlachter, K., Halász, J., Nagy, P., … Kiss, A. (2014). MicroRNA expression might predict prognosis of epithelial hepatoblastoma. Virchows Archiv: an international journal of pathology, 464(4), 419–27. https://doi.org/10.1007/s00428-014-1549-y
Jiao, C., Zhu, A., Jiao, X., Ge, J., & Xu, X. (2016). Combined low miR-34s are associated with unfavorable prognosis in children with hepatoblastoma: a Chinese population-based study. Journal of pediatric surgery, 51(8), 1355–61. https://doi.org/10.1016/j.jpedsurg.2016.02.091
Liu, S., Xie, F., Xiang, X., Liu, S., Dong, S., Qu, K., & Lin, T. (2017). Identification of differentially expressed genes, lncRNAs and miRNAs which are associated with tumor malignant phenotypes in hepatoblastoma patients. Oncotarget, 8(57), 97554–97564. https://doi.org/10.18632/oncotarget.22181
Kelly, D., Sharif, K., Brown, R. M., & Morland, B. (2015). Hepatocellular carcinoma in children. Clinics in Liver Disease, 19(2), 433–447. https://doi.org/10.1016/j.cld.2015.01.010
Schmid, I., & von Schweinitz, D. (2017). Pediatric hepatocellular carcinoma: challenges and solutions. Journal of hepatocellular carcinoma, 4, 15–21. https://doi.org/10.2147/JHC.S94008
Chen, E., Xu, X., Liu, R., & Liu, T. (2018). Small but heavy role: microRNAs in hepatocellular carcinoma progression. BioMed Research International, 2018, 1–9. https://doi.org/10.1155/2018/6784607
ATCC: The Global Bioresource Center. (n.d.). 2018. Retrieved October 10, 2018, from https://www.atcc.org/
Gramantieri, L., Ferracin, M., Fornari, F., Veronese, A., Sabbioni, S., Liu, C.-G., … Negrini, M. (2007). Cyclin G1 is a target of miR-122a, a microRNA frequently down-regulated in human hepatocellular carcinoma. Cancer Research, 67(13), 6092–6099. https://doi.org/10.1158/0008-5472.CAN-06-4607
Lin, C. J.-F., Gong, H.-Y., Tseng, H.-C., Wang, W.-L., & Wu, J.-L. (2008). miR-122 targets an anti-apoptotic gene, Bcl-w, in human hepatocellular carcinoma cell lines. Biochemical and biophysical research communications, 375(3), 315–20. https://doi.org/10.1016/j.bbrc.2008.07.154
Xu, Q., Zhang, M., Tu, J., Pang, L., Cai, W., & LIU, X. (2015). MicroRNA-122 affects cell aggressiveness and apoptosis by targeting PKM2 in human hepatocellular carcinoma, 34(4). https://doi.org/10.3892/or.2015.4175
Xu, J., Zhu, X., Wu, L., Yang, R., Yang, Z., Wang, Q., & Wu, F. (2012). MicroRNA-122 suppresses cell proliferation and induces cell apoptosis in hepatocellular carcinoma by directly targeting Wnt/β-catenin pathway, 32(5). https://doi.org/10.1111/j.1478-3231.2011.02750.x
Kumar, S., Batra, A., Kanthaje, S., Ghosh, S., & Chakraborti, A. (2017). Crosstalk between microRNA-122 and FOX family genes in HepG2 cells, 242(4), 436–440. https://doi.org/10.1177/1535370216681548
Huang, H., Zhu, Y., & Li, S. (2015). MicroRNA-122 mimic transfection contributes to apoptosis in HepG2 cells. Molecular medicine reports, 12(5), 6918–24. https://doi.org/10.3892/mmr.2015.4254
Shu, X.-L., Fan, C.-B., Long, B., Zhou, X., & Wang, Y. (2016). The anti-cancer effects of cisplatin on hepatic cancer are associated with modulation of miRNA-21 and miRNA-122 expression. European review for medical and pharmacological sciences, 20(21), 4459–4465. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/27874954
Yahya, S. M. M., Fathy, S. A., El-Khayat, Z. A., El-Toukhy, S. E., Hamed, A. R., Hegazy, M. G. A., & Nabih, H. K. (2018). Possible role of microRNA-122 in modulating multidrug resistance of hepatocellular carcinoma. Indian journal of clinical biochemistry: IJCB, 33(1), 21–30. https://doi.org/10.1007/s12291-017-0651-8
Zeng, X., Yuan, Y., Wang, T., Wang, H., Hu, X., Fu, Z., … Lu, G. (2017). Targeted imaging and induction of apoptosis of drug-resistant hepatoma cells by miR-122-loaded graphene-InP nanocompounds. Journal of Nanobiotechnology, 15(1), 9. https://doi.org/10.1186/s12951-016-0237-2
LI, A., QIAN, J., HE, J., ZHANG, Q., ZHAI, A., SONG, W., … ZHANG, F. (2013). Modulation of miR-122 expression affects the interferon response in human hepatoma cells. Molecular Medicine Reports, 7(2), 585–590. https://doi.org/10.3892/mmr.2012.1233
Najafi, Z., Sharifi, M., & Javadi, G. (2015). Degradation of miR-21 induces apoptosis and inhibits cell proliferation in human hepatocellular carcinoma. Cancer gene therapy, 22(11), 530–5. https://doi.org/10.1038/cgt.2015.51
Connolly, E. C., Van Doorslaer, K., Rogler, L. E., & Rogler, C. E. (2010). Overexpression of miR-21 promotes an in vitro metastatic phenotype by targeting the tumor suppressor RHOB. Molecular cancer research: MCR, 8(5), 691–700. https://doi.org/10.1158/1541-7786.MCR-09-0465
Hu, S., Tao, R., Wang, S., Wang, C., Zhao, X., Zhao, H., … Gao, Y. (2015). MicroRNA-21 promotes cell proliferation in human hepatocellular carcinoma partly by targeting HEPN1, 36(7). https://doi.org/10.1007/s13277-015-3213-9
Yin, D., Wang, Y., Sai, W., Zhang, L., Miao, Y., Cao, L., … Yang, L. (2016). HBx-induced miR-21 suppresses cell apoptosis in hepatocellular carcinoma by targeting interleukin-12. Oncology reports, 36(4), 2305–12. https://doi.org/10.3892/or.2016.5026
Xu, G., Zhang, Y., Wei, J., Jia, W., Ge, Z., Zhang, Z., & Liu, X. (2013). MicroRNA-21 promotes hepatocellular carcinoma HepG2 cell proliferation through repression of mitogen-activated protein kinase-kinase 3. BMC cancer, 13(1), 469. https://doi.org/10.1186/1471-2407-13-469
Zhu, Q., Wang, Z., Hu, Y., Li, J., Li, X., Zhou, L., & Huang, Y. (2012). miR-21 promotes migration and invasion by the miR-21-PDCD4-AP-1 feedback loop in human hepatocellular carcinoma. Oncology reports, 27(5), 1660–8. https://doi.org/10.3892/or.2012.1682
Tomimaru, Y., Eguchi, H., Nagano, H., Wada, H., Tomokuni, A., Kobayashi, S., … Mori, M. (2010). MicroRNA-21 induces resistance to the anti-tumour effect of interferon-α/5-fluorouracil in hepatocellular carcinoma cells, 103(10). https://doi.org/10.1038/sj.bjc.6605958
Zhu, X., Wu, L., Yao, J., Jiang, H., Wang, Q., Yang, Z., & Wu, F. (2015). MicroRNA let-7c inhibits cell proliferation and induces cell cycle arrest by targeting CDC25A in human hepatocellular carcinoma. PloS one, 10(4), e0124266. https://doi.org/10.1371/journal.pone.0124266
Long, J., Jiang, C., Liu, B., Fang, S., & Kuang, M. (2016). MicroRNA-15a-5p suppresses cancer proliferation and division in human hepatocellular carcinoma by targeting BDNF. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine, 37(5), 5821–8. https://doi.org/10.1007/s13277-015-4427-6
Wang, Y., Zhao, Y.-R., Zhang, A.-Y., Ma, J., Wang, Z.-Z., & Zhang, X. (2017). Targeting of miR-20a against CFLAR to potentiate TRAIL-induced apoptotic sensitivity in HepG2 cells. European review for medical and pharmacological sciences, 21(9), 2087–2097. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/28537677
