Abstract
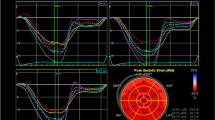
Systemic lupus erythematosus (SLE) often leads to various cardiovascular diseases. We aimed to investigate the value of peak strain dispersion (PSD) in evaluating left ventricular dysfunction in patients with uncomplicated SLE. Eighty-seven female SLE patients and fifty-nine healthy female controls were recruited. The SLE patients were divided into inactive disease (SLE disease activity index (SLEDAI) ≤ 4; n = 48) and active disease (SLEDAI ≥ 5; n = 39) subgroups. Traditional echocardiography and two-dimensional speckle-tracking echocardiography were performed using a GE VividE9 ultrasound diagnostic system and an advanced quantitative analysis EchoPAC workstation (version 201), respectively. The global longitudinal strain (GLS) in the SLE with SLEDAI ≤ 4 group was comparable to that in the control group (− 19.89% vs − 20.7%; P = 0.061). However, GLS was obviously damaged in the SLE with SLEDAI ≥ 5 group compared with that in the control group (− 19.07% vs − 20.7%; P < 0.001). PSD impairment was observed in the SLE with SLEDAI ≤ 4 group (33.83 ms vs 31.44 ms; P = 0.012) and SLE with SLEDAI ≥ 5 groups (52.31 ms vs 31.44 ms; P < 0.001), but the largest difference was observed in the active disease group. Linear regression analysis showed that PSD was moderately correlated with the SLEDAI (r = 0.535; P < 0.001) in SLE patients with SLEDAI ≤ 4 and showed the best correlation with the SLEDAI (r = 0.646; P < 0.001) in the SLE patients with SLEDAI ≥ 5. A correlation between GLS and the SLEDAI (r = 0.359; P = 0.025) was found in the active disease group but not in the inactive disease group (r = 0.253; P = 0.082). PSD is more comprehensive and accurate for evaluating left ventricular subclinical dysfunction in SLE patients. In inactive SLE patients, PSD is a more sensitive index to evaluate early systolic dysfunction of the left ventricle. GLS may be a more vulnerable indicator of early left ventricular cardiac dysfunction in active SLE patients. Controlling disease activity may reduce the events of cardiac dysfunction.





Similar content being viewed by others
References
Mok CC (2011) Epidemiology and survival of systemic lupus erythematosus in Hong Kong Chinese. Lupus 20(7):767–771
Doria A et al (2005) Cardiac involvement in systemic lupus erythematosus. Lupus 14(9):683–686
Leung WH et al (1990) Association between antiphospholipid antibodies and cardiac abnormalities in patients with systemic lupus erythematosus. Am J Med 89(4):411–419
Vila LM et al (1999) Lymphocyte populations and cytokine concentrations in pericardial fluid from a systemic lupus erythematosus patient with cardiac tamponade. Ann Rheum Dis 58(11):720–721
Bienias P et al (2019) Comparison of non-invasive assessment of arrhythmias, conduction disturbances and cardiac autonomic tone in systemic sclerosis and systemic lupus erythematosus. Rheumatol Int 39(2):301–310
Bartels CM et al (2014) Mortality and cardiovascular burden of systemic lupus erythematosus in a US population-based cohort. J Rheumatol 41(4):680–687
Bengtsson C et al (2012) Cardiovascular event in systemic lupus erythematosus in northern Sweden: incidence and predictors in a 7-year follow-up study. Lupus 21(4):452–459
Keeling SO et al (2018) Measuring disease activity and damage with validated metrics: a systematic review on mortality and damage in systemic lupus erythematosus. J Rheumatol 45(10):1448–1461
Hesselvig JH et al (2017) Cutaneous lupus erythematosus and systemic lupus erythematosus are associated with clinically significant cardiovascular risk: a Danish nationwide cohort study. Lupus 26(1):48–53
Haque S, Bruce IN (2005) Therapy insight: systemic lupus erythematosus as a risk factor for cardiovascular disease. Nat Clin Pract Cardiovasc Med 2(8):423–430
Leitman M et al (2004) Two-dimensional strain-a novel software for real-time quantitative echocardiographic assessment of myocardial function. J Am Soc Echocardiogr 17(10):1021–1029
Carasso S et al (2013) Left ventricular function and functional recovery early and late after myocardial infarction: a prospective pilot study comparing two-dimensional strain, conventional echocardiography, and radionuclide myocardial perfusion imaging. J Am Soc Echocardiogr 26(11):1235–1244
Blessberger H, Binder T (2010) NON-invasive imaging: two dimensional speckle tracking echocardiography: basic principles. Heart 96(9):716–722
Amundsen BH et al (2006) Noninvasive myocardial strain measurement by speckle tracking echocardiography: validation against sonomicrometry and tagged magnetic resonance imaging. J Am Coll Cardiol 47(4):789–793
Skaarup KG et al (2018) Association between layer-specific global longitudinal strain and adverse outcomes following acute coronary syndrome. Eur Heart J Cardiovasc Imaging 19(12):1334–1342
Kim D et al (2017) Differences in left ventricular functional adaptation to arterial stiffness and neurohormonal activation in patients with hypertension: a study with two-dimensional layer-specific speckle tracking echocardiography. Clin Hypertens 23:21
