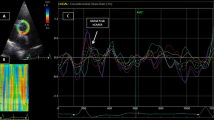
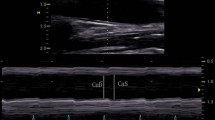
Abstract
Arterial stiffness is a marker of cardiovascular damage and an independent predictor of major cardiovascular events, usually assessed via Carotid-Femoral Pulse Wave Velocity (cfPWV). The aim of this study was to test the feasibility and usefulness of aortic strain analysis using a simplified transthoracic echocardiography (TTE) speckle-tracking (ST) based method. 60 consecutive patients with normal blood pressure and aortic dimensions underwent TTE and cfPWV evaluation. Strain analysis was performed on the largest section of the ascending aorta (aA) with a dedicated software; three couples of speckles were identified within the anterior and posterior aortic wall and transverse segments connecting each couple traced. Peak ascending Aorta Strain (PaAS) was defined as the averaged peak percentage deformation of these segments during the cardiac cycle; aortic stiffness index β2 was defined as 100 × Ln(SBP/DBP)/PaAS. 17 patients were excluded for suboptimal TTE images. In the 43 analyzed individuals (male 58%, age 63 years) PaAS was 5.5 [3.7–8.6] % with excellent inter and intra observer reproducibility (ICC 95% for both). PaAS showed significant correlation with age (r = − 0.64), aA diameter (r = − 0.45), cfPWV (r = − 0.41), stroke volume (r = − 0.41) and heart rate (r = − 0.49). Age remained the only independent determinant of PaAS in a multivariate analysis. β2 (10.8 [5.9–15.0]) showed significant direct correlation with age, Pulse pressure, aA diameter and cfPWV. A simplified strain analysis of the ascending aorta is technically feasible with good reproducibility; PaAS and β2 index correlate well with established parameters of arterial stiffness and might represent a new index in evaluating vascular biomechanics.


Similar content being viewed by others
Abbreviations
- aA:
-
Ascending aorta
- cfPWV:
-
Carotid-femoral pulse wave velocity
- PaAS:
-
Peak ascending aorta strain
- ST:
-
Speckle tracking
- TTE:
-
Transthoracic echocardiography
References
Milan A et al (2011) Arterial stiffness: from physiology to clinical implications. High Blood Press Cardiovasc Prev 18:1–12
Cheng S et al (2010) Correlates of echocardiographic indices of cardiac remodeling over the adult life course: longitudinal observations from the Framingham Heart Study. Circulation 122:570–578
Pluta A et al (2015) Left ventricular remodeling and arterial remodeling in patients with chronic kidney disease stage 1–3. Ren Fail 37:1105–1110
Laurent S et al (2006) Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 27:2588–2605
Bruno RM et al (2017) Carotid and aortic stiffness in essential hypertension and their relation with target organ damage: the CATOD study. J Hypertens 35:310–318
Chirinos JA et al (2012) Arterial stiffness: basic concepts and measurement techniques. J Cardiovasc Transl Res 5:243–255
Ecobici M, Voiculescu M (2017) Importance of arterial stiffness in predicting cardiovascular events. Rom J Intern Med 55:8–13
Mancia G et al (2013) 2013 Practice guidelines for the management of arterial hypertension of the European Society of Hypertension (ESH) and the European Society of Cardiology (ESC): ESH/ESC Task Force for the Management of Arterial Hypertension. J Hypertens 31:1925–1938
Lang RM et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults:an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28:1–39.e14
Townsend RR et al (2017) Arterial stiffness: recommendations and standardization. Pulse (Basel) 4:3–7
Cavalcante JL et al (2011) Aortic stiffness: current understanding and future directions. J Am Coll Cardiol 57:1511–1522
Weber T et al (2009) Noninvasive determination of carotid-femoral pulse wave velocity depends critically on assessment of travel distance: a comparison with invasive measurement. J Hypertens 27:1624–1630
Cho JY et al (2016) Evaluation of arterial stiffness by echocardiography: methodological aspects. Chonnam Med J 52:101–106
Tosello F et al (2016) Out of proportion proximal aortic remodeling: a subclinical marker of early vascular ageing? A systematic review. Int J Cardiol 223:999–1006
Safar ME et al (2010) Arterial aging–hemodynamic changes and therapeutic options. Nat Rev Cardiol 7:442–449
Milan A et al (2013) Ascending aortic dilatation, arterial stiffness and cardiac organ damage in essential hypertension. J Hypertens 31:109–116
Vlachopoulos C et al (2010) Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol 55:1318–1327
Davies JI et al (2003) Pulse wave analysis and pulse wave velocity: a critical review of their strengths and weaknesses. J Hypertens 21:463–472
Morrison TM et al (2009) Circumferential and longitudinal cyclic strain of the human thoracic aorta: age-related changes. J Vasc Surg 49:1029–1036
Redheuil A et al (2010) Reduced ascending aortic strain and distensibility: earliest manifestations of vascular aging in humans. Hypertension 55:319–326
Cuspidi C et al (2015) How to identify hypertensive patients at high cardiovascular risk? The role of echocardiography. High Blood Press Cardiovasc Prev 22:113–117
Vogt S et al (2016) Detailed measurement of wall strain with 3D speckle tracking in the aortic root: a case of bionic support for clinical decision making. Thorac Cardiovasc Surg Rep 5:77–80
Li Y et al (2016) Evaluation of myocardial strain and artery elasticity using speckle tracking echocardiography and high-resolution ultrasound in patients with bicuspid aortic valve. Int J Cardiovasc Imaging 32:1063–1069
Geyer H et al (2010) Assessment of myocardial mechanics using speckle tracking echocardiography: fundamentals and clinical applications. J Am Soc Echocardiogr 23:351–369 (quiz 453–355)
Oishi Y et al (2008) A novel approach to assess aortic stiffness related to changes in aging using a two-dimensional strain imaging. Echocardiography 25:941–945
Kim KH et al (2009) Usefulness of aortic strain analysis by velocity vector imaging as a new echocardiographic measure of arterial stiffness. J Am Soc Echocardiogr 22:1382–1388
Teixeira R et al (2015) Circumferential vascular strain rate to estimate vascular load in aortic stenosis: a speckle tracking echocardiography study. Int J Cardiovasc Imaging 31:681–689
Fritze O et al (2012) Age-related changes in the elastic tissue of the human aorta. J Vasc Res 49:77–86
Honda T, Yano K et al (1994) Evaluation of aortic distensibility in patients with essential hypertension by using cine magnetic resonance imaging. Angiology 45:207–212
Donato Aquaro G et al (2011) Elastic properties of aortic wall in patients with bicuspid aortic valve by magnetic resonance imaging. Am J Cardiol 108:81–87
O’Rourke MF (2007) Arterial aging: pathophysiological principles. Vasc Med 12:329–341
Tomiyama H et al (2018) Longitudinal association among endothelial function, arterial stiffness and subclinical organ damage in hypertension. Int J Cardiol 253:161–166
Safar ME (2018) Arterial stiffness as a risk factor for clinical hypertension. Nat Rev Cardiol 15:97–105
Azadani AN et al (2012) Comparison of mechanical properties of human ascending aorta and aortic sinuses. Ann Thorac Surg 93:87–94
Van Bortel LM et al (2012) Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J Hypertens 30:445–448
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Sabia, L., Avenatti, E., Cesareo, M. et al. Evaluation of aortic stiffness by a new simplified 2D speckle tracking analysis. Int J Cardiovasc Imaging 34, 1753–1760 (2018). https://doi.org/10.1007/s10554-018-1400-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-018-1400-7