Fan, M.-Q., Huang, C.-B., Gu, Y., Xiao, Y., Sheng, J.-X., & Zhong, L. (2013). Decrease expression of microRNA-20a promotes cancer cell proliferation and predicts poor survival of hepatocellular carcinoma. Journal of experimental & clinical cancer research: CR, 32(1), 21. https://doi.org/10.1186/1756-9966-32-21
Li, S., Li, J., Fei, B.-Y., Shao, D., Pan, Y., Mo, Z.-H., … Chen, L. (2015). MiR-27a promotes hepatocellular carcinoma cell proliferation through suppression of its target gene peroxisome proliferator-activated receptor γ. Chinese medical journal, 128(7), 941–7. https://doi.org/10.4103/0366-6999.154302
Zhao, N., Sun, H., Sun, B., Zhu, D., Zhao, X., Wang, Y., … Li, X. (2016). miR-27a-3p suppresses tumor metastasis and VM by down-regulating VE-cadherin expression and inhibiting EMT: an essential role for Twist-1 in HCC, 6(1), 23091. https://doi.org/10.1038/srep23091
Li, J.-M., Zhou, J., Xu, Z., Huang, H.-J., Chen, M.-J., & Ji, J.-S. (2018). MicroRNA-27a-3p inhibits cell viability and migration through down-regulating DUSP16 in hepatocellular carcinoma, 119(7), 5143–5152. https://doi.org/10.1002/jcb.26526
Wu, X.-J., Li, Y., Liu, D., Zhao, L.-D., Bai, B., & Xue, M.-H. (2013). miR-27a as an oncogenic microRNA of hepatitis B virus- related hepatocellular carcinoma. Asian Pacific journal of cancer prevention: APJCP, 14(2), 885–9. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/23621256
Wang, C.-M., Wang, Y., Fan, C.-G., Xu, F.-F., Sun, W.-S., Liu, Y.-G., & Jia, J.-H. (2011). miR-29c targets TNFAIP3, inhibits cell proliferation and induces apoptosis in hepatitis B virus-related hepatocellular carcinoma. Biochemical and biophysical research communications, 411(3), 586–92. https://doi.org/10.1016/j.bbrc.2011.06.191
He, R., Yang, L., Lin, X., Chen, X., Lin, X., Wei, F., … Chen, G. (2015). MiR-30a-5p suppresses cell growth and enhances apoptosis of hepatocellular carcinoma cells via targeting AEG-1. International journal of clinical and experimental pathology, 8(12), 15632–41. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26884832
Li, W.-F., Dai, H., Ou, Q., Zuo, G.-Q., & Liu, C.-A. (2016). Overexpression of microRNA-30a-5p inhibits liver cancer cell proliferation and induces apoptosis by targeting MTDH/PTEN/AKT pathway. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine, 37(5), 5885–95. https://doi.org/10.1007/s13277-015-4456-1
Zhou, K., Luo, X., Wang, Y., Cao, D., & Sun, G. (2017). MicroRNA-30a suppresses tumor progression by blocking Ras/Raf/MEK/ERK signaling pathway in hepatocellular carcinoma. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 93, 1025–1032. https://doi.org/10.1016/j.biopha.2017.07.029
Jiang, T., Li, M., Li, Q., Guo, Z., Sun, X., Zhang, X., … Xiao, P. (2017). MicroRNA-98-5p inhibits cell proliferation and induces cell apoptosis in hepatocellular carcinoma via targeting IGF2BP1. Oncology research, 25(7), 1117–1127. https://doi.org/10.3727/096504016X14821952695683
Zhao, C., Li, Y., Zhang, M., Yang, Y., & Chang, L. (2015). miR-126 inhibits cell proliferation and induces cell apoptosis of hepatocellular carcinoma cells partially by targeting Sox2. Human cell, 28(2), 91–9. https://doi.org/10.1007/s13577-014-0105-z
Zheng, C., Li, J., Wang, Q., Liu, W., Zhou, J., Liu, R., … Cao, K. (2015). MicroRNA-195 functions as a tumor suppressor by inhibiting CBX4 in hepatocellular carcinoma. Oncology reports, 33(3), 1115–22. https://doi.org/10.3892/or.2015.3734
Lin, L., Liang, H., Wang, Y., Yin, X., Hu, Y., Huang, J., … Chen, X. (2014). microRNA-141 inhibits cell proliferation and invasion and promotes apoptosis by targeting hepatocyte nuclear factor-3β in hepatocellular carcinoma cells. BMC cancer, 14(1), 879. https://doi.org/10.1186/1471-2407-14-879
Shi, L., Wu, L., Chen, Z., Yang, J., Chen, X., Yu, F., … Lin, X. (2015). MiR-141 activates Nrf2-dependent antioxidant pathway via down-regulating the expression of Keap1 conferring the resistance of hepatocellular carcinoma cells to 5-fluorouracil. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology, 35(6), 2333–48. https://doi.org/10.1159/000374036
Lou, K., Chen, N., Li, Z., Zhang, B., Wang, X., Chen, Y., … Wang, H. (2017). MicroRNA-142-5p overexpression inhibits cell growth and induces apoptosis by regulating FOXO in hepatocellular carcinoma cells. Oncology research, 25(1), 65–73. https://doi.org/10.3727/096504016X14719078133366
Zhang, J., Shi, Y., Hong, D., Song, M., Huang, D., Wang, C., & Zhao, G. (2015). MiR-148b suppresses cell proliferation and invasion in hepatocellular carcinoma by targeting WNT1/β-catenin pathway. Scientific reports, 5(1), 8087. https://doi.org/10.1038/srep08087
Zhao, Y., Li, F., Zhang, X., Liu, A., Qi, J., Cui, H., & Zhao, P. (2015). MicroRNA-194 acts as a prognostic marker and inhibits proliferation in hepatocellular carcinoma by targeting MAP4K4. International journal of clinical and experimental pathology, 8(10), 12446–54. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26722431
Yang, Y., Li, M., Chang, S., Wang, L., Song, T., Gao, L., … Huang, C. (2014). MicroRNA-195 acts as a tumor suppressor by directly targeting Wnt3a in HepG2 hepatocellular carcinoma cells. Molecular medicine reports, 10(5), 2643–8. https://doi.org/10.3892/mmr.2014.2526
Rebucci, M., Sermeus, A., Leonard, E., Delaive, E., Dieu, M., Fransolet, M., … Michiels, C. (2015). miRNA-196b inhibits cell proliferation and induces apoptosis in HepG2 cells by targeting IGF2BP1. Molecular cancer, 14(1), 79. https://doi.org/10.1186/s12943-015-0349-6
Gui, R., Huang, R., Zhang, J.-H., Wen, X.-H., & Nie, X.-M. (2016). MicroRNA-199a-5p inhibits VEGF-induced tumorigenesis through targeting oxidored-nitro domain-containing protein 1 in human HepG2 cells. Oncology reports, 35(4), 2216–22. https://doi.org/10.3892/or.2016.4550
Dou, C., Wang, Y., Li, C., Liu, Z., Jia, Y., Li, Q., … Tu, K. (2015). MicroRNA-212 suppresses tumor growth of human hepatocellular carcinoma by targeting FOXA1. Oncotarget, 6(15), 13216–28. https://doi.org/10.18632/oncotarget.3916
Tu, H., Wei, G., Cai, Q., Chen, X., Sun, Z., Cheng, C., … Zeng, T. (2015). MicroRNA-212 inhibits hepatocellular carcinoma cell proliferation and induces apoptosis by targeting FOXA1. OncoTargets and therapy, 8, 2227–35. https://doi.org/10.2147/OTT.S87976
Zheng, W.-W., Zhou, J., Zhang, C.-H., & Liu, X.-S. (2016). MicroRNA-216b is downregulated in hepatocellular carcinoma and inhibits HepG2 cell growth by targeting Forkhead box protein M1. European review for medical and pharmacological sciences, 20(12), 2541–50. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/27383303
Dong, Z., Qi, R., Guo, X., Zhao, X., Li, Y., Zeng, Z., … Lu, Y. (2017). MiR-223 modulates hepatocellular carcinoma cell proliferation through promoting apoptosis via the Rab1-mediated mTOR activation. Biochemical and biophysical research communications, 483(1), 630–637. https://doi.org/10.1016/j.bbrc.2016.12.091
