Abstract
Background
Sexually transmitted infections and chronic inflammation have been associated with an increased risk of prostate cancer. Inflammatory mediators, such as cytokines and free radicals, have been hypothesized to play a role.
Methods
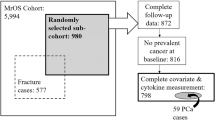
To explore the role of inflammation in prostate cancer risk further, we examined the association between pre-diagnostic serum levels of interleukin-16 (IL-16), an important pleiotropic cytokine, and prostate cancer risk among 932 Caucasian cases and 942 controls and 154 African-American cases and 302 controls in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Serum IL-16 was quantified using enzyme-linked immunoassay. Logistic regression was used to estimate associations between IL-16 and prostate cancer risk, separately by race.
Results
Although no association between IL-16 and prostate cancer overall was observed among Caucasians (p = 0.27), a significantly increased risk of high-grade prostate cancer, defined as Gleason ≥ 7 (phet = 0.02), was observed with increasing levels of IL-16 (OR3rd vs. 1st tertile = 1.37, 95% CI 1.04–1.81, ptrend = 0.02). We also discovered a significant interaction between IL-16 and history of gonorrhea (p = 0.04). Among Caucasian men with a history of gonorrhea, elevated IL-16 levels were associated with an increased risk of prostate cancer (OR3rd vs. 1st tertile = 3.64, 95% CI 1.14–11.6) but no association was seen among those without a history of gonorrhea (OR3rd vs. 1st tertile = 1.06, 95% CI 0.83–1.34). No associations were observed among African-Americans.
Conclusions
This study found evidence that higher pre-diagnostic IL-16 levels may be associated with increased risk of high-grade disease, supporting inflammation as potential mechanism by which sexually transmitted diseases may increase risk.
Similar content being viewed by others
References
Dennis LK, Lynch CF, Torner JC (2002) Epidemiologic association between prostatitis and prostate cancer. Urology 60(1):78–83
Roberts RO, Bergstralh EJ, Bass SE, Lieber MM, Jacobsen SJ (2004) Prostatitis as a risk factor for prostate cancer. Epidemiology 15(1):93–99
Caini S, Gandini S, Dudas M, Bremer V, Severi E, Gherasim A (2014) Sexually transmitted infections and prostate cancer risk: a systematic review and meta-analysis. Cancer Epidemiol 38(4):329–338
Lian WQ, Luo F, Song XL, Lu YJ, Zhao SC (2015) Gonorrhea and prostate cancer incidence: an updated meta-analysis of 21 epidemiologic studies. Med Sci Monit 21:1902–1910
Zhang X, Zhou G, Sun B, Zhao G, Liu D, Sun J et al (2015) Impact of obesity upon prostate cancer-associated mortality: a meta-analysis of 17 cohort studies. Oncol Lett 9(3):1307–1312
Vidal AC, Howard LE, Moreira DM, Castro-Santamaria R, Andriole GL, Freedland SJ (2015) Aspirin, NSAIDs, and risk of prostate cancer: results from the REDUCE study. Clin Cancer Res 21(4):756–762
Dasgupta K, Di Cesar D, Ghosn J, Rajan R, Mahmud S, Rahme E (2006) Association between nonsteroidal anti-inflammatory drugs and prostate cancer occurrence. Cancer J 12(2):130–135
Nelson JE, Harris RE (2000) Inverse association of prostate cancer and non-steroidal anti-inflammatory drugs (NSAIDs): results of a case-control study. Oncol Rep 7(1):169–170
Roberts RO, Jacobson DJ, Girman CJ, Rhodes T, Lieber MM, Jacobsen SJ (2002) A population-based study of daily nonsteroidal anti-inflammatory drug use and prostate cancer. Mayo Clin Proc 77(3):219–225
Mahmud SM, Franco EL, Aprikian AG (2010) Use of nonsteroidal anti-inflammatory drugs and prostate cancer risk: a meta-analysis. Int J Cancer 127(7):1680–1691
Shah R, Mucci NR, Amin A, Macoska JA, Rubin MA (2001) Postatrophic hyperplasia of the prostate gland: neoplastic precursor or innocent bystander? Am J Pathol 158(5):1767–1773
Woenckhaus J, Fenic I (2008) Proliferative inflammatory atrophy: a background lesion of prostate cancer? Andrologia 40(2):134–137
Wang W, Bergh A, Damber JE (2009) Morphological transition of proliferative inflammatory atrophy to high-grade intraepithelial neoplasia and cancer in human prostate. Prostate 69(13):1378–1386
