Abstract
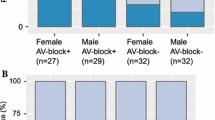
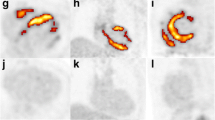
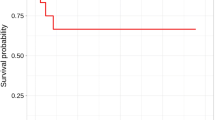
Cardiac sarcoidosis (CS) is known to be associated with ventricular tachycardia (VT); however, most investigations to date have focused on patients with known extra-cardiac sarcoidosis. The presence of CS is typically evaluated using 18F-fluorodeoxyglucose (18F-FDG) uptake on cardiac positron emission tomography (PET) or late gadolinium enhancement (LGE) on cardiac magnetic resonance (CMR). In this study, we sought to determine the prevalence of primary CS and the relationship between myocardial 18F-FDG uptake and LGE in patients with VT without known sarcoidosis. We retrospectively identified 67 patients without known sarcoidosis or active ischemic heart disease (i.e. significant ischemic disease that had not been previously revascularized) referred for both CMR and PET for evaluation of VT. Standard cine- and LGE- CMR and cardiac PET protocols were used. Myocardial LGE was defined as signal intensity > 5 SDs above the mean signal intensity of normal myocardium. Cardiac PET images were considered positive if there was focal myocardial 18F-FDG uptake having greater activity than the left ventricular blood pool. 45 patients (67%) had LGE, while only 4 (6%) had myocardial FDG uptake. Nine percent of patients with LGE had FDG-uptake while none without LGE did, and 10% of the cohort had indeterminate FDG uptake presumably from poor dietary preparation. Of those with both FDG uptake and LGE, 3/4 ultimately received a clinical diagnosis of CS. 4.5% of patients without previously known sarcoidosis or active ischemic heart disease presenting with VT have newly diagnosed CS. Detection of CS can be increased using a CMR first approach followed by cardiac PET for patients with non-ischemic LGE.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CMR:
-
Cardiac magnetic resonance
- CS:
-
Cardiac sarcoidosis
- EF:
-
Ejection fraction
- ICD:
-
Implantable cardioverter-defibrillator
- LGE:
-
Late gadolinium enhancement
- LV:
-
Left ventricle, left ventricular
- NSVT:
-
Non-sustained ventricular tachycardia
- PET:
-
Positron emission tomography
- PVC:
-
Premature ventricular contraction
- SUV:
-
Standardized uptake value
- VT:
-
Ventricular tachycardia
- 18F-FDG:
-
18F-fluorodeoxyglucose
References
Tung R, Bauer B, Schelbert H, Lynch JP 3rd, Auerbach M, Gupta P, Schiepers C, Chan S, Ferris J, Barrio M, Ajijola O, Bradfield J, Shivkumar K (2015) Incidence of abnormal positron emission tomography in patients with unexplained cardiomyopathy and ventricular arrhythmias: the potential role of occult inflammation in arrhythmogenesis. Heart Rhythm 12:2488–2498
Dweck MR, Abgral R, Trivieri MG, Robson PM, Karakatsanis N, Mani V, Palmisano A, Miller MA, Lala A, Chang HL, Sanz J, Contreras J, Narula J, Fuster V, Padilla M, Fayad ZA, Kovacic JC (2018) Hybrid magnetic resonance imaging and positron emission tomography with fluorodeoxyglucose to diagnose active cardiac sarcoidosis. JACC Cardiovasc Imaging 11:94–107
Blankstein R, Osborne M, Naya M, Waller A, Kim CK, Murthy VL, Kazemian P, Kwong RY, Tokuda M, Skali H, Padera R, Hainer J, Stevenson WG, Dorbala S, Di Carli MF (2014) Cardiac positron emission tomography enhances prognostic assessments of patients with suspected cardiac sarcoidosis. J Am Coll Cardiol 63:329–336
Bravo PE, Raghu G, Rosenthal DG, Elman S, Petek BJ, Soine LA, Maki JH, Branch KR, Masri SC, Patton KK, Caldwell JH, Krieger EV (2017) Risk assessment of patients with clinical manifestations of cardiac sarcoidosis with positron emission tomography and magnetic resonance imaging. Int J Cardiol 241:457–462