Fang, L., Zhang, H.-B., Li, H., Fu, Y., & Yang, G.-S. (2012). miR-548c-5p inhibits proliferation and migration and promotes apoptosis in CD90(+) HepG2 cells. Radiology and oncology, 46(3), 233–41. https://doi.org/10.2478/v10019-012-0025-z
Guo, W., Tan, W., Liu, S., Huang, X., Lin, J., Liang, R., … Wang, C. (2015). MiR-570 inhibited the cell proliferation and invasion through directly targeting B7-H1 in hepatocellular carcinoma. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine, 36(11), 9049–57. https://doi.org/10.1007/s13277-015-3644-3
Shan, X., Miao, Y., Fan, R., Qian, H., Chen, P., Liu, H., … Zhou, F. (2013). MiR-590-5P inhibits growth of HepG2 cells via decrease of S100A10 expression and inhibition of the Wnt pathway, 14(4). https://doi.org/10.3390/ijms14048556
Huang, Y., Liu, J., Fan, L., Wang, F., Yu, H., Wei, W., & Sun, G. (2016). miR-663 overexpression induced by endoplasmic reticulum stress modulates hepatocellular carcinoma cell apoptosis via transforming growth factor beta 1. OncoTargets and therapy, 9, 1623–33. https://doi.org/10.2147/OTT.S96902
Chu, Y., Fan, W., Guo, W., Zhang, Y., Wang, L., Guo, L., … Xu, G. (2017). miR-1247-5p functions as a tumor suppressor in human hepatocellular carcinoma by targeting Wnt3. Oncology reports, 38(1), 343–351. https://doi.org/10.3892/or.2017.5702
Liu, Y., Liang, H., & Jiang, X. (2015). MiR-1297 promotes apoptosis and inhibits the proliferation and invasion of hepatocellular carcinoma cells by targeting HMGA2. International journal of molecular medicine, 36(5), 1345–52. https://doi.org/10.3892/ijmm.2015.2341
Liu, C., Wang, C., Wang, J., & Huang, H. (2016). miR-1297 promotes cell proliferation by inhibiting RB1 in liver cancer. Oncology letters, 12(6), 5177–5182. https://doi.org/10.3892/ol.2016.5326
Li, D., Yang, P., Li, H., Cheng, P., Zhang, L., Wei, D., … Zhang, T. (2012). MicroRNA-1 inhibits proliferation of hepatocarcinoma cells by targeting endothelin-1. Life sciences, 91(11–12), 440–447. https://doi.org/10.1016/j.lfs.2012.08.015
WEI, W., HU, Z., FU, H., TIE, Y., ZHANG, H., WU, Y., & ZHENG, X. (2012). MicroRNA-1 and microRNA-499 downregulate the expression of the ets1 proto-oncogene in HepG2 cells. Oncology Reports, 28(2), 701–706. https://doi.org/10.3892/or.2012.1850
Li, Q., Zhou, L., Yang, F., Wang, G., Zheng, H., Wang, D., … Dou, K. (2012). MicroRNA-10b promotes migration and invasion through CADM1 in human hepatocellular carcinoma cells. Tumor Biology, 33(5), 1455–1465. https://doi.org/10.1007/s13277-012-0396-1
Zeng, Y.-B., Liang, X.-H., Zhang, G.-X., Jiang, N., Zhang, T., Huang, J.-Y., … Zeng, X.-C. (2016). miRNA-135a promotes hepatocellular carcinoma cell migration and invasion by targeting forkhead box O1. Cancer cell international, 16(1), 63. https://doi.org/10.1186/s12935-016-0328-z
Han, Z.-B., Chen, H.-Y., Fan, J.-W., Wu, J.-Y., Tang, H.-M., & Peng, Z.-H. (2012). Up-regulation of microRNA-155 promotes cancer cell invasion and predicts poor survival of hepatocellular carcinoma following liver transplantation. Journal of Cancer Research and Clinical Oncology, 138(1), 153–161. https://doi.org/10.1007/s00432-011-1076-z
Xie, Q., Chen, X., Lu, F., Zhang, T., Hao, M., Wang, Y., … Zhuang, H. (2012). Aberrant expression of microRNA 155 may accelerate cell proliferation by targeting sex-determining region Y box 6 in hepatocellular carcinoma. Cancer, 118(9), 2431–42. https://doi.org/10.1002/cncr.26566
Shan, S. W., Fang, L., Shatseva, T., Rutnam, Z. J., Yang, X., Du, W., … Yang, B. B. (2013). Mature miR-17-5p and passenger miR-17-3p induce hepatocellular carcinoma by targeting PTEN, GalNT7 and vimentin in different signal pathways, 126. https://doi.org/10.1242/jcs.122895
Zhang, X., Yu, B., Zhang, F., Guo, Z., & Li, L. (2017). microRNA-18a promotes cell migration and invasion through inhibiting Dicer l expression in hepatocellular carcinoma in vitro. Chinese medical sciences journal = Chung-kuo i hsueh k’o hsueh tsa chih, 32(1), 34–3. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/28399983
Tu, K., Zheng, X., Dou, C., Li, C., Yang, W., Yao, Y., & Liu, Q. (2014). MicroRNA-130b promotes cell aggressiveness by inhibiting peroxisome proliferator-activated receptor gamma in human hepatocellular carcinoma. International journal of molecular sciences, 15(11), 20486–99. https://doi.org/10.3390/ijms151120486
Chuang, K.-H., Whitney-Miller, C. L., Chu, C.-Y., Zhou, Z., Dokus, M. K., Schmit, S., & Barry, C. T. (2015). MicroRNA-494 is a master epigenetic regulator of multiple invasion-suppressor microRNAs by targeting ten eleven translocation 1 in invasive human hepatocellular carcinoma tumors. Hepatology (Baltimore, Md.), 62(2), 466–80. https://doi.org/10.1002/hep.27816
Li, Z.-B., Li, Z.-Z., Li, L., Chu, H.-T., & Jia, M. (2015). MiR-21 and miR-183 can simultaneously target SOCS6 and modulate growth and invasion of hepatocellular carcinoma (HCC) cells. European review for medical and pharmacological sciences, 19(17), 3208–17. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26400524
Mao, B., Xiao, H., Zhang, Z., Wang, D., & Wang, G. (2015). MicroRNA-21 regulates the expression of BTG2 in HepG2 liver cancer cells. Molecular medicine reports, 12(4), 4917–24. https://doi.org/10.3892/mmr.2015.4051
Chai, Z.-T., Kong, J., Zhu, X.-D., Zhang, Y.-Y., Lu, L., Zhou, J.-M., … Sun, H.-C. (2013). MicroRNA-26a inhibits angiogenesis by down-regulating VEGFA through the PIK3C2α/Akt/HIF-1α pathway in hepatocellular carcinoma. PloS one, 8(10), e77957. https://doi.org/10.1371/journal.pone.0077957
Wang, G., Sun, Y., He, Y., Ji, C., Hu, B., & Sun, Y. (2015). miR-26a promoted by interferon-alpha inhibits hepatocellular carcinoma proliferation and migration by blocking EZH2. Genetic testing and molecular biomarkers, 19(1), 30–6. https://doi.org/10.1089/gtmb.2014.0245
Ma, D.-N., Chai, Z.-T., Zhu, X.-D., Zhang, N., Zhan, D.-H., Ye, B.-G., … Tang, Z.-Y. (2016). MicroRNA-26a suppresses epithelial-mesenchymal transition in human hepatocellular carcinoma by repressing enhancer of zeste homolog 2, 9(1), 1. https://doi.org/10.1186/s13045-015-0229-y
Wang, Y., Cui, M., Sun, B., Liu, F., Zhang, X., & Ye, L. (2014). MiR-506 suppresses proliferation of hepatoma cells through targeting YAP mRNA 3’UTR. Acta pharmacologica Sinica, 35(9), 1207–14. https://doi.org/10.1038/aps.2014.59
Lu, Z., Zhang, W., Gao, S., Jiang, Q., Xiao, Z., Ye, L., & Zhang, X. (2015). MiR-506 suppresses liver cancer angiogenesis through targeting sphingosine kinase 1 (SPHK1) mRNA. Biochemical and biophysical research communications, 468(1–2), 8–13. https://doi.org/10.1016/j.bbrc.2015.11.008
Shen, G., Lin, Y., Yang, X., Zhang, J., Xu, Z., & Jia, H. (2014). MicroRNA-26b inhibits epithelial-mesenchymal transition in hepatocellular carcinoma by targeting USP9X. BMC cancer, 14(1), 393. https://doi.org/10.1186/1471-2407-14-393