Schatteman PH, Hoekx L, Wyndaele JJ, Jeuris W, Van Marck E (2000) Inflammation in prostate biopsies of men without prostatic malignancy or clinical prostatitis: correlation with total serum PSA and PSA density. Eur Urol 37(4):404–412
Gerstenbluth RE, Seftel AD, MacLennan GT, Rao RN, Corty EW, Ferguson K et al (2002) Distribution of chronic prostatitis in radical prostatectomy specimens with up-regulation of bcl-2 in areas of inflammation. J Urol 167(5):2267–2270
Di Silverio F, Gentile V, De Matteis A, Mariotti G, Giuseppe V, Luigi PA et al (2003) Distribution of inflammation, pre-malignant lesions, incidental carcinoma in histologically confirmed benign prostatic hyperplasia: a retrospective analysis. Eur Urol 43(2):164–175
Gurel B, Lucia MS, Thompson IM Jr, Goodman PJ, Tangen CM, Kristal AR et al (2014) Chronic inflammation in benign prostate tissue is associated with high-grade prostate cancer in the placebo arm of the prostate cancer prevention trial. Cancer Epidemiol Biomark Prev 23(5):847–856
Glass WG, Sarisky RT, Vecchio AM (2006) Not-so-sweet sixteen: the role of IL-16 in infectious and immune-mediated inflammatory diseases. J Interferon Cytokine Res 26(8):511–520
Cruikshank WW, Kornfeld H, Center DM (2000) Interleukin-16. J Leukoc Biol 67(6):757–766
Bisset LR, Rothen M, Joller-Jemelka HI, Dubs RW, Grob PJ, Opravil M (1997) Change in circulating levels of the chemokines macrophage inflammatory proteins 1 alpha and 11 beta, RANTES, monocyte chemotactic protein-1 and interleukin-16 following treatment of severely immunodeficient HIV-infected individuals with indinavir. Aids 11(4):485–491
Scala E, D’Offizi G, Rosso R, Turriziani O, Ferrara R, Mazzone AM et al (1997) C-C chemokines, IL-16, and soluble antiviral factor activity are increased in cloned T cells from subjects with long-term nonprogressive HIV infection. J Immunol 158(9):4485–4492
Lee S, Kaneko H, Sekigawa I, Tokano Y, Takasaki Y, Hashimoto H (1998) Circulating interleukin-16 in systemic lupus erythematosus. Br J Rheumatol 37(12):1334–1337
Lard LR, Roep BO, Verburgh CA, Zwinderman AH, Huizinga TW (2002) Elevated IL-16 levels in patients with systemic lupus erythematosus are associated with disease severity but not with genetic susceptibility to lupus. Lupus 11(3):181–185
Kaufmann J, Franke S, Kientsch-Engel R, Oelzner P, Hein G, Stein G (2001) Correlation of circulating interleukin 16 with proinflammatory cytokines in patients with rheumatoid arthritis. Rheumatology 40(4):474–475
Duan H, Fleming J, Pritchard DK, Amon LM, Xue J, Arnett HA et al (2008) Combined analysis of monocyte and lymphocyte messenger RNA expression with serum protein profiles in patients with scleroderma. Arthritis Rheum 58(5):1465–1474
Alexandrakis MG, Passam FH, Kyriakou DS, Christophoridou AV, Perisinakis K, Hatzivasili A et al (2004) Serum level of interleukin-16 in multiple myeloma patients and its relationship to disease activity. Am J Hematol 75(2):101–106
Gao LB, Rao L, Wang YY, Liang WB, Li C, Xue H et al (2009) The association of interleukin-16 polymorphisms with IL-16 serum levels and risk of colorectal and gastric cancer. Carcinogenesis 30(2):295–299
Comperat E, Roupret M, Drouin SJ, Camparo P, Bitker MO, Houlgatte A et al (2010) Tissue expression of IL16 in prostate cancer and its association with recurrence after radical prostatectomy. Prostate 70(15):1622–1627
Mathy NL, Scheuer W, Lanzendorfer M, Honold K, Ambrosius D, Norley S et al (2000) Interleukin-16 stimulates the expression and production of pro-inflammatory cytokines by human monocytes. Immunology 100(1):63–69
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140(6):883–899
Gohagan JK, Prorok PC, Hayes RB, Kramer BS (2000) The Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial of the National Cancer Institute: history, organization, and status. Control Clin Trials 21(6 Suppl):251 s-272 s
Hayes RB, Reding D, Kopp W, Subar AF, Bhat N, Rothman N et al (2000) Etiologic and early marker studies in the prostate, lung, colorectal and ovarian (PLCO) cancer screening trial. Control Clin Trials 21(6 Suppl):349s–355s