Osborne MT, Hulten EA, Singh A, Waller AH, Bittencourt MS, Stewart GC, Hainer J, Murthy VL, Skali H, Dorbala S, Di Carli MF, Blankstein R (2014) Reduction in (1)(8)F-fluorodeoxyglucose uptake on serial cardiac positron emission tomography is associated with improved left ventricular ejection fraction in patients with cardiac sarcoidosis. J Nucl Cardiol 21:166–174
Vita T, Okada DR, Veillet-Chowdhury M, Bravo PE, Mullins E, Hulten E, Agrawal M, Madan R, Taqueti VR, Steigner M, Skali H, Kwong RY, Stewart GC, Dorbala S, Di Carli MF, Blankstein R (2018) Complementary value of cardiac magnetic resonance imaging and positron emission tomography/computed tomography in the assessment of cardiac sarcoidosis. Circ Cardiovasc Imaging 11:e007030
Singh A, Kawaji K, Goyal N, Nazir NT, Beaser A, O’Keefe-Baker V, Addetia K, Tung R, Hu P, Mor-Avi V, Patel AR (2019) Feasibility of cardiac magnetic resonance wideband protocol in patients with implantable cardioverter defibrillators and its utility for defining scar. Am J Cardiol 123:1329–1335
Kuruvilla S, Adenaw N, Katwal AB, Lipinski MJ, Kramer CM, Salerno M (2014) Late gadolinium enhancement on cardiac magnetic resonance predicts adverse cardiovascular outcomes in nonischemic cardiomyopathy: a systematic review and meta-analysis. Circ Cardiovasc Imaging 7:250–258
Thomas KW, Hunninghake GW (2003) Sarcoidosis. JAMA 289:3300–3303
Rizzato G, Palmieri G, Agrati AM, Zanussi C (2004) The organ-specific extrapulmonary presentation of sarcoidosis: a frequent occurrence but a challenge to an early diagnosis. A 3-year-long prospective observational study. Sarcoidosis Vasc Diffuse Lung Dis 21:119–126
Okada DR, Bravo PE, Vita T, Agarwal V, Osborne MT, Taqueti VR, Skali H, Chareonthaitawee P, Dorbala S, Stewart G, Di Carli M, Blankstein R (2018) Isolated cardiac sarcoidosis: a focused review of an under-recognized entity. J Nucl Cardiol 25:1136–1146
Crouser ED, Ono C, Tran T, He X, Raman SV (2014) Improved detection of cardiac sarcoidosis using magnetic resonance with myocardial T2 mapping. Am J Respir Crit Care Med 189:109–112
Crouser ED, Ruden E, Julian MW, Raman SV (2016) Resolution of abnormal cardiac MRI T2 signal following immune suppression for cardiac sarcoidosis. J Investig Med 64:1148–1150
Messroghli DR, Moon JC, Ferreira VM, Grosse-Wortmann L, He T, Kellman P, Mascherbauer J, Nezafat R, Salerno M, Schelbert EB, Taylor AJ, Thompson R, Ugander M, van Heeswijk RB, Friedrich MG (2017) Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: a consensus statement by the society for cardiovascular magnetic resonance (SCMR) endorsed by the European association for cardiovascular imaging (EACVI). J Cardiovasc Magn Reson 19:75
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
AP (principal investigator), KK, and AS, designed the study, read MRI’s, and developed the manuscript. SC collected and reviewed clinical data and contributed to the manuscript. RW, DA, and RL read PET studies. EF, JM, and RT helped develop the study concept and performed ablations on several patients. All authors reviewed the manuscript prior to submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
The University of Chicago IRB approved with a waiver of consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kebed, K.Y., Carter, S.V., Flatley, E. et al. Prevalence of newly diagnosed sarcoidosis in patients with ventricular arrhythmias: a cardiac magnetic resonance and 18F-FDG cardiac PET study. Int J Cardiovasc Imaging 37, 1361–1369 (2021). https://doi.org/10.1007/s10554-020-02090-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-02090-2