Wang, J., Li, J., Wang, X., Zheng, C., & Ma, W. (2013). Downregulation of microRNA-214 and overexpression of FGFR-1 contribute to hepatocellular carcinoma metastasis. Biochemical and biophysical research communications, 439(1), 47–53. https://doi.org/10.1016/j.bbrc.2013.08.032
Li, Y., Li, Y., Chen, Y., Xie, Q., Dong, N., Gao, Y., … Wang, S. (2017). MicroRNA-214-3p inhibits proliferation and cell cycle progression by targeting MELK in hepatocellular carcinoma and correlates cancer prognosis. Cancer cell international, 17(1), 102. https://doi.org/10.1186/s12935-017-0471-1
Li, H., Wang, H., & Ren, Z. (2018). MicroRNA-214-5p inhibits the invasion and migration of hepatocellular carcinoma cells by targeting Wiskott-Aldrich syndrome like. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology, 46(2), 757–764. https://doi.org/10.1159/000488734
Cui, W., Li, Y., Xu, K., Chen, G., Lu, X., Duan, Q., & Kang, Z. (2016). miR-361-5p inhibits hepatocellular carcinoma cell proliferation and invasion by targeting VEGFA. Biochemical and biophysical research communications, 479(4), 901–906. https://doi.org/10.1016/j.bbrc.2016.09.076
Guo, C., Zhao, D., Zhang, Q., Liu, S., & Sun, M.-Z. (2018). miR-429 suppresses tumor migration and invasion by targeting CRKL in hepatocellular carcinoma via inhibiting Raf/MEK/ERK pathway and epithelial-mesenchymal transition. Scientific reports, 8(1), 2375. https://doi.org/10.1038/s41598-018-20258-8
Yang, Z., Tsuchiya, H., Zhang, Y., Hartnett, M. E., & Wang, L. (2013). MicroRNA-433 inhibits liver cancer cell migration by repressing the protein expression and function of cAMP response element-binding protein. The Journal of biological chemistry, 288(40), 28893–9. https://doi.org/10.1074/jbc.M113.502682
Xue, J., Chen, L.-Z., Li, Z.-Z., Hu, Y., Yan, S., & Liu, L.-Y. (2015). MicroRNA-433 inhibits cell proliferation in hepatocellular carcinoma by targeting p21 activated kinase (PAK4). Molecular and cellular biochemistry, 399(1–2), 77–86. https://doi.org/10.1007/s11010-014-2234-9
Mao, J., Hu, X., Pang, P., Zhou, B., Li, D., & Shan, H. (2017). miR-30e acts as a tumor suppressor in hepatocellular carcinoma partly via JAK1/STAT3 pathway. Oncology reports, 38(1), 393–401. https://doi.org/10.3892/or.2017.5683
Zhao, G., Han, C., Zhang, Z., Wang, L., & Xu, J. (2017). Increased expression of microRNA-31-5p inhibits cell proliferation, migration, and invasion via regulating Sp1 transcription factor in HepG2 hepatocellular carcinoma cell line. Biochemical and biophysical research communications, 490(2), 371–377. https://doi.org/10.1016/j.bbrc.2017.06.050
Han, S.-Y., Han, H.-B., Tian, X.-Y., Sun, H., Xue, D., Zhao, C., … Li, P.-P. (2016). MicroRNA-33a-3p suppresses cell migration and invasion by directly targeting PBX3 in human hepatocellular carcinoma. Oncotarget, 7(27), 42461–42473. https://doi.org/10.18632/oncotarget.9886
Tian, Q., Xiao, Y., Wu, Y., Liu, Y., Song, Z., Gao, W., … Sun, Z. (2016). MicroRNA-33b suppresses the proliferation and metastasis of hepatocellular carcinoma cells through the inhibition of Sal-like protein 4 expression. International journal of molecular medicine, 38(5), 1587–1595. https://doi.org/10.3892/ijmm.2016.2754
Song, J., Wang, Q., Luo, Y., Yuan, P., Tang, C., Hui, Y., & Wang, Z. (2015). miR-34c-3p inhibits cell proliferation, migration and invasion of hepatocellular carcinoma by targeting MARCKS. International journal of clinical and experimental pathology, 8(10), 12728–37. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26722462
Wang, L., Wu, J., & Xie, C. (2017). miR-92a promotes hepatocellular carcinoma cells proliferation and invasion by FOXA2 targeting. Iranian journal of basic medical sciences, 20(7), 783–790. https://doi.org/10.22038/IJBMS.2017.9010
Liu, Z., Wang, J., Mao, Y., Zou, B., & Fan, X. (2016). MicroRNA-101 suppresses migration and invasion via targeting vascular endothelial growth factor-C in hepatocellular carcinoma cells. Oncology letters, 11(1), 433–438. https://doi.org/10.3892/ol.2015.3832
Cao, K., Li, J., Zhao, Y., Wang, Q., Zeng, Q., He, S., … Cao, P. (2016). miR-101 inhibiting cell proliferation, migration and invasion in hepatocellular carcinoma through downregulating girdin, 39(2), 96–102. https://doi.org/10.14348/molcells.2016.2161
Jia, H., Wang, H., Yao, Y., Wang, C., & Li, P. (2018). MiR-136 inhibits malignant progression of hepatocellular carcinoma cells by targeting cyclooxygenase 2. Oncology research, 26(6), 967–976. https://doi.org/10.3727/096504018X15148192843443
Yang, L., Guo, Y., Liu, X., Wang, T., Tong, X., Lei, K., … Xu, Q. (2018). The tumor suppressive miR-302c-3p inhibits migration and invasion of hepatocellular carcinoma cells by targeting TRAF4. Journal of Cancer, 9(15), 2693–2701. https://doi.org/10.7150/jca.25569
Ding, W., Tan, H., Zhao, C., Li, X., Li, Z., Jiang, C., … Wang, L. (2015). MiR-145 suppresses cell proliferation and motility by inhibiting ROCK1 in hepatocellular carcinoma. Tumor Biology, 37(5), 1–6. https://doi.org/10.1007/s13277-015-4462-3
Zu, Y., Yang, Y., Zhu, J., Bo, X., Hou, S., Zhang, B., … Zheng, J. (2016). MiR-146a suppresses hepatocellular carcinoma by downregulating TRAF6. American journal of cancer research, 6(11), 2502–2513. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/27904767
Liu, W., Xu, C., Wan, H., Liu, C., Wen, C., Lu, H., & Wan, F. (2014). MicroRNA-206 overexpression promotes apoptosis, induces cell cycle arrest and inhibits the migration of human hepatocellular carcinoma HepG2 cells. International journal of molecular medicine, 34(2), 420–8. https://doi.org/10.3892/ijmm.2014.1800
Jiang, G., Cui, Y., Yu, X., Wu, Z., Ding, G., & Cao, L. (2015). miR-211 suppresses hepatocellular carcinoma by downregulating SATB2. Oncotarget, 6(11), 9457–66. https://doi.org/10.18632/oncotarget.3265
Xie, F., Yuan, Y., Xie, L., Ran, P., Xiang, X., Huang, Q., … Zheng, S. (2017). miRNA-320a inhibits tumor proliferation and invasion by targeting c-Myc in human hepatocellular carcinoma. OncoTargets and therapy, 10, 885–894. https://doi.org/10.2147/OTT.S122992
Liu, H., Li, W., Chen, C., Pei, Y., & Long, X. (2015). MiR-335 acts as a potential tumor suppressor miRNA via downregulating ROCK1 expression in hepatocellular carcinoma. Tumor Biology, 36(8), 6313–6319. https://doi.org/10.1007/s13277-015-3317-2
Liu, Y., Zhang, W., Liu, S., Liu, K., Ji, B., & Wang, Y. (2017). miR-365 targets ADAM10 and suppresses the cell growth and metastasis of hepatocellular carcinoma. Oncology reports, 37(3), 1857–1864. https://doi.org/10.3892/or.2017.5423
Huang, X.-P., Hou, J., Shen, X.-Y., Huang, C.-Y., Zhang, X.-H., Xie, Y.-A., & Luo, X.-L. (2015). MicroRNA-486-5p, which is downregulated in hepatocellular carcinoma, suppresses tumor growth by targeting PIK3R1. The FEBS journal, 282(3), 579–94. https://doi.org/10.1111/febs.13167
