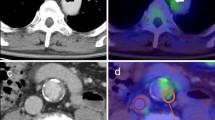
Abstract
Baseline predictors of response to treatment of patients with coronary heart disease (CHD) with respect to vascular inflammation and atherosclerotic plaque burden are poorly understood. From post hoc analysis of the dal-PLAQUE study (NCT00655473), 18F-fluorodeoxyglucose-positron emission tomography (18-FDG-PET) imaging and carotid black blood magnetic resonance imaging (MRI) were used to track changes in these vascular parameters. Baseline demographics, imaging, and biomarkers were collected/measured in 130 patients with CHD or CHD risk-equivalents, and imaging follow-up at 6 months (PET) and 24 months (MRI) was performed. Using stepwise linear regression, predictors of change in carotid plaque inflammation by PET [target-to-background ratio (TBR), n = 92] and plaque burden by MRI [wall area (WA) and total vessel area (TVA), n = 89] were determined. Variables with p < 0.05 in multivariable models were considered independently significant. Interleukin-6, systolic blood pressure and standard deviation of wall thickness (WT) at baseline were independently positively associated with 18-FDG uptake (mean of maximum [MeanMax] TBR change over 6 months). Mean of mean TBR, phospholipase A2, apolipoprotein A-I, and high-sensitivity C-reactive protein at baseline were independently negatively associated with MeanMax TBR change over 6 months. Mean WT and plasminogen activator inhibitor-1 (PAI-1) activity at baseline, and age, were independently associated with change in WA over 24 months. For TVA changes; mean WA and PAI-1 activity at baseline, age, and female gender were independent predictors. These findings may help determine patients most suitable for clinical trials employing plaque inflammation or burden changes as endpoints.




Similar content being viewed by others

References
Lobatto ME, Fuster V, Fayad ZA, Mulder WJ (2011) Perspectives and opportunities for nanomedicine in the management of atherosclerosis. Nat Rev Drug Discov 10:835–852
Underhill HR, Hatsukami TS, Fayad ZA, Fuster V, Yuan C (2010) MRI of carotid atherosclerosis: clinical implications and future directions. Nat Rev Cardiol 7:165–173
Sekikawa A, Curb JD, Edmundowicz D, Okamura T, Choo J, Fujiyoshi A, Masaki K, Miura K, Kuller LH, Shin C, Ueshima H (2012) Coronary artery calcification by computed tomography in epidemiologic research and cardiovascular disease prevention. J Epidemiol 22:188–198
Paraskevas KI, Wierzbicki AS, Mikhailidis DP (2012) Statins and noncardiac vascular disease. Curr Opin Cardiol 27:392–397
Fryburg DA, Vassileva MT (2011) Atherosclerosis drug development in jeopardy: the need for predictive biomarkers of treatment response. Sci Transl Med 3(72):1–5
Fayad ZA, Mani V, Woodward M, Kallend D, Bansilal S, Pozza J, Burgess T, Fuster V, Rudd JH, Tawakol A, Farkouh ME (2011) Rationale and design of dal-PLAQUE: a study assessing efficacy and safety of dalcetrapib on progression or regression of atherosclerosis using magnetic resonance imaging and 18F-fluorodeoxyglucose positron emission tomography/computed tomography. Am Heart J 162(2):214–221
Rudd JH, Warburton EA, Fryer TD, Jones HA, Clark JC, Antoun N, Johnström P, Davenport AP, Kirkpatrick PJ, Arch BN, Pickard JD, Weissberg PL (2002) Imaging atherosclerotic plaque inflammation with [18F]-fluorodeoxyglucose positron emission tomography. Circulation 105:2708–2711
James OG, Christensen JD, Wong TZ, Borges-Neto S, Koweek LM (2011) Utility of FDG PET/CT in inflammatory cardiovascular disease. Radiographics 31:1271–1286
