Abstract
Background
There is a well-recognized male excess in childhood cancer incidence; however, it is unclear whether there is etiologic heterogeneity by sex when defined by epidemiologic risk factors.
Methods
Using a 5-state registry-linkage study (cases n = 16,411; controls n = 69,816), we estimated sex-stratified odds ratios (OR) and 95% confidence intervals (95% CI) between birth and demographic characteristics for 16 pediatric cancers. Evidence of statistical interaction (p-interaction < 0.01) by sex was evaluated for each characteristic in each cancer.
Results
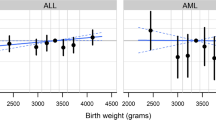
Males comprised > 50% of cases for all cancers, except Wilms tumor (49.6%). Sex interacted with a number of risk factors (all p-interaction < 0.01) including gestational age for ALL (female, 40 vs. 37–39 weeks OR: 0.84, 95% CI 0.73–0.97) and ependymoma (female, 40 vs. 37–39 OR: 1.78, 95% CI 1.14–2.79; female, ≥ 41 OR: 2.01. 95% CI 1.29–3.14), birth order for AML (female, ≥ 3rd vs. 1st OR: 1.39, 95% CI 1.01–1.92), maternal education for Hodgkin lymphoma (male, any college vs. < high school[HS] OR: 1.47, 95% CI 1.03–2.09) and Wilms tumor (female, any college vs. HS OR: 0.74, 95% CI 0.59–0.93), maternal race/ethnicity for neuroblastoma (male, black vs. white OR: 2.21, 95% CI 1.21–4.03; male, Hispanic vs. white OR: 1.86, 95% CI 1.26–2.75; female, Asian/Pacific Islander vs. white OR: 0.28, 95% CI 0.12–0.69), and paternal age (years) for hepatoblastoma in males (< 24 vs. 25–29 OR: 2.17, 95% CI 1.13–4.19; ≥ 35 vs. 25–29 OR: 2.44, 95% CI 1.28–4.64).
Conclusions
These findings suggest etiologic heterogeneity by sex for childhood cancers for gestational age, maternal education, and race/ethnicity and paternal age.

Similar content being viewed by others
Data availability
The data are proprietary and sharing is limited based on each registry’s rules.
References
Williams LA, Richardson M, Marcotte EL et al (2019) Sex ratio among childhood cancers by single year of age. Pediatr Blood Cancer. https://doi.org/10.1002/pbc.27620
Williams LA, Hubbard AK, Scheurer ME et al (2020) Trends in pediatric central nervous system tumor incidence by global region from 1988 to 2012. Int J Epidemiol dyaa. https://doi.org/10.1093/ije/dyaa176
Ward E, Desantis C, Robbins A et al (2014) Childhood and adolescent cancer statistics, 2014. CA: Cancer J Clin 64:83–103. https://doi.org/10.3322/caac.21219
Libert C, Dejager L, Pinheiro I (2010) The X chromosome in immune functions: When a chromosome makes the difference. Nat Rev Immunol 10:594–604. https://doi.org/10.1038/nri2815
Klein SL, Flanagan KL (2016) Sex differences in immune responses. Nat Rev Immunol 16:626–638. https://doi.org/10.1038/nri.2016.90
Washburn T, Medearis D, Child B (1965) Sex differences in susceptibilty to infections. Pediatrics 35:57–64
Piccini P, Montagnani C, De Martino M (2018) Gender disparity in pediatrics: A review of the current literature. Ital J Pediatr 44:4–9. https://doi.org/10.1186/s13052-017-0437-x
Spatz A, Borg C, Feunteun J (2004) X-chromosome genetics and human cancer. Nat Rev Cancer 4:617–629. https://doi.org/10.1038/nrc1413
Khan D, Ahmed SA (2016) The immune system is a natural target for estrogen action: opposing effects of estrogen in two prototypical autoimmune diseases. Front Immunol 6:1–8. https://doi.org/10.3389/fimmu.2015.00635
Khan D, Ahmed A (2012) Estrogen and signaling in the cells of the immune system. Adv Neuroimmune Biol 3:73–93. https://doi.org/10.3233/NIB-2012-012039
Furman D, Hejblum BP, Simon N et al (2014) Systems analysis of sex differences reveals an immunosuppressive role for testosterone in the response to influenza vaccination. Proc Natl Acad Sci 111:869–874. https://doi.org/10.1073/pnas.1321060111
