Abstract
Purpose
Metronomic chemotherapy has the potential to offer tumor control with reduced toxicity when compared to standard dose chemotherapy in patients with metastatic breast cancer. As metronomic chemotherapy may target the tumor microvasculature, it has the potential for synergistic effects with antiangiogenic agents such as the VEGF-A inhibitor bevacizumab.
Methods
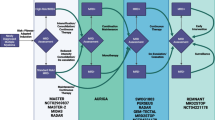
In this randomized phase II study, patients with metastatic breast cancer were randomized to receive metronomic oral cyclophosphamide and methotrexate (CM) combined with bevacizumab (Arm A) or CM alone (Arm B). The primary endpoint was objective response rate (ORR). Secondary endpoints included progression-free survival (PFS), overall survival (OS), and safety and tolerability.
Results
A total of 55 patients were enrolled, with 34 patients treated on Arm A and 21 patients treated on Arm B. The ORR was modestly higher in Arm A (26%) than in Arm B (10%); neither met the 40% cutoff for further clinical evaluation. The median time to progression (TTP) was 5.52 months (3.22–13.6) on Arm A and 1.82 months (1.54–6.70) on Arm B (log-rank p = 0.008). The median OS was 29.6 months (17.2–NA) on Arm A and 16.2 months (15.7–NA) on Arm B (log-rank p = 0.7). Common all-grade adverse events in both arms included nausea, fatigue, and elevated AST.
Conclusion
The combination of metronomic CM with bevacizumab significantly improved PFS over CM alone, although there was no significant difference in OS. Oral metronomic chemotherapy alone has limited activity in advanced breast cancer.
ClinicalTrials.gov Identifier: NCT00083031.
Date of Registration: May 17, 2004.


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin. https://doi.org/10.3322/caac.21660
Saphner T, Tormey DC, Gray R (1996) Annual hazard rates of recurrence for breast cancer after primary therapy. J Clin Oncol 14(10):2738–2746. https://doi.org/10.1200/JCO.1996.14.10.2738
Dawood S, Broglio K, Ensor J, Hortobagyi GN, Giordano SH (2010) Survival differences among women with de novo stage IV and relapsed breast cancer. Ann Oncol 21(11):2169–2174. https://doi.org/10.1093/annonc/mdq220
Andre F, Slimane K, Bachelot T, Dunant A, Namer M, Barrelier A, Kabbaj O, Spano JP, Marsiglia H, Rouzier R, Delaloge S, Spielmann M (2004) Breast cancer with synchronous metastases: trends in survival during a 14-year period. J Clin Oncol 22(16):3302–3308. https://doi.org/10.1200/JCO.2004.08.095
Scharovsky OG, Mainetti LE, Rozados VR (2009) Metronomic chemotherapy: changing the paradigm that more is better. Curr Oncol 16(2):7–15. https://doi.org/10.3747/co.v16i2.420
Munzone E, Colleoni M (2015) Clinical overview of metronomic chemotherapy in breast cancer. Nat Rev Clin Oncol 12(11):631–644. https://doi.org/10.1038/nrclinonc.2015.131
Colleoni M, Rocca A, Sandri MT, Zorzino L, Masci G, Nole F, Peruzzotti G, Robertson C, Orlando L, Cinieri S, de BF, Viale G, Goldhirsch A, (2002) Low-dose oral methotrexate and cyclophosphamide in metastatic breast cancer: antitumor activity and correlation with vascular endothelial growth factor levels. Ann Oncol 13(1):73–80
Orlando L, Cardillo A, Rocca A, Balduzzi A, Ghisini R, Peruzzotti G, Goldhirsch A, D’Alessandro C, Cinieri S, Preda L, Colleoni M (2006) Prolonged clinical benefit with metronomic chemotherapy in patients with metastatic breast cancer. Anticancer Drugs 17(8):961–967