Wang, Y., Xie, Y., Li, X., Lin, J., Zhang, S., Li, Z., … Gong, R. (2018). MiR-876-5p acts as an inhibitor in hepatocellular carcinoma progression by targeting DNMT3A. Pathology, research and practice, 214(7), 1024–1030. https://doi.org/10.1016/j.prp.2018.04.012
Fang, L., Yang, N., Ma, J., Fu, Y., & Yang, G.-S. (2012). microRNA-1301-mediated inhibition of tumorigenesis. Oncology reports, 27(4), 929–34. https://doi.org/10.3892/or.2011.1589
Chang, W., Zhang, L., Xian, Y., & Yu, Z. (2017). MicroRNA-33a promotes cell proliferation and inhibits apoptosis by targeting PPARα in human hepatocellular carcinoma. Experimental and therapeutic medicine, 13(5), 2507–2514. https://doi.org/10.3892/etm.2017.4236
Li, N., Fu, H., Tie, Y., Hu, Z., Kong, W., Wu, Y., & Zheng, X. (2009). miR-34a inhibits migration and invasion by down-regulation of c-Met expression in human hepatocellular carcinoma cells. Cancer letters, 275(1), 44–53. https://doi.org/10.1016/j.canlet.2008.09.035
Wen, F., Li, B., Huang, C., Wei, Z., Zhou, Y., Liu, J., & Zhang, H. (2015). MiR-34a is involved in the decrease of ATP contents induced by resistin through target on ATP5S in HepG2 cells. Biochemical genetics, 53(11–12), 301–9. https://doi.org/10.1007/s10528-015-9693-x
Ye, J., Yao, Y., Song, Q., Li, S., Hu, Z., Yu, Y., … Wang, Q. K. (2016). Up-regulation of miR-95-3p in hepatocellular carcinoma promotes tumorigenesis by targeting p21 expression. Scientific reports, 6(1), 34034. https://doi.org/10.1038/srep34034
Yu, L., Zhou, L., Cheng, Y., Sun, L., Fan, J., Liang, J., … Zhu, L. (2014). MicroRNA-543 acts as an oncogene by targeting PAQR3 in hepatocellular carcinoma. American journal of cancer research, 4(6), 897–906. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/25520877
Liu, T., Zhang, X., Sha, K., Liu, X., Zhang, L., & Wang, B. (2015). miR-709 up-regulated in hepatocellular carcinoma, promotes proliferation and invasion by targeting GPC5. Cell proliferation, 48(3), 330–7. https://doi.org/10.1111/cpr.12181
Xu, D., He, X., Chang, Y., Xu, C., Jiang, X., Sun, S., & Lin, J. (2013). Inhibition of miR-96 expression reduces cell proliferation and clonogenicity of HepG2 hepatoma cells. Oncology reports, 29(2), 653–61. https://doi.org/10.3892/or.2012.2138
Gao, B., Gao, K., Li, L., Huang, Z., & Lin, L. (2014). miR-184 functions as an oncogenic regulator in hepatocellular carcinoma (HCC). Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 68(2), 143–8. https://doi.org/10.1016/j.biopha.2013.09.005
Zhang, J.-J., Wang, C.-Y., Hua, L., Yao, K.-H., Chen, J.-T., & Hu, J.-H. (2015). miR-107 promotes hepatocellular carcinoma cell proliferation by targeting Axin2. International journal of clinical and experimental pathology, 8(5), 5168–74. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26191213
Wu, N., Liu, X., Xu, X., Fan, X., Liu, M., Li, X., … Tang, H. (2011). MicroRNA-373, a new regulator of protein phosphatase 6, functions as an oncogene in hepatocellular carcinoma. The FEBS journal, 278(12), 2044–54. https://doi.org/10.1111/j.1742-4658.2011.08120.x
Zhang, S., Yang, Z., Cai, X., Zhao, M., Sun, M.-M., Li, J., … Zhang, X. (2017). miR-511 promotes the proliferation of human hepatoma cells by targeting the 3′UTR of B cell translocation gene 1 (BTG1) mRNA, 38(8), 1161–1170. https://doi.org/10.1038/aps.2017.62
Lang, Q., & Ling, C. (2012). MiR-124 suppresses cell proliferation in hepatocellular carcinoma by targeting PIK3CA., 426(2). https://doi.org/10.1016/j.bbrc.2012.08.075
Zhao, L., & Wang, W. (2015). miR-125b suppresses the proliferation of hepatocellular carcinoma cells by targeting Sirtuin7. International journal of clinical and experimental medicine, 8(10), 18469–75. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26770454
Zhou, J., Zhang, Y., Qi, Y., Yu, D., Shao, Q., & Liang, J. (2017). MicroRNA-152 inhibits tumor cell growth by directly targeting RTKN in hepatocellular carcinoma. Oncology reports, 37(2), 1227–1234. https://doi.org/10.3892/or.2016.5290
Li, X.-Y., Feng, X.-Z., Tang, J.-Z., Dong, K., Wang, J.-F., Meng, C.-C., … Sun, Z.-W. (2016). MicroRNA-200b inhibits the proliferation of hepatocellular carcinoma by targeting DNA methyltransferase 3a. Molecular medicine reports, 13(5), 3929–35. https://doi.org/10.3892/mmr.2016.4995
Wei, W., Wanjun, L., Hui, S., Dongyue, C., Xinjun, Y., & Jisheng, Z. (2013). miR-203 inhibits proliferation of HCC cells by targeting survivin. Cell biochemistry and function, 31(1), 82–5. https://doi.org/10.1002/cbf.2863
Zhang, A., Lakshmanan, J., Motameni, A., & Harbrecht, B. G. (2018). MicroRNA-203 suppresses proliferation in liver cancer associated with PIK3CA, p38 MAPK, c-Jun, and GSK3 signaling. Molecular and cellular biochemistry, 441(1–2), 89–98. https://doi.org/10.1007/s11010-017-3176-9
Liu, K., Li, X., Cao, Y., Ge, Y., Wang, J., & Shi, B. (2015). MiR-132 inhibits cell proliferation, invasion and migration of hepatocellular carcinoma by targeting PIK3R3. International journal of oncology, 47(4), 1585–93. https://doi.org/10.3892/ijo.2015.3112
Zhang, W., Liu, K., Liu, S., Ji, B., Wang, Y., & Liu, Y. (2015). MicroRNA-133a functions as a tumor suppressor by targeting IGF-1R in hepatocellular carcinoma. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine, 36(12), 9779–88. https://doi.org/10.1007/s13277-015-3749-8
Chen, X., Bo, L., Zhao, X., & Chen, Q. (2015). MicroRNA-133a inhibits cell proliferation, colony formation ability, migration and invasion by targeting matrix metallopeptidase 9 in hepatocellular carcinoma. Molecular medicine reports, 11(5), 3900–7. https://doi.org/10.3892/mmr.2015.3232
Pang, X., Huang, K., Zhang, Q., Zhang, Y., & Niu, J. (2015). miR-154 targeting ZEB2 in hepatocellular carcinoma functions as a potential tumor suppressor. Oncology reports, 34(6), 3272–9. https://doi.org/10.3892/or.2015.4321
Tian, Z., Jiang, H., Liu, Y., Huang, Y., Xiong, X., Wu, H., & Dai, X. (2016). MicroRNA-133b inhibits hepatocellular carcinoma cell progression by targeting Sirt1. Experimental cell research, 343(2), 135–147. https://doi.org/10.1016/j.yexcr.2016.03.027
Li, H., Xiang, Z., Liu, Y., Xu, B., & Tang, J. (2017). MicroRNA-133b inhibits proliferation, cellular migration, and invasion via targeting LASP1 in hepatocarcinoma cells. Oncology research, 25(8), 1269–1282. https://doi.org/10.3727/096504017X14850151453092
Zhu, M., Li, M., Wang, T., Linghu, E., & Wu, B. (2016). MicroRNA-137 represses FBI-1 to inhibit proliferation and in vitro invasion and migration of hepatocellular carcinoma cells. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine, 37(10), 13995–14008. https://doi.org/10.1007/s13277-016-5230-8