Yeager M, Chatterjee N, Ciampa J, Jacobs KB, Gonzalez-Bosquet J, Hayes RB et al (2009) Identification of a new prostate cancer susceptibility locus on chromosome 8q24. Nat Genet 41(10):1055–1057
Huang WY, Hayes R, Pfeiffer R, Viscidi RP, Lee FK, Wang YF et al (2008) Sexually transmissible infections and prostate cancer risk. Cancer Epidemiol Biomark Prev 17(9):2374–2381
Tuzova M, Richmond J, Wolpowitz D, Curiel-Lewandrowski C, Chaney K, Kupper T et al (2015) CCR4 + T cell recruitment to the skin in mycosis fungoides: potential contributions by thymic stromal lymphopoietin and interleukin-16. Leuk Lymphoma 56(2):440–449
Yellapa A, Bitterman P, Sharma S, Guirguis AS, Bahr JM, Basu S et al (2014) Interleukin 16 expression changes in association with ovarian malignant transformation. Am J Obstet Gynecol 210(3):272.e1-10
Purzycka-Bohdan D, Szczerkowska-Dobosz A, Zablotna M, Wierzbicka J, Piotrowska A, Zmijewski MA et al (2016) Assessment of interleukin 16 serum levels and skin expression in psoriasis patients in correlation with clinical severity of the disease. PLoS ONE 11(10):e0165577
Nischwitz S, Faber H, Samann PG, Domingues HS, Krishnamoorthy G, Knop M et al (2014) Interferon beta-1a reduces increased interleukin-16 levels in multiple sclerosis patients. Acta Neurol Scand 130(1):46–52
Skundric DS, Dai R, Zakarian VL, Bessert D, Skoff RP, Cruikshank WW et al (2005) Anti-IL-16 therapy reduces CD4 + T-cell infiltration and improves paralysis and histopathology of relapsing EAE. J Neurosci Res 79(5):680–693
Paalani M, Lee JW, Haddad E, Tonstad S (2011) Determinants of inflammatory markers in a bi-ethnic population. Ethn Dis 21(2):142–149
Morimoto Y, Conroy SM, Ollberding NJ, Kim Y, Lim U, Cooney RV et al (2014) Ethnic differences in serum adipokine and C-reactive protein levels: the multiethnic cohort. Int J Obes 38(11):1416–1422
Fisher G, Hyatt TC, Hunter GR, Oster RA, Desmond RA, Gower BA (2012) Markers of inflammation and fat distribution following weight loss in African-American and white women. Obesity 20(4):715–720
Stowe RP, Peek MK, Cutchin MP, Goodwin JS (2010) Plasma cytokine levels in a population-based study: relation to age and ethnicity. J Gerontol A Biol Sci Med Sci 65(4):429–433
Contreras-Sesvold C, Revenis BD, O’Connor FG, Deuster PA (2015) Association of plasma heat shock protein 70, interleukin 6, and creatine kinase concentrations in a healthy, young adult population. J Biomark 2015:967120
Sutcliffe S, Giovannucci E, De Marzo AM, Leitzmann MF, Willett WC, Platz EA (2006) Gonorrhea, syphilis, clinical prostatitis, and the risk of prostate cancer. Cancer Epidemiol Biomark Prev 15(11):2160–2166
Patel DA, Bock CH, Schwartz K, Wenzlaff AS, Demers RY, Severson RK (2005) Sexually transmitted diseases and other urogenital conditions as risk factors for prostate cancer: a case–control study in Wayne County, Michigan. Cancer Causes Control 16(3):263–273
Pelucchi C, Talamini R, Negri E, Franceschi S, La Vecchia C (2006) Genital and urinary tract diseases and prostate cancer risk. Eur J Cancer Prev 15(3):254–257
De Marzo AM, Platz EA, Sutcliffe S, Xu J, Gronberg H, Drake CG et al (2007) Inflammation in prostate carcinogenesis. Nat Rev Cancer 7(4):256–269
McDonald AC, Vira MA, Vidal AC, Gan W, Freedland SJ, Taioli E (2014) Association between systemic inflammatory markers and serum prostate-specific antigen in men without prostatic disease—the 2001–2008 National Health and Nutrition Examination Survey. Prostate 74(5):561–567
Elzanaty S, Rezanezhad B, Borgquist R (2016) Association between PSA Levels and biomarkers of subclinical systemic inflammation in middle-aged healthy men from the general population. Curr Urol 9(3):148–152
Funding
This study was supported by the Intramural Research Program of the Division of Cancer Epidemiology and Genetics, National Cancer Institute, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moore, A., Huang, WY., Danforth, K. et al. Prospective evaluation of serum IL-16 and risk of prostate cancer in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Cancer Causes Control 29, 455–464 (2018). https://doi.org/10.1007/s10552-018-1012-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-018-1012-5