Abstract
Purpose
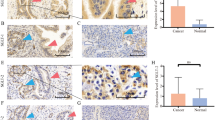
Sodium/glucose cotransporter (SGLT) 1 and 2 expression in carcinoma cells was recently examined, but their association with the clinicopathological factors of the patients and their biological effects on breast carcinoma cells have remained remain virtually unknown. Therefore, in this study, we explored the expression status of SGLT1 and SGLT2 in breast cancer patients and examined the effects of SGLT1 inhibitors on breast carcinoma cells in vitro.
Methods
SGLT1 and SGLT2 were immunolocalized and we first correlated the findings with clinicopathological factors of the patients. We then administered mizagliflozin and KGA-2727, SGLT1 specific inhibitors to MCF-7 and MDA-MB-468 breast carcinoma cell lines, and their growth-inhibitory effects were examined. Protein arrays were then used to further explore their effects on the growth factors.
Results
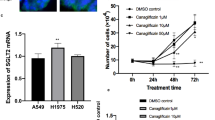
The SGLT1 high group had significantly worse clinical outcome including both overall survival and disease-free survival than low group. SGLT2 status was not significantly correlated with clinical outcome of the patients. Both mizagliflozin and KGA-2727 inhibited the growth of breast cancer cell lines. Of particular interest, mizagliflozin inhibited the proliferation of MCF-7 cells, even under very low glucose conditions. Mizagliflozin downregulated vascular endothelial growth factor receptor 2 phosphorylation.
Conclusion
High SGLT1 expression turned out as an adverse clinical prognostic factor in breast cancer patient. This is the first study demonstrating that SGLT1 inhibitors suppressed breast carcinoma cell proliferation. These results indicated that SGLT1 inhibitors could be used as therapeutic agents for breast cancer patients with aggressive biological behaviors.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article and its supplementary information files.
Abbreviations
- ATCC:
-
American type culture collection
- DAB:
-
3,3′-Diaminobenzidine
- DFS:
-
Disease-free survival
- DMSO:
-
Dimethyl sulfoxide
- EGFR:
-
Epidermal growth factor receptor
- ER:
-
Estrogen receptor
- DCIS:
-
Ductal carcinoma in situ
- HER2:
-
Human epidermal growth factor receptor 2
- IDC:
-
Invasive ductal carcinoma
- GLUT:
-
Glucose transporters
- OS:
-
Overall survival
- PET:
-
Positron emission tomography
- PI3K:
-
Phosphoinositide-3 kinase
- PgR:
-
Progesterone receptor
- PVDF:
-
Polyvinylidene difluoride
- TBS-T:
-
Tris-buffered saline containing Tween-20
- SGLT:
-
Sodium/glucose cotransporters
- VEGFR-2:
-
Vascular endothelial growth factor receptor-2
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Carey LA, Perou CM, Livasy CA et al (2006) Race, breast cancer subtypes, and survival in the Carolina breast cancer study. JAMA 295:2492–2502
Ryser MD, Weaver DL, Zhao F et al (2019) Cancer outcomes in DCIS patients without locoregional treatment. J Natl Cancer Inst 111:952–960
Warburg O (1956) On the origin of cancer cells. Science 123:309–314
Szablewski L (2013) Expression of glucose transporters in cancers. Biochim Biophys Acta 1835:164–169
Wright EM, Loo DDF, Hirayama BA (2011) Biology of human sodium glucose transporters. Physiol Rev 91:733–794
Mueckler M, Thorens B (2013) The SLC2 (GLUT) family of membrane transporters. Mol Aspects Med 34:121–138
Hediger MA, Rhoads DB (1994) Molecular physiology of sodium-glucose cotransporters. Physiol Rev 74:993–1026
Vrhovac I, Balen Eror D, Klessen D et al (2015) Localizations of Na(+)-D-glucose cotransporters SGLT1 and SGLT2 in human kidney and of SGLT1 in human small intestine, liver, lung, and heart. Pflugers Arch 467:1881–1898
Chen J, Williams S, Ho S et al (2010) Quantitative PCR tissue expression profiling of the human SGLT2 gene and related family members. Diabetes Ther 1:57–92
Kanai Y, Lee WS, You G et al (1994) The human kidney low affinity Na+/glucose cotransporter SGLT2. Delineation of the major renal reabsorptive mechanism for D-glucose. J Clin Invest 93:397–404
Guo GF, Cai YC, Zhang B et al (2011) Overexpression of SGLT1 and EGFR in colorectal cancer showing a correlation with the prognosis. Med Oncol. https://doi.org/10.1007/s12032-010-9696-8
Hanabata Y, Nakajima Y, Morita K-I et al (2012) Coexpression of SGLT1 and EGFR is associated with tumor differentiation in oral squamous cell carcinoma. Odontology 100:156–163
Helmke BM, Reisser C, Idzkoe M et al (2004) Expression of SGLT-1 in preneoplastic and neoplastic lesions of the head and neck. Oral Oncol 40:28–35
Kobayashi M, Uematsu T, Tokura Y et al (2019) Immunohistochemical expression of sodium-dependent glucose transporter - 2 (SGLT-2) in clear cell renal carcinoma: possible prognostic implications. Int Braz J Urol 45:169–178
Ishikawa N, Oguri T, Isobe T et al (2001) SGLT gene expression in primary lung cancers and their metastatic lesions. Jpn J Cancer Res 92:874–879
Villani LA, Smith BK, Marcinko K et al (2016) The diabetes medication Canagliflozin reduces cancer cell proliferation by inhibiting mitochondrial complex-I supported respiration. Mol Metabol 5:1048–1056
Scafoglio C, Hirayama BA, Kepe V et al (2015) Functional expression of sodium-glucose transporters in cancer. Proc Natl Acad Sci USA 112:E4111–E4119
Kaji K, Nishimura N, Seki K et al (2018) Sodium glucose cotransporter 2 inhibitor canagliflozin attenuates liver cancer cell growth and angiogenic activity by inhibiting glucose uptake. Int J Cancer 142:1712–1722
Saito T, Okada S, Yamada E et al (2015) Effect of dapagliflozin on colon cancer cell [rapid communication]. Endocr J 62:1133–1137
Yamamoto L, Yamashita S, Nomiyama T et al (2021) Sodium-glucose cotransporter 2 inhibitor canagliflozin attenuates lung cancer cell proliferation in vitro. Diabetol Int 12:389–398
Zhou J, Zhu J, Yu S-J et al (2020) Sodium-glucose co-transporter-2 (SGLT-2) inhibition reduces glucose uptake to induce breast cancer cell growth arrest through AMPK/mTOR pathway. Biomed Pharmacother 132:110821
Komatsu S, Nomiyama T, Numata T et al (2020) SGLT2 inhibitor ipragliflozin attenuates breast cancer cell proliferation. Endocr J 67:99–106
Song P, Onishi A, Koepsell H, Vallon V (2016) Sodium glucose cotransporter SGLT1 as a therapeutic target in diabetes mellitus. Expert Opin Ther Targets 20:1109–1125
Liu H, Ertay A, Peng P et al (2019) SGLT1 is required for the survival of triple-negative breast cancer cells via potentiation of EGFR activity