Cui, S., Sun, Y., Liu, Y., Liu, C., Wang, J., Hao, G., & Sun, Q. (2018). [Corrigendum] MicroRNA-137 has a suppressive role in liver cancer via targeting EZH2. Molecular medicine reports, 17(5), 7460. https://doi.org/10.3892/mmr.2018.8785
Liu, X., Gong, J., & Xu, B. (2015). miR-143 down-regulates TLR2 expression in hepatoma cells and inhibits hepatoma cell proliferation and invasion. International journal of clinical and experimental pathology, 8(10), 12738–47. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26722463
Xue, F., Yin, J., Xu, L., & Wang, B. (2017). MicroRNA-143 inhibits tumorigenesis in hepatocellular carcinoma by downregulating GATA6. Experimental and therapeutic medicine, 13(6), 2667–2674. https://doi.org/10.3892/etm.2017.4348
Wang, L., Wang, Y. M., Xu, S., Wang, W. G., Chen, Y., MAO, J. Y., … Le Tian, B. (2015). MicroRNA-215 is upregulated by treatment with Adriamycin and leads to the chemoresistance of hepatocellular carcinoma cells and tissues, 12(4). https://doi.org/10.3892/mmr.2015.4012
Li, Q., Wang, G., Shan, J.-L., Yang, Z.-X., Wang, H.-Z., Feng, J., … Wang, D. (2010). MicroRNA-224 is upregulated in HepG2 cells and involved in cellular migration and invasion. Journal of gastroenterology and hepatology, 25(1), 164–71. https://doi.org/10.1111/j.1440-1746.2009.05971.x
Ma, D., Tao, X., Gao, F., Fan, C., & Wu, D. (2012). miR-224 functions as an onco-miRNA in hepatocellular carcinoma cells by activating AKT signaling. Oncology letters, 4(3), 483–488. https://doi.org/10.3892/ol.2012.742
Xu, H., Zhao, L., Fang, Q., Sun, J., Zhang, S., Zhan, C., … Zhang, Y. (2014). MiR-338-3p inhibits hepatocarcinoma cells and sensitizes these cells to sorafenib by targeting hypoxia-induced factor 1α. PloS one, 9(12), e115565. https://doi.org/10.1371/journal.pone.0115565
Xiao, Y., Tian, Q., He, J., Huang, M., Yang, C., & Gong, L. (2016). MiR-503 inhibits hepatocellular carcinoma cell growth via inhibition of insulin-like growth factor 1 receptor. OncoTargets and therapy, 9, 3535–44. https://doi.org/10.2147/OTT.S106351
Yang, X., Zang, J., Pan, X., Yin, J., Xiang, Q., Yu, J., … Lei, X. (2017). miR-503 inhibits proliferation making human hepatocellular carcinoma cells susceptible to 5-fluorouracil by targeting EIF4E. Oncology reports, 37(1), 563–570. https://doi.org/10.3892/or.2016.5220
Yu, M., Xue, H., Wang, Y., Shen, Q., Jiang, Q., Zhang, X., Tian, Y. (2017). miR-345 inhibits tumor metastasis and EMT by targeting IRF1-mediated mTOR/STAT3/AKT pathway in hepatocellular carcinoma. International journal of oncology, 50(3), 975–983. https://doi.org/10.3892/ijo.2017.3852
Zheng, Q., Sheng, Q., Jiang, C., Shu, J., Chen, J., Nie, Z., Zhang, Y. (2014). MicroRNA-452 promotes tumorigenesis in hepatocellular carcinoma by targeting cyclin-dependent kinase inhibitor 1B. Molecular and cellular biochemistry, 389(1–2), 187–95. https://doi.org/10.1007/s11010-013-1940-z
Tang, H., Zhang, J., Yu, Z., Ye, L., Li, K., Ding, F., Meng, W. (2017). Mir-452-3p: a potential tumor promoter that targets the CPEB3/EGFR axis in human hepatocellular carcinoma. Technology in cancer research & treatment, 16(6), 1136–1149. https://doi.org/10.1177/1533034617735931
Yu, L., Gong, X., Sun, L., Yao, H., Lu, B., & Zhu, L. (2015). miR-454 functions as an oncogene by inhibiting CHD5 in hepatocellular carcinoma. Oncotarget, 6(36), 39225–34. https://doi.org/10.18632/oncotarget.4407
Zhou, Y., Li, Y., Ye, J., Jiang, R., Yan, H., Yang, X., … Zhang, J. (2013). MicroRNA-491 is involved in metastasis of hepatocellular carcinoma by inhibitions of matrix metalloproteinase and epithelial to mesenchymal transition. Liver international: official journal of the International Association for the Study of the Liver, 33(8), 1271–80. https://doi.org/10.1111/liv.12190
Zhao, G., Wang, T., Huang, Q.-K., Pu, M., Sun, W., Zhang, Z.-C., Tao, K.-S. (2016). MicroRNA-548a-5p promotes proliferation and inhibits apoptosis in hepatocellular carcinoma cells by targeting Tg737. World journal of gastroenterology, 22(23), 5364–73. https://doi.org/10.3748/wjg.v22.i23.5364
Li, X., Zhang, W., Zhou, L., Yue, D., & Su, X. (2015). MicroRNA-592 targets DEK oncogene and suppresses cell growth in the hepatocellular carcinoma cell line HepG2. International journal of clinical and experimental pathology, 8(10), 12455–63. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/26722432
Han, G., Zhang, L., Ni, X., Chen, Z., Pan, X., Zhu, Q., Wang, X. (2018). MicroRNA-873 promotes cell proliferation, migration, and invasion by directly targeting TSLC1 in hepatocellular carcinoma. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology, 46(6), 2261–2270. https://doi.org/10.1159/000489594
Lu, S., Wu, J., Gao, Y., Han, G., Ding, W., & Huang, X. (2016). MicroRNA-4262 activates the NF-κB and enhances the proliferation of hepatocellular carcinoma cells. International journal of biological macromolecules, 86, 43–9. https://doi.org/10.1016/j.ijbiomac.2016.01.019
Etzioni, R., Urban, N., Ramsey, S., McIntosh, M., Schwartz, S., Reid, B., Hartwell, L. (2003). The case for early detection. Nature reviews. Cancer, 3(4), 243–52. https://doi.org/10.1038/nrc1041
Murray, M. J., Raby, K. L., Saini, H. K., Bailey, S., Wool, S. V, Tunnacliffe, J. M., Coleman, N. (2015). Solid tumors of childhood display specific serum microRNA profiles. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology, 24(2), 350–60. https://doi.org/10.1158/1055-9965.EPI-14-0669
Larrea, E., Sole, C., Manterola, L., Goicoechea, I., Armesto, M., Arestin, M., Lawrie, C. H. (2016). New concepts in cancer biomarkers: circulating miRNAs in liquid biopsies, 17(5), 627. https://doi.org/10.3390/ijms17050627
Pezuk, J. A., Miller, T. L. A., Bevilacqua, J. L. B., de Barros, A. C. S. D., de Andrade, F. E. M., E Macedo, L. F. de A., Reis, L. F. L. (2017). Measuring plasma levels of three microRNAs can improve the accuracy for identification of malignant breast lesions in women with BI-RADS 4 mammography. Oncotarget, 8(48), 83940–83948. https://doi.org/10.18632/oncotarget.20806
Valadi, H., Ekström, K., Bossios, A., Sjöstrand, M., Lee, J. J., & Lötvall, J. O. (2007). Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nature cell biology, 9(6), 654–9. https://doi.org/10.1038/ncb1596
Mitchell, P. S., Parkin, R. K., Kroh, E. M., Fritz, B. R., Wyman, S. K., Pogosova-Agadjanyan, E. L., Tewari, M. (2008). Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. U.S.A., 105(30), 10513–10518. https://doi.org/10.1073/pnas.0804549105
Chim, S. S. C., Shing, T. K. F., Hung, E. C. W., Leung, T. -y., Lau, T. -k., Chiu, R. W. K., & Dennis Lo, Y. M. (2008). Detection and characterization of placental microRNAs in maternal plasma. Clinical Chemistry, 54(3), 482–490. https://doi.org/10.1373/clinchem.2007.097972