Perdreau E et al (2016) Postoperative assessment of left ventricular function by two-dimensional strain (speckle tracking) after paediatric cardiac surgery. Arch Cardiovasc Dis 109(11):599–606
Cusma Piccione M et al (2013) Role of 2D strain in the early identification of left ventricular dysfunction and in the risk stratification of systemic sclerosis patients. Cardiovasc Ultrasound 11:6
Hassanin N, Alkemary A (2016) Early detection of subclinical uremic cardiomyopathy using two-dimensional speckle tracking echocardiography. Echocardiography 33(4):527–536
Wierzbowska-Drabik K, Plewka M, Kasprzak JD (2017) Variability of longitudinal strain in left ventricular segments supplied by non-stenosed coronary artery: insights from speckle tracking analysis of dobutamine stress echocardiograms in patients with high coronary risk profile. AMS 13(1):82–92
Aagaard EN et al (2020) Left ventricular mechanical dispersion in a general population: data from the Akershus Cardiac Examination 1950 study. Eur Heart J Cardiovasc Imaging 21(2):183–190
Kosiuk J et al (2015) Association between ventricular arrhythmias and myocardial mechanical dispersion assessed by strain analysis in patients with nonischemic cardiomyopathy. Clin Res Cardiol 104(12):1072–1077
Kvisvik B et al (2019) Mechanical dispersion as a marker of left ventricular dysfunction and prognosis in stable coronary artery disease. Int J Cardiovasc Imaging 35(7):1265–1275
Ermakov S et al (2019) Left ventricular mechanical dispersion predicts arrhythmic risk in mitral valve prolapse. Heart 105(14):1063–1069
Klaeboe LG et al (2017) Prognostic value of left ventricular deformation parameters in patients with severe aortic stenosis: a pilot study of the usefulness of strain echocardiography. J Am Soc Echocardiogr 30(8):727-735.e1
Prihadi EA et al (2019) Determinants and prognostic implications of left ventricular mechanical dispersion in aortic stenosis. Eur Heart J Cardiovasc Imaging 20(7):740–748
Zhu M et al (2019) Left ventricular global longitudinal strain and mechanical dispersion predict response to multipoint pacing for cardiac resynchronization therapy. J Clin Ultrasound 47(6):356–365
Hensen LCR et al (2018) Left ventricular mechanical dispersion and global longitudinal strain and ventricular arrhythmias in predialysis and dialysis patients. J Am Soc Echocardiogr 31(7):777–783
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40(9):1725
Gladman DD, Ibanez D, Urowitz MB (2002) Systemic lupus erythematosus disease activity index 2000. J Rheumatol 29(2):288–291
Bernatsky S et al (2007) A population-based assessment of systemic lupus erythematosus incidence and prevalence—results and implications of using administrative data for epidemiological studies. Rheumatology (Oxford) 46(12):1814–1818
Knight JS, Kaplan MJ (2013) Cardiovascular disease in lupus: insights and updates. Curr Opin Rheumatol 25(5):597–605
Ng AC et al (2008) Comparison of myocardial tissue velocities measured by two-dimensional speckle tracking and tissue Doppler imaging. Am J Cardiol 102(6):784–789
Henein MY, Gibson DG (1999) Normal long axis function. Heart 81(2):111–113
Haugaa KH et al (2010) Mechanical dispersion assessed by myocardial strain in patients after myocardial infarction for risk prediction of ventricular arrhythmia. JACC Cardiovasc Imaging 3(3):247–256
Jain D, Halushka MK (2009) Cardiac pathology of systemic lupus erythematosus. J Clin Pathol 62(7):584–592
Kobayashi H et al (2010) Cardiac magnetic resonance imaging abnormalities in patients with systemic lupus erythematosus: a preliminary report. Mod Rheumatol 20(3):319–323
Yap DY, Lai KN (2010) Cytokines and their roles in the pathogenesis of systemic lupus erythematosus: from basics to recent advances. J Biomed Biotechnol 2010:365083
Liu Y, Kaplan MJ (2018) Cardiovascular disease in systemic lupus erythematosus: an update. Curr Opin Rheumatol 30(5):441–448
Roman MJ et al (2003) Prevalence and correlates of accelerated atherosclerosis in systemic lupus erythematosus. N Engl J Med 349(25):2399–2406
Yip GW et al (2009) Disease chronicity and activity predict subclinical left ventricular systolic dysfunction in patients with systemic lupus erythematosus. Heart 95(12):980–987
Tincani A et al (2006) Heart involvement in systemic lupus erythematosus, anti-phospholipid syndrome and neonatal lupus. Rheumatology (Oxford) 45(Suppl 4):iv8–iv13
Funding
This study was funded by the Key Research & Development Projects of Sichuan Province (Grant Number 2020YFS0245).
Author information
Authors and Affiliations
Contributions
CL, WB and LR contributed to the study conception and design. Data collection and analysis were performed by CL, KL and MY. The first draft of the manuscript was written by CL. All the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Consent for publication
All authors provided their consent for publication.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, C., Li, K., Yuan, M. et al. Peak strain dispersion within the left ventricle detected by two-dimensional speckle tracking in patients with uncomplicated systemic lupus erythematosus. Int J Cardiovasc Imaging 37, 2197–2205 (2021). https://doi.org/10.1007/s10554-021-02201-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-021-02201-7