Wong NS, Buckman RA, Clemons M, Verma S, Dent S, Trudeau ME, Roche K, Ebos J, Kerbel R, Deboer GE, Sutherland DJ, Emmenegger U, Slingerland J, Gardner S, Pritchard KI (2010) Phase I/II trial of metronomic chemotherapy with daily dalteparin and cyclophosphamide, twice-weekly methotrexate, and daily prednisone as therapy for metastatic breast cancer using vascular endothelial growth factor and soluble vascular endothelial growth factor receptor levels as markers of response. J Clin Oncol 28(5):723–730
Orlando L, Cardillo A, Ghisini R, Rocca A, Balduzzi A, Torrisi R, Peruzzotti G, Goldhirsch A, Pietri E, Colleoni M (2006) Trastuzumab in combination with metronomic cyclophosphamide and methotrexate in patients with HER-2 positive metastatic breast cancer. BMC Cancer 6:225
Gately S, Kerbel R (2001) Antiangiogenic scheduling of lower dose cancer chemotherapy. Cancer J 7(5):427–436
Hanahan D, Bergers G, Bergsland E (2000) Less is more, regularly: metronomic dosing of cytotoxic drugs can target tumor angiogenesis in mice. J Clin Invest 105(8):1045–1047. https://doi.org/10.1172/JCI9872
Man S, Bocci G, Francia G, Green SK, Jothy S, Hanahan D, Bohlen P, Hicklin DJ, Bergers G, Kerbel RS (2002) Antitumor effects in mice of low-dose (metronomic) cyclophosphamide administered continuously through the drinking water. Cancer Res 62(10):2731–2735
Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, Shenkier T, Cella D, Davidson NE (2007) Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med 357(26):2666–2676
Miles DW, Chan A, Dirix LY, Cortes J, Pivot X, Tomczak P, Delozier T, Sohn JH, Provencher L, Puglisi F, Harbeck N, Steger GG, Schneeweiss A, Wardley AM, Chlistalla A, Romieu G (2010) Phase III study of bevacizumab plus docetaxel compared with placebo plus docetaxel for the first-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J Clin Oncol 28(20):3239–3247
Robert NJ, Dieras V, Glaspy J, Brufsky AM, Bondarenko I, Lipatov ON, Perez EA, Yardley DA, Chan SY, Zhou X, Phan SC, O’Shaughnessy J (2011) RIBBON-1: randomized, double-blind, placebo-controlled, phase III trial of chemotherapy with or without bevacizumab for first-line treatment of human epidermal growth factor receptor 2-negative, locally recurrent or metastatic breast cancer. J Clin Oncol 29(10):1252–1260
O’Shaughnessy J, Miles D, Gray R, Dieras V, Perez E, Zon R, Cortes J, Zhou X, Phan S, Miller K (2010) A meta-analysis of overall survival data from three randomized trials of bevacizumab (BV) and first-line chemotherapy as treatment for patients with metastatic breast cancer (MBC). J Clin Oncol 28(15S):A1005
Montagna E, Cancello G, Bagnardi V, Pastrello D, Dellapasqua S, Perri G, Viale G, Veronesi P, Luini A, Intra M, Calleri A, Rampinelli C, Goldhirsch A, Bertolini F, Colleoni M (2012) Metronomic chemotherapy combined with bevacizumab and erlotinib in patients with metastatic HER2-negative breast cancer: clinical and biological activity. Clin Breast Cancer 12(3):207–214. https://doi.org/10.1016/j.clbc.2012.03.008
Dellapasqua S, Bertolini F, Bagnardi V, Campagnoli E, Scarano E, Torrisi R, Shaked Y, Mancuso P, Goldhirsch A, Rocca A, Pietri E, Colleoni M (2008) Metronomic cyclophosphamide and capecitabine combined with bevacizumab in advanced breast cancer. J Clin Oncol 26(30):4899–4905