Arroyo, J. D., Chevillet, J. R., Kroh, E. M., Ruf, I. K., Pritchard, C. C., Gibson, D. F., … Tewari, M. (2011). Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proceedings of the National Academy of Sciences of the United States of America, 108(12), 5003–8. https://doi.org/10.1073/pnas.1019055108
Noferesti, S. S., Sohel, M. M. H., Hoelker, M., Salilew-Wondim, D., Tholen, E., Looft, C., … Tesfaye, D. (2015). Controlled ovarian hyperstimulation induced changes in the expression of circulatory miRNA in bovine follicular fluid and blood plasma. Journal of ovarian research, 8(1), 81. https://doi.org/10.1186/s13048-015-0208-5
Cortez, M. A., Bueso-Ramos, C., Ferdin, J., Lopez-Berestein, G., Sood, A. K., & Calin, G. A. (2011). MicroRNAs in body fluids—the mix of hormones and biomarkers. Nature Reviews Clinical Oncology, 8(8), 467–477. https://doi.org/10.1038/nrclinonc.2011.76
Turchinovich, A., Weiz, L., & Burwinkel, B. (2012). Extracellular miRNAs: the mystery of their origin and function, 37(11). https://doi.org/10.1016/j.tibs.2012.08.003
Zhang, L., Xu, Y., Jin, X., Wang, Z., Wu, Y., Zhao, D., Liang, Z. (2015). A circulating miRNA signature as a diagnostic biomarker for non-invasive early detection of breast cancer. Breast Cancer Research and Treatment, 154(2), 423–434. https://doi.org/10.1007/s10549-015-3591-0
Zhang, H., Mao, F., Shen, T., Luo, Q., Ding, Z., Qian, L., & Huang, J. (2017). Plasma miR-145, miR-20a, miR-21 and miR-223 as novel biomarkers for screening early-stage non-small cell lung cancer. Oncology letters, 13(2), 669–676. https://doi.org/10.3892/ol.2016.5462
Wang, J., Yan, F., Zhao, Q., Zhan, F., Wang, R., Wang, L., Huang, X. (2017). Circulating exosomal miR-125a-3p as a novel biomarker for early-stage colon cancer. Scientific reports, 7(1), 4150. https://doi.org/10.1038/s41598-017-04386-1
Arab, A., Karimipoor, M., Irani, S., Kiani, A., Zeinali, S., Tafsiri, E., & Sheikhy, K. (2017). Potential circulating miRNA signature for early detection of NSCLC. Cancer genetics, 216–217, 150–158. https://doi.org/10.1016/j.cancergen.2017.07.006
Wang, Y., Yin, W., Lin, Y., Yin, K., Zhou, L., Du, Y., Lu, J. (2018). Downregulated circulating microRNAs after surgery: potential noninvasive biomarkers for diagnosis and prognosis of early breast cancer. Cell death discovery, 5(1), 21. https://doi.org/10.1038/s41420-018-0089-7
Bertoli, G., Cava, C., & Castiglioni, I. (2015). Micrornas: new biomarkers for diagnosis, prognosis, therapy prediction and therapeutic tools for breast cancer. Theranostics, 5(10), 1122–1143. https://doi.org/10.7150/thno.11543
Alečković, M., & Kang, Y. (2015). Regulation of cancer metastasis by cell-free miRNAs. Biochimica et biophysica acta, 1855(1), 24–42. https://doi.org/10.1016/j.bbcan.2014.10.005
Wang, H., Peng, R., Wang, J., Qin, Z., & Xue, L. (2018). Circulating microRNAs as potential cancer biomarkers: the advantage and disadvantage. Clinical epigenetics, 10(1), 59. https://doi.org/10.1186/s13148-018-0492-1
Witwer, K. W. (2015). Circulating microRNA biomarker studies: pitfalls and potential solutions. Clinical chemistry, 61(1), 56–63. https://doi.org/10.1373/clinchem.2014.221341
Leong, S. P., Ballesteros-Merino, C., Jensen, S. M., Marwitz, S., Bifulco, C., Fox, B. A., & Skoberne, M. (2018). Novel frontiers in detecting cancer metastasis. Clin Exp Metastasis, 35(5–6), 403–412. https://doi.org/10.1007/s10585-018-9918-6
Chaudhry, M., Steiner, R., Claussen, C., Patel, K., Lee, H., Weber, D., Manasanch, E. E. (2018). Carfilzomib-based combination regimens are highly effective frontline therapies for multiple myeloma and Waldenström’s macroglobulinemia. Leukemia & lymphoma, 1–7. https://doi.org/10.1080/10428194.2018.1508668
Haug, B. H., Hald, Ø. H., Utnes, P., Roth, S. A., Løkke, C., Flægstad, T., & Einvik, C. (2015). Exosome-like extracellular vesicles from MYCN-amplified neuroblastoma cells contain oncogenic miRNAs. Anticancer Research, 35(5), 2521–2530.
Ramraj, S. K., Aravindan, S., Somasundaram, D. B., Herman, T. S., Natarajan, M., & Aravindan, N. (2016). Serum-circulating miRNAs predict neuroblastoma progression in mouse model of high-risk metastatic disease. Oncotarget, 7(14), 18605–19. https://doi.org/10.18632/oncotarget.7615
Zeka, F., Decock, A., Van Goethem, A., Vanderheyden, K., Demuynck, F., Lammens, T., … Vandesompele, J. (2018). Circulating microRNA biomarkers for metastatic disease in neuroblastoma patients. JCI insight, 3(23). https://doi.org/10.1172/jci.insight.97021
Ma, J., Xu, M., Yin, M., Hong, J., Chen, H., Gao, Y., Mo, X. (2019). Exosomal hsa-miR199a-3p promotes proliferation and migration in neuroblastoma. Frontiers in oncology, 9, 459. https://doi.org/10.3389/fonc.2019.00459
Ludwig, N., Nourkami-Tutdibi, N., Backes, C., Lenhof, H.-P., Graf, N., Keller, A., & Meese, E. (2015). Circulating serum miRNAs as potential biomarkers for nephroblastoma. Pediatric blood & cancer, 62(8), 1360–7. https://doi.org/10.1002/pbc.25481
Liu, W., Chen, S., & Liu, B. (2016). Diagnostic and prognostic values of serum exosomal microRNA-21 in children with hepatoblastoma: a Chinese population-based study. Pediatric surgery international, 32(11), 1059–1065. https://doi.org/10.1007/s00383-016-3960-8
Patel, D., Boufraqech, M., Jain, M., Zhang, L., He, M., Gesuwan, K., Kebebew, E. (2013). MiR-34a and miR-483-5p are candidate serum biomarkers for adrenocortical tumors. Surgery, 154(6). https://doi.org/10.1016/j.surg.2013.06.022
Decmann, A., Bancos, I., Khanna, A., Thomas, M. A., Turai, P., Perge, P., Igaz, P. (2019). Comparison of plasma and urinary microRNA-483-5p for the diagnosis of adrenocortical malignancy. Journal of Biotechnology, 297, 49–53. https://doi.org/10.1016/j.jbiotec.2019.03.017
Pezzuto, F., Buonaguro, L., Buonaguro, F. M., & Tornesello, M. L. (2018). The role of circulating free DNA and microRNA in non-invasive diagnosis of HBV- and HCV-related hepatocellular carcinoma. International Journal of Molecular Sciences, 19(4), 1007. https://doi.org/10.3390/ijms19041007
Mourad, L., El-Ahwany, E., Zoheiry, M., Abu-Taleb, H., Hassan, M., Ouf, A., Zada, S. (2018). Expression analysis of liver-specific circulating microRNAs in HCV-induced hepatocellular carcinoma in Egyptian patients. Cancer biology & therapy, 19(5), 400–406. https://doi.org/10.1080/15384047.2018.1423922
Ali, H. E. A., Abdel Hameed, R., Effat, H., Ahmed, E. K., Atef, A. A., Sharawi, S. K., … Abdel Wahab, A. H. (2017). Circulating microRNAs panel as a diagnostic tool for discrimination of HCV-associated hepatocellular carcinoma. Clinics and research in hepatology and gastroenterology, 41(4), e51–e62. https://doi.org/10.1016/j.clinre.2017.06.004
Huang, Y.-H., Liang, K.-H., Chien, R.-N., Hu, T.-H., Lin, K.-H., Hsu, C.-W., Yeh, C.-T. (2017). A circulating microRNA signature capable of assessing the risk of hepatocellular carcinoma in cirrhotic patients. Scientific reports, 7(1), 523. https://doi.org/10.1038/s41598-017-00631-9