Mizoguchi M, Tahara N, Tahara A, Nitta Y, Kodama N, Oba T, Mawatari K, Yasukawa H, Kaida H, Ishibashi M, Hayabuchi N, Harada H, Ikeda H, Yamagishi S, Imaizumi T (2011) Pioglitazone attenuates atherosclerotic plaque inflammation in patients with impaired glucose tolerance or diabetes a prospective, randomized, comparator-controlled study using serial FDG PET/CT imaging study of carotid artery and ascending aorta. JACC Cardiovasc Imaging 4:1110–1118
Ogawa M, Nakamura S, Saito Y, Kosugi M, Magata Y (2012) What can be seen by 18F-FDG PET in atherosclerosis imaging? The effect of foam cell formation on 18F-FDG uptake to macrophages in vitro. J Nucl Med 53:55–58
Rosenbaum D, Millon A, Fayad ZA (2012) Molecular imaging in atherosclerosis: FDG PET. Curr Atheroscler Rep 14:429–437
Tardif JC, Lesage F, Harel F, Romeo P, Pressacco J (2011) Imaging biomarkers in atherosclerosis trials. Circ Cardiovasc Imaging 4:319–333
Corti R, Fuster V (2011) Imaging of atherosclerosis: magnetic resonance imaging. Eur Heart J 32(14):1709–1719
Fayad ZA, Mani V, Woodward M, Kallend D, Abt M, Burgess T, Fuster V, Ballantyne CM, Stein EA, Tardif JC, Rudd JH, Farkouh ME, Tawakol A, dal-PLAQUE Investigators (2011) Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial. Lancet 378:1547–1559
Fayad ZA, Mani V, Fuster V (2012) The time has come for clinical cardiovascular trials with plaque characterization as an endpoint. Eur Heart J 33:160–161
Rudd JH, Myers KS, Bansilal S, Machac J, Pinto CA, Tong C, Rafique A, Hargeaves R, Farkouh M, Fuster V, Fayad ZA (2008) Atherosclerosis inflammation imaging with 18F-FDG PET: carotid, iliac, and femoral uptake reproducibility, quantification methods, and recommendations. J Nucl Med 49:871–878
El Aidi H, Mani V, Weinshelbaum KB, Aguiar SH, Taniguchi H, Postley JE, Samber DD, Cohen EI, Stern J, van der Geest RJ, Reiber JH, Woodward M, Fuster V, Gidding SS, Fayad ZA (2009) Cross-sectional, prospective study of MRI reproducibility in the assessment of plaque burden of the carotid arteries and aorta. Nat Clin Pract Cardiovasc Med 6:219–228
Investigators AIM-HIGH, Boden WE, Probstfield JL, Anderson T, Chaitman BR, Desvignes-Nickens P, Koprowicz K, McBride R, Teo K, Weintraub W (2011) Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med 365:2255–2267
Keech A, Simes RJ, Barter P, Best J, Scott R, Taskinen MR, Forder P, Pillai A, Davis T, Glasziou P, Drury P, Kesäniemi YA, Sullivan D, Hunt D, Colman P, d’Emden M, Whiting M, Ehnholm C, Laakso M, FIELD study investigators (2005) Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 366:1849–1861
Schwartz GG, Olsson AG, Abt M, Ballantyne CM, Barter PJ, Brumm J, Chaitman BR, Holme IM, Kallend D, Leiter LA, Leitersdorf E, McMurray JJ, Mundl H, Nicholls SJ, Shah PK, Tardif JC, Wright RS, dal-OUTCOMES Investigators (2012) Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med 367:2089–2099
Hayashi K, Mani V, Nemade A, Aguiar S, Postley JE, Fuster V, Fayad ZA (2010) Variations in atherosclerosis and remodeling patterns in aorta and carotids. J Cardiovasc Magn Reson 12:10
Mani V, Muntner P, Gidding SS, Aguiar SH, El Aidi H, Weinshelbaum KB, Taniguchi H, van der Geest R, Reiber JH, Bansilal S, Farkouh M, Fuster V, Postley JE, Woodward M, Fayad ZA (2009) Cardiovascular magnetic resonance parameters of atherosclerotic plaque burden improve discrimination of prior major adverse cardiovascular events. J Cardiovasc Magn Reson 11:10
Bucerius J, Duivenvoorden R, Mani V, Moncrieff C, Rudd JH, Calcagno C, Machac J, Fuster V, Farkouh ME, Fayad ZA (2011) Prevalence and risk factors of carotid vessel wall inflammation in coronary artery disease patients: FDG-PET and CT imaging study. JACC Cardiovasc Imaging 4:1195–1205
Hashizume M, Mihara M (2012) Atherogenic effects of TNF-α and IL-6 via up-regulation of scavenger receptors. Cytokine 58:424–430
