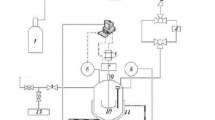
The factors affecting the experimental determination of the conditions of gas hydrates phase equilibria in various systems using the stepwise heating and constant-rate heating procedures in the GHA350 stirring autoclave or the RCS6 rocking cells were analyzed. It was found that more reliable results are obtained with the GHA350 autoclave, which gives more intense agitation of the fluids. The stepwise heating method is a priori a more reliable way for determination of the equilibrium conditions of hydrate formation suitable for any system. However, its use requires a greater expenditure of time. With the alternative method of heating at a constant rate of 0.1-1 deg/h it is possible to reduce the length of a single measurement considerably. Anyway, if it is used to obtain reliable experimental data it is necessary to select a heating rate in line with the mixing efficiency of the employed apparatus, the type of system being investigated, and other factors. For most systems the results of measuring the hydrate equilibrium temperatures by the stepwise method and by continuous heating at 0.1 deg/h using the GHA350 autoclave agree within the experimental error limits. Greater discrepancy between the results of the two methods is observed for gas hydrates in concentrated salt solutions that have high viscosity at low temperatures (below –25°C).





Similar content being viewed by others
References
V. A. Istomin, V. G. Kvon, Prevention and Elimination of Gas Hydrates in Gas Production Systems [in Russian], IRTs Gazprom, Moscow (2005), 556 pp.
A. Y. Manakov, A. S. Stoporev, Russ. Chem. Rev., 90, No. 5, 566-600 (2021).
M. E. Semenov et al., Pet. Chem., 62, No. 2, 127-140 (2022).
B. Tohidi et al., Chem. Eng. Sci., 52, No. 19, 3257-3263 (1997).
A. P. Semenov et al., J. Nat. Gas Sci. Eng., 82, 103491 (2020).
A. P. Semenov et al., Chem. Eng. J., 423, 130227 (2021).
Y. Gong et al., Chem. Eng. J., 429, 132386 (2022).
B. Tohidi et al., Ann. N. Y. Acad. Sci., 912, 924-931 (2000).
A. P. Semenov et al., J. Chem. Thermodyn., 137, 119-130 (2019).
A. P. Semenov et al., Data Br., 42, 108289 (2022).
A. P. Semenov et al., Chem. Eng. Sci., 255, 117670 (2022).
P. Gayet et al., Chem. Eng. Sci., 60, No. 21, 5751-5758 (2005).
A. P. Semenov et al., Chem. Eng. Sci., 137, 161-169 (2015).
A. P. Semenov et al., Fluid Phase Equilib., 432, 1-9 (2017).
A. S. Stoporev et al., J. Pet. Sci. Eng., 176, 952-961 (2019).
M. Tariq et al., Chem. Eng. Sci., 249, 117319 (2022).
S. Adisasmito, R. J. Frank, E. D. Sloan, J. Chem. Eng. Data, 36, No. 1, 68-71 (1991).
A. H. Mohammadi, R. Anderson, B. Tohidi, AIChE J, 51, No. 10, 2825-2833 (2005).
D. R. Marshall, S. Saito., R. Kobayashi, AIChE J, 10, No. 2, 202-205 (1964).
K. Sugahara et al., Open Thermodyn. J., 2, No. 1, 95-99 (2008).
R. Kumar et al., AIChE J, 54, No. 8, 2132-2144 (2008).
A. P. Semenov, A. S. Viktorov, A. B. Berberov, et al., Tekhnologiya Nefti i Gaza, No. 5, 24-32 (2012).
T. Uchida et al., ChemPhysChem, 6, No. 4, 646-654 (2005).
T. Uchida et al., AIChE J, 50, No. 2, 518-523 (2004).
V. I. Medvedev et al., Chem. Technol. Fuels Oils, 51, No. 5, 470-479 (2015).
M. Aminnaji, R. Anderson, B. Tohidi, Energy and Fuels, 35, No. 11, 9341-9348 (2021).
E. D. Sloan, Clathrate Hydrates of Natural Gases. Second, Marcel Dekker (1998).
E. D. Sloan, C. A. Koh, Clathrate Hydrates of Natural Gases. Third, CRC Press (2007).
A. P. Semenov et al., Chem. Technol. Fuels Oils, 54, No. 6, 738-742 (2019).
Acknowledgement
The investigation was supported by a grant from the Russian Science Foundation (project no. 20-79-10377).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya i Tekhnologiya Topliv i Masel, No. 4, pp. 50–56 July –August, 2022.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Semenov, A.P., Mendgaziev, R.I., Tulegenov, T.B. et al. Analysis of the Techniques for Measuring the Equilibrium Conditions of Gas Hydrates Formation. Chem Technol Fuels Oils 58, 628–636 (2022). https://doi.org/10.1007/s10553-022-01429-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10553-022-01429-w