Spector LG, Pankratz N, Marcotte EL (2015) Genetic and nongenetic risk factors for childhood cancer. Pediatr Clin North Am 62:11–25. https://doi.org/10.1016/j.pcl.2014.09.013.Genetic
Von Behren J, Spector LG, Mueller BA et al (2011) Birth order and risk of childhood cancer: a pooled analysis from five US States. Int J Cancer 128:2709–2716. https://doi.org/10.1002/ijc.25593
Johnson KJ, Carozza SE, Chow EJ et al (2009) Parental age and risk of childhood cancer: a pooled analysis. Epidemiology 20:475–483. https://doi.org/10.1097/EDE.0b013e3181a5a332
Chow EJ, Puumala SE, Mueller BA et al (2010) Childhood cancer in relation to parental race and ethnicity: a 5-state pooled analysis. Cancer 116:3045–3053. https://doi.org/10.1002/cncr.25099
Williams LA, Richardson M, Kehm RD et al (2018) The association between sex and most childhood cancers is not mediated by birthweight. Cancer Epidemiol 57:7–12
Puumala SE, Carozza SE, Chow EJ et al (2009) Childhood cancer among twins and higher order multiples. Cancer Epidemiol Biomarkers Prev 18:162–168. https://doi.org/10.1158/1055-9965.EPI-08-0660
Kehm RD, Spector LG, Poynter JN et al (2017) Socioeconomic status and childhood cancer incidence: a population-based multilevel analysis. Am J Epidemiol 187:612–626. https://doi.org/10.1093/aje/kwx292/4080179/Cigarette-Smoking-and-Risk-of-Early-Natural
O’Neill KA, Murphy MFG, Bunch KJ et al (2015) Infant birthweight and risk of childhood cancer: international population-based case control studies of 40 000 cases. Int J Epidemiol 44:153–168. https://doi.org/10.1093/ije/dyu265
Johnson KJ, Carozza SE, Chow EJ et al (2011) Birth characteristics and childhood carcinomas. Br J Cancer 105:1396–1401. https://doi.org/10.1038/bjc.2011.359
Steliarova-Foucher E, Stiller C, Lacour B, Kaatsch P (2005) International classification of childhood cancer, third edition. Cancer 103:1457-1467. https://doi.org/10.1002/cncr.20910
Ostrom QT, De Blank PM, Kruchko C et al (2014) Alex’s Lemonade stand foundation infant and childhood primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol 16:x1–x35. https://doi.org/10.1093/neuonc/nou327
Daniels JL, Pan IJ, Olshan AF et al (2008) Obstetric history and birth characteristics and Wilms tumor: a report from the children’s oncology group. Cancer Causes Control 19:1103–1110. https://doi.org/10.1007/s10552-008-9174-1
Oksuzyan S, Crespi C, Cockburn M et al (2015) Race/ethnicity and the risk childhood leukemia: a case-control study in California. J Epidemiol Community Heal 69:795–802. https://doi.org/10.1136/jech-2014-204975.Race/ethnicity
Schuz J, Kaatsch P, Kaletsch U et al (1999) Association of childhood cancer with factors related to pregnancy and birth. Int J Epidemiol 28:631–639. https://doi.org/10.1038/sj.bjc.6690395
Lupo PJ, Danysh HE, Skapek SX et al (2014) Maternal and birth characteristics and childhood rhabdomyosarcoma: a report from the Children’s Oncology Group. Cancer Causes Control 25:905–913. https://doi.org/10.1007/s10552-014-0390-6
Smith A, Lightfoot T, Simpson J, Roman E (2009) Birth weight, sex and childhood cancer: a report from the United Kingdom Childhood Cancer Study. Cancer Epidemiol 33:363–367. https://doi.org/10.1016/j.canep.2009.10.012
Contreras ZA, Hansen J, Ritz B et al (2017) Parental age and childhood cancer risk: a danish population-based registry study. Cancer Epidemiol 49:202–215. https://doi.org/10.1016/j.canep.2017.06.010
Clocchiatti A, Cora E, Zhang Y, Dotto GP (2016) Sexual dimorphism in cancer. Nat Rev Cancer 16:330–339. https://doi.org/10.1038/nrc.2016.30
Rubin JB, Lagas JS, Broestl L et al (2020) Sex differences in cancer mechanisms. Biol Sex Differ 11:1–29. https://doi.org/10.1186/s13293-020-00291-x