Reimers GJ, Jackson CL, Rickards J, Chan PY, Cohn JS, Rye KA, Barter PJ, Rodgers KJ (2011) Inhibition of rupture of established atherosclerotic plaques by treatment with apolipoprotein A-I. Cardiovasc Res 91:37–44
Morgantini C, Imaizumi S, Grijalva V, Navab M, Fogelman AM, Reddy ST (2010) Apolipoprotein A-I mimetic peptides prevent atherosclerosis development and reduce plaque inflammation in a murine model of diabetes. Diabetes 59:3223–3228
Cimmino G, Ibanez B, Vilahur G, Speidl WS, Fuster V, Badimon L, Badimon JJ (2009) Up-regulation of reverse cholesterol transport key players and rescue from global inflammation by ApoA-I(Milano). J Cell Mol Med 13:3226–3235
Hu MM, Zhang J, Wang WY, Wu WY, Ma YL, Chen WH, Wang YP (2011) The inhibition of lipoprotein-associated phospholipase A2 exerts beneficial effects against atherosclerosis in LDLR-deficient mice. Acta Pharmacol Sin 32:1253–1258
Gonçalves I, Edsfeldt A, Ko NY, Grufman H, Berg K, Björkbacka H, Nitulescu M, Persson A, Nilsson M, Prehn C, Adamski J, Nilsson J (2012) Evidence supporting a key role of Lp-PLA2-generated lysophosphatidylcholine in human atherosclerotic plaque inflammation. Arterioscler Thromb Vasc Biol 32:1505–1512
Agirbasli M (2005) Pivotal role of plasminogen-activator inhibitor 1 in vascular disease. Int J Clin Pract 59:102–106
Raiko JR, Oikonen M, Wendelin-Saarenhovi M, Siitonen N, Kähönen M, Lehtimäki T, Viikari J, Jula A, Loo BM, Huupponen R, Saarikoski L, Juonala M, Raitakari OT (2012) Plasminogen activator inhitor-1 associates with cardiovascular risk factors in healthy young adults in the Cardiovascular Risk in Young Finns Study. Atherosclerosis 224:208–212
Mani V, Aguiar SH, Itskovich VV, Weinshelbaum KB, Postley JE, Wasenda EJ, Aguinaldo JG, Samber DD, Fayad ZA (2006) Carotid black blood MRI burden of atherosclerotic disease assessment correlates with ultrasound intima-media thickness. J Cardiovasc Magn Reson 8:529–534
Bucerius J, Mani V, Moncrieff C, Rudd JH, Machac J, Fuster V, Farkouh ME, Fayad ZA (2012) Impact of noninsulin-dependent type 2 diabetes on carotid wall 18F-fluorodeoxyglucose positron emission tomography uptake. J Am Coll Cardiol 59:2080–2088
Acknowledgments
The authors thank Michael E. Farkouh and Valentin Fuster for their review and feedback of the manuscript. This study was funded by F. Hoffmann-La Roche Ltd. Editorial assistance was provided by Prime Healthcare during the preparation of this report, and funded by F. Hoffmann-La Roche Ltd. All opinions expressed are those of the authors.
Conflict of interest
V.M., J.B. and D.S. indicate they have nothing to disclose. M.W. discloses that he has received honoraria from Roche. A.T. discloses that he has received honoraria from Roche, BMS and Novartis, and research grants from Merck, BMS, Genentech, GSK and VBL. D.K. was an employee of F. Hoffman-La Roche Ltd at the time the study was performed. M.A. is an employee of F. Hoffman-La Roche Ltd and receives share options. J.H.F.R. discloses that he has received honoraria from Roche and is part-supported by the National Institute for Health Research Cambridge Biomedical Research Centre. Z.A.F. discloses that he has received research grants from Roche, GlaxoSmithKline, Merck, VBL Therapeutics, Novartis, Bristol-Myers Squibb, and Via Pharmaceuticals and honoraria from Roche.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mani, V., Woodward, M., Samber, D. et al. Predictors of change in carotid atherosclerotic plaque inflammation and burden as measured by 18-FDG-PET and MRI, respectively, in the dal-PLAQUE study. Int J Cardiovasc Imaging 30, 571–582 (2014). https://doi.org/10.1007/s10554-014-0370-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-014-0370-7