Nishida, N., Arizumi, T., Hagiwara, S., Ida, H., Sakurai, T., & Kudo, M. (2017). MicroRNAs for the prediction of early response to sorafenib treatment in human hepatocellular carcinoma. Liver cancer, 6(2), 113–125. https://doi.org/10.1159/000449475
Shi, M., Jiang, Y., Yang, L., Yan, S., Wang, Y.-G., & Lu, X.-J. (2018). Decreased levels of serum exosomal miR-638 predict poor prognosis in hepatocellular carcinoma. Journal of cellular biochemistry, 119(6), 4711–4716. https://doi.org/10.1002/jcb.26650
Ding, Y., Yan, J.-L., Fang, A.-N., Zhou, W.-F., & Huang, L. (2017). Circulating miRNAs as novel diagnostic biomarkers in hepatocellular carcinoma detection: a meta-analysis based on 24 articles. Oncotarget, 8(39), 66402–66413. https://doi.org/10.18632/oncotarget.18949
Zhang, Y.-C., Xu, Z., Zhang, T.-F., & Wang, Y.-L. (2015). Circulating microRNAs as diagnostic and prognostic tools for hepatocellular carcinoma. World journal of gastroenterology, 21(34), 9853–62. https://doi.org/10.3748/wjg.v21.i34.9853
Moshiri, F., Salvi, A., Gramantieri, L., Sangiovanni, A., Guerriero, P., De Petro, G., … Negrini, M. (2018). Circulating miR-106b-3p, miR-101-3p and miR-1246 as diagnostic biomarkers of hepatocellular carcinoma. Oncotarget, 9(20), 15350–15364. https://doi.org/10.18632/oncotarget.24601
Yin, D., Wang, Y., Sai, W., Zhang, L., Miao, Y., Cao, L., … Yang, L. (2016). Serum microRNA panel for early diagnosis of the onset of hepatocellular carcinoma, 36(4). https://doi.org/10.3892/or.2016.5026
Simonson, B., & Das, S. (2015). MicroRNA therapeutics: the next magic bullet? Mini reviews in medicinal chemistry, 15(6), 467–74. Retrieved from http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC4410078
Bravo, V., Rosero, S., Ricordi, C., & Pastori, R. L. (2007). Instability of miRNA and cDNAs derivatives in RNA preparations. Biochemical and biophysical research communications, 353(4), 1052–5. https://doi.org/10.1016/j.bbrc.2006.12.135
Nguyen, T., Menocal, E. M., Harborth, J., & Fruehauf, J. H. (2008). RNAi therapeutics: an update on delivery. Current opinion in molecular therapeutics, 10(2), 158–67. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/18386228
Janssen, H. L. A., Reesink, H. W., Lawitz, E. J., Zeuzem, S., Rodriguez-Torres, M., Patel, K., … Hodges, M. R. (2013). Treatment of HCV infection by targeting microRNA. The New England journal of medicine, 368(18), 1685–94. https://doi.org/10.1056/NEJMoa1209026
Tivnan, A., Orr, W. S., Gubala, V., Nooney, R., Williams, D. E., McDonagh, C., … Stallings, R. L. (2012). Inhibition of neuroblastoma tumor growth by targeted delivery of microRNA-34a using anti-disialoganglioside GD2 coated nanoparticles. PloS one, 7(5), e38129. https://doi.org/10.1371/journal.pone.0038129
Hsu, S., Yu, B., Wang, X., Lu, Y., Schmidt, C. R., Lee, R. J., … Ghoshal, K. (2013). Cationic lipid nanoparticles for therapeutic delivery of siRNA and miRNA to murine liver tumor. Nanomedicine: nanotechnology, biology, and medicine, 9(8), 1169–80. https://doi.org/10.1016/j.nano.2013.05.007
Li, F., Wang, F., Zhu, C., Wei, Q., Zhang, T., & Zhou, Y. L. (2018). miR-221 suppression through nanoparticle-based miRNA delivery system for hepatocellular carcinoma therapy and its diagnosis as a potential biomarker. International journal of nanomedicine, 13, 2295–2307. https://doi.org/10.2147/IJN.S157805
Meissner, J. M., Toporkiewicz, M., Czogalla, A., Matusewicz, L., Kuliczkowski, K., & Sikorski, A. F. (2015). Novel antisense therapeutics delivery systems: in vitro and in vivo studies of liposomes targeted with anti-CD20 antibody. Journal of controlled release: official journal of the Controlled Release Society, 220(Pt A), 515–528. https://doi.org/10.1016/j.jconrel.2015.11.015
Ekin, A., Karatas, O. F., Culha, M., & Ozen, M. (2014). Designing a gold nanoparticle-based nanocarrier for microRNA transfection into the prostate and breast cancer cells. The journal of gene medicine, 16(11–12), 331–5. https://doi.org/10.1002/jgm.2810
Piacenti, V., Langella, E., Autiero, I., Nolan, J. C., Piskareva, O., Adamo, M. F. A., … Moccia, M. (2019). A combined experimental and computational study on peptide nucleic acid (PNA) analogues of tumor suppressive miRNA-34a. Bioorganic Chemistry, 91, 103165. https://doi.org/10.1016/j.bioorg.2019.103165
Gebert, L. F. R., Rebhan, M. A. E., Crivelli, S. E. M., Denzler, R., Stoffel, M., & Hall, J. (2014). Miravirsen (SPC3649) can inhibit the biogenesis of miR-122. Nucleic acids research, 42(1), 609–21. https://doi.org/10.1093/nar/gkt852
van der Ree, M. H., van der Meer, A. J., van Nuenen, A. C., de Bruijne, J., Ottosen, S., Janssen, H. L., … Reesink, H. W. (2016). Miravirsen dosing in chronic hepatitis C patients results in decreased microRNA-122 levels without affecting other microRNAs in plasma. Alimentary Pharmacology & Therapeutics, 43(1), 102–113. https://doi.org/10.1111/apt.13432
Ottosen, S., Parsley, T. B., Yang, L., Zeh, K., van Doorn, L.-J., van der Veer, E., … Patick, A. K. (2015). In vitro antiviral activity and preclinical and clinical resistance profile of miravirsen, a novel anti-hepatitis C virus therapeutic targeting the human factor miR-122. Antimicrobial agents and chemotherapy, 59(1), 599–608. https://doi.org/10.1128/AAC.04220-14
Cai, C.-K., Zhao, G.-Y., Tian, L.-Y., Liu, L., Yan, K., Ma, Y.-L., … Ma, B.-A. (2012). miR-15a and miR-16-1 downregulate CCND1 and induce apoptosis and cell cycle arrest in osteosarcoma. Oncology reports, 28(5), 1764–70. https://doi.org/10.3892/or.2012.1995
van der Ree, M. H., de Vree, J. M., Stelma, F., Willemse, S., van der Valk, M., Rietdijk, S., … Reesink, H. W. (2017). Safety, tolerability, and antiviral effect of RG-101 in patients with chronic hepatitis C: a phase 1B, double-blind, randomised controlled trial. Lancet (London, England), 389(10070), 709–717. https://doi.org/10.1016/S0140-6736(16)31715-9
Northcott, P. A., Jones, D. T. W., Kool, M., Robinson, G. W., Gilbertson, R. J., Cho, Y.-J., … Pfister, S. M. (2012). Medulloblastomics: the end of the beginning. Nature reviews. Cancer, 12(12), 818–34. https://doi.org/10.1038/nrc3410
Schmidt, M. F. (2017). miRNA targeting drugs: the next blockbusters? Methods in molecular biology (Clifton, N.J.), 1517, 3–22. https://doi.org/10.1007/978-1-4939-6563-2_1
Funding
The authors would like to thank FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) for the financial support along all these years.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salomão, K.B., Pezuk, J.A., de Souza, G.R. et al. MicroRNA dysregulation interplay with childhood abdominal tumors. Cancer Metastasis Rev 38, 783–811 (2019). https://doi.org/10.1007/s10555-019-09829-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-019-09829-x