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Vergato C, Doshi KA, Roblyer D, Waxman DJ (2022) Type-I interferon signaling is essential for robust metronomic chemo-immunogenic tumor regression in murine breast cancer. Cancer Res Commun 4:246–257. https://doi.org/10.1158/2767-9764.Crc-21-0148
He X, Du Y, Wang Z, Wang X, Duan J, Wan R, Xu J, Zhang P, Wang D, Tian Y, Han J, Fei K, Bai H, Tian J, Wang J (2020) Upfront dose-reduced chemotherapy synergizes with immunotherapy to optimize chemoimmunotherapy in squamous cell lung carcinoma. J Immunother Cancer. https://doi.org/10.1136/jitc-2020-000807
Kaneno R, Shurin GV, Kaneno FM, Naiditch H, Luo J, Shurin MR (2011) Chemotherapeutic agents in low noncytotoxic concentrations increase immunogenicity of human colon cancer cells. Cell Oncol (Dordr) 34(2):97–106. https://doi.org/10.1007/s13402-010-0005-5
Kaneno R, Shurin GV, Tourkova IL, Shurin MR (2009) Chemomodulation of human dendritic cell function by antineoplastic agents in low noncytotoxic concentrations. J Transl Med 7:58. https://doi.org/10.1186/1479-5876-7-58
Shurin GV, Tourkova IL, Kaneno R, Shurin MR (2009) Chemotherapeutic agents in noncytotoxic concentrations increase antigen presentation by dendritic cells via an IL-12-dependent mechanism. J Immunol 183(1):137–144. https://doi.org/10.4049/jimmunol.0900734
Ghiringhelli F, Menard C, Puig PE, Ladoire S, Roux S, Martin F, Solary E, Le Cesne A, Zitvogel L, Chauffert B (2007) Metronomic cyclophosphamide regimen selectively depletes CD4+CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunol Immunother 56(5):641–648. https://doi.org/10.1007/s00262-006-0225-8
Banissi C, Ghiringhelli F, Chen L, Carpentier AF (2009) Treg depletion with a low-dose metronomic temozolomide regimen in a rat glioma model. Cancer Immunol Immunother 58(10):1627–1634. https://doi.org/10.1007/s00262-009-0671-1
Zhao J, Cao Y, Lei Z, Yang Z, Zhang B, Huang B (2010) Selective depletion of CD4+CD25+Foxp3+ regulatory T cells by low-dose cyclophosphamide is explained by reduced intracellular ATP levels. Cancer Res 7(12):4850–4858. https://doi.org/10.1158/0008-5472.CAN-10-0283
Kan S, Hazama S, Maeda K, Inoue Y, Homma S, Koido S, Okamoto M, Oka M (2012) Suppressive effects of cyclophosphamide and gemcitabine on regulatory T-cell induction in vitro. Anticancer Res 32(12):5363–5369
Khan KA, Ponce de Leon JL, Benguigui M, Xu P, Chow A, Cruz-Munoz W, Man S, Shaked Y, Kerbel RS (2020) Immunostimulatory and anti-tumor metronomic cyclophosphamide regimens assessed in primary orthotopic and metastatic murine breast cancer. NPJ Breast Cancer 6:29. https://doi.org/10.1038/s41523-020-0171-1
Schutz FA, Jardim DL, Je Y, Choueiri TK (2011) Haematologic toxicities associated with the addition of bevacizumab in cancer patients. Eur J Cancer 47(8):1161–1174. https://doi.org/10.1016/j.ejca.2011.03.005
Choueiri TK, Mayer EL, Je Y, Rosenberg JE, Nguyen PL, Azzi GR, Bellmunt J, Burstein HJ, Schutz FA (2011) Congestive heart failure risk in patients with breast cancer treated with bevacizumab. J Clin Oncol 29(6):632–638. https://doi.org/10.1200/JCO.2010.31.9129