Lunde A, Melve KK, Gjessing HK et al (2007) Genetic and environmental influences on birth weight, birth length, head circumference, and gestational age by use of population-based parent-offspring data. Am J Epidemiol 165:734–741. https://doi.org/10.1093/aje/kwk107
Schüz J, Weihkopf T, Kaatsch P (2007) Medication use during pregnancy and the risk of childhood cancer in the offspring. Eur J Pediatr 166:433–441. https://doi.org/10.1007/s00431-006-0401-z
Marcotte EL, Thomopoulos TP, Infante-Rivard C et al (2016) Caesarean delivery and risk of childhood leukaemia: a pooled analysis from the Childhood Leukemia International Consortium (CLIC). Lancet Haematol 3:e176–e185. https://doi.org/10.1016/S2352-3026(16)00002-8
Deoni SC, Adams SH, Li X et al (2019) Cesarean delivery impacts infant brain development. Am J Neuroradiol 40:169–177. https://doi.org/10.3174/ajnr.A5887
Patel DM, Jones RR, Booth BJ et al (2019) Parental occupational exposure to pesticides, animals and organic dust and risk of childhood leukemia and central nervous system tumors: Findings from the International Childhood Cancer Cohort Consortium (I4C). Int J Cancer. https://doi.org/10.1002/ijc.32388
Williams LA, Richardson M, Spector LG, Marcotte EL (2021) Cesarean section is associated with an increased risk of acute lymphoblastic leukemia and hepatoblastoma in children from minnesota. Cancer Epidemiol Biomarkers Prev 30:736–742. https://doi.org/10.1158/1055-9965.EPI-20-1406
Juraschka K, Taylor MD (2019) Medulloblastoma in the age of molecular subgroups: a review: JNSPG 75th Anniversary invited review article. J Neurosurg Pediatr 24:353–363. https://doi.org/10.3171/2019.5.PEDS18381
Williams LA, Spector LG (2019) Survival differences between males and females diagnosed with childhood cancer. JNCI Cancer Spectr 3:1–11
Williams LA, Yang JJ, Hirsch BA et al (2019) Is there etiologic heterogeneity between subtypes of childhood acute lymphoblastic leukemia? A review of variation in risk by subtype. Cancer Epidemiol Biomarkers Prev. https://doi.org/10.1158/1055-9965.EPI-18-0801
Brumback B, Berg A (2008) On effect-measure modification: Relationships among changes in the relative risk, odds ratio, and risk difference. Stat Methods Med Res 27:3453–3465
Weiss NS (2008) Subgroup-specific associations in the face of overall null results: Should we rush in or fear to tread? Cancer Epidemiol Biomarkers Prev 17:1297–1299. https://doi.org/10.1158/1055-9965.EPI-08-0144
Galante L, Milan AM, Reynolds CM et al (2018) Sex-specific human milk composition: The role of infant sex in determining early life nutrition. Nutrients 10:1–11. https://doi.org/10.3390/nu10091194
Funding
The Children’s Cancer Research Fund (LAW), the Washington State Cancer Registry and the Cancer Surveillance System of Western Washington, which provided data, are supported by contract N01-CN-05230 from the National Cancer Institute and the Fred Hutchinson Cancer Research Center. In California and Texas, National Cancer Institute grants R01 CA717450 and R01 CA92670 supported assembly of their respective datasets. In New York, partial support for assembly of the dataset was received from the Centers for Disease Control and Prevention’s National Program of Cancer Registries by cooperative agreement U58DP000783-01 awarded to the New York State Department of Health; contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of the Centers for Disease Control and Prevention.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Williams, L.A., Sample, J., McLaughlin, C.C. et al. Sex differences in associations between birth characteristics and childhood cancers: a five-state registry-linkage study. Cancer Causes Control 32, 1289–1298 (2021). https://doi.org/10.1007/s10552-021-01479-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-021-01479-1