Gray R, Bhattacharya S, Bowden C, Miller K, Comis RL (2009) Independent review of E2100: a phase III trial of bevacizumab plus paclitaxel versus paclitaxel in women with metastatic breast cancer. J Clin Oncol 27(30):4966–4972. https://doi.org/10.1200/JCO.2008.21.6630
Tanne JH (2011) FDA cancels approval for bevacizumab in advanced breast cancer. BMJ 343:d7684. https://doi.org/10.1136/bmj.d7684
Colleoni M, Orlando L, Sanna G, Rocca A, Maisonneuve P, Peruzzotti G, Ghisini R, Sandri MT, Zorzino L, Nole F, Viale G, Goldhirsch A (2006) Metronomic low-dose oral cyclophosphamide and methotrexate plus or minus thalidomide in metastatic breast cancer: antitumor activity and biological effects. Ann Oncol 17(2):232–238. https://doi.org/10.1093/annonc/mdj066
Krajnak S, Schnatz C, Almstedt K, Brenner W, Haertner F, Heimes AS, Lebrecht A, Makris GM, Schwab R, Hasenburg A, Schmidt M, Battista MJ (2020) Low-dose metronomic chemotherapy as an efficient treatment option in metastatic breast cancer-results of an exploratory case-control study. Breast Cancer Res Treat 182(2):389–399. https://doi.org/10.1007/s10549-020-05711-5
Krajnak S, Battista M, Brenner W, Almstedt K, Elger T, Heimes AS, Hasenburg A, Schmidt M (2018) Explorative analysis of low-dose metronomic chemotherapy with cyclophosphamide and methotrexate in a cohort of metastatic breast cancer patients. Breast Care (Basel) 13(4):272–276. https://doi.org/10.1159/000487629
Lu Q, Lee K, Xu F, Xia W, Zheng Q, Hong R, Jiang K, Zhai Q, Li Y, Shi Y, Yuan Z, Wang S (2020) Metronomic chemotherapy of cyclophosphamide plus methotrexate for advanced breast cancer: real-world data analyses and experience of one center. Cancer Commun (London) 40(5):222–233. https://doi.org/10.1002/cac2.12029
Hussein MM, Gaafar RM, Abdel-Warith AM, Ahmed WA, Allahloubi NMA, Salem SE, Abdel-Salam IM (2017) Efficacy and toxicity of metronomic chemotherapy in metastatic breast cancer: Egyptian experience. Clin Breast Cancer 17(8):618–628. https://doi.org/10.1016/j.clbc.2017.05.001
Munzone E, Regan MM, Cinieri S, Montagna E, Orlando L, Shi R, Campadelli E, Gianni L, Palleschi M, Petrelli F, Bengala C, Generali D, Collova E, Puglisi F, Cretella E, Zamagni C, Chini C, Ruepp B, Loi S, Colleoni M, International Breast Cancer Study G (2023) Efficacy of metronomic oral vinorelbine, cyclophosphamide, and capecitabine vs weekly intravenous paclitaxel in patients with estrogen receptor-positive, ERBB2-negative metastatic breast cancer: final results from the phase 2 METEORA-II randomized clinical trial. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2023.2150
Acknowledgements
We would like to thank Timothy Erick, PhD for medical writing support, and Valerie Hope Goldstein for medical editing and submission support; both are full-time employees of Dana-Farber Cancer Institute.
Funding
This work was supported by a National Cancer Institute (NCI)—Avon Partners for Progress Award, the NCI DF/HCC SPORE in Breast Cancer (NIH Grant Number: P50CA168504), and Genentech, Inc.
Author information
Authors and Affiliations
Contributions
Study Conception and Design: DRS, DAY, HAB, PDR, LNH, EPW, HJB. Data Collection and Curation: JJS, DRS, DAY, HAB, PDR, LNH, EPW, HJB. Data Analysis: NT, SR, ELM, HJB. Writing, Original Draft: HJB. Writing, Review and Editing: All Authors. Approval of Final Manuscript: All Authors.
Corresponding author
Ethics declarations
Competing interests
ELM reports a consulting role with Lilly, Novartis, AstraZeneca, and Gilead. HAB reports research grants paid to institution from Abbvie, Agios, ARMO Biosciences, Array BioPharma, Arvinas, AstraZeneca, Bayer, BeiGene, BioAtla, BioMed Valley Discoveries, BioTheryX, Boehringer Ingelheim, Bristol Myers Squibb, CALGB, Celgene, CicloMed, Coordination Pharmaceuticals, eFFECTOR Therapeutics, Lilly, EMD Serono, Roche/Genentech, GlaxoSmithKline, Gossamer Bio, Harpoon Therapeutics, Hengrui Therapeutics, InCyte, Janssen, Jounce Therapeutics, Kymab, MacroGenics, MedImmune, Merck, Millennium/Takeda, Moderna, NGM Biopharmaceuticals, Novartis, Pfizer, Revolution Medicines, Ryvu Therapeutics, Foundation Medicine, SeaGen, Tesaro, TG Therapeutics, Varastem, Vertex, XBiotech, Zymeworks; Consulting—Paid to Institution from GRAIL, Roche, Vincerx Pharma; Non-Compensated Consulting from Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Novartis, and TG Therapeutics. DRS reports research grants paid to institution from Abbvie, Aeglea BioTherapeutics, Agios, Amgen, AnHeart Therapeutics, Apollomics, Arcus, Arrys Therapeutics, Ascendis Pharma, Asher Biotherapeutics, Astellas, AstraZeneca, Bayer, BeiGene, Bind Therapeutics, BioNTech, Blueprint Medicine, Boehringer Ingelheim, Bristol Myers Squibb, Calithera, Celgene, Celldex, Clovis, Cyteir Therapeutics, Daiichi Sankyo, Eisai, Elevation Oncology, Endeavor, Erasca, Faeth Therapeutics, Fujifilm Pharmaceuticals, G1 Therapeutics, Roche/Genentech, Gilead Sciences, GlaxoSmithKline, GRAIL, Hutchison MediPharma, lmClose Systems, lncyte, Ipsen, Janssen, Jazz Pharmaceuticals, Kronos Bio, Lilly, Loxo Oncology, Lyell lmmunopharma, MacroGenics, Medlmmune, Merck, Millennium Pharmaceuticals, Moderna, Molecular Template, Monte Rosa Therapeutics, Nektar, Neon Therapeutics, Novartis, Novocure, Peloton Therapeutics, PureTech Health, Razor Genomics, Repare Therapeutics, Rgenix (Inspirna), SeaGen, Shenzhen Chipscreen Biosciences, Stemline Therapeutics, Strata Oncology, Synthekine, Taiho, Tango Therapeutics, Tarveda, Tesaro, Tizona Therapeutics, Transgene, UT Southwestern, Verastem, and Zai Laboratory; and payment made to institution for consulting work conducted for Abbvie, AstraZeneca, BeiGene, Bristol Myers Squibb, Evidera, GlaxoSmithKline, Ipsen Biopharmaceuticals, Janssen, Jazz Pharmaceuticals, Lilly, Molecular Templates, Monte Rosa Therapeutics, Novartis, Novosure, Pfizer, Regeneron Pharmaceuticals, Roche/Genentech, and Sanofi-Aventis. The remaining authors declare no competing interests.
Ethical approval
This study was conducted in line with the principles of the Declaration of Helsinki. Approval was granted by the Dana-Farber/Harvard Cancer Center (DF/HCC) Institutional Review Board (IRB). All participants provided written informed consent before being enrolled on the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Prior Presentations: Results of this study were presented at the 2005 San Antonio Breast Cancer Symposium in San Antonio, TX, USA.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mayer, E.L., Tayob, N., Ren, S. et al. A randomized phase II study of metronomic cyclophosphamide and methotrexate (CM) with or without bevacizumab in patients with advanced breast cancer. Breast Cancer Res Treat 204, 123–132 (2024). https://doi.org/10.1007/s10549-023-07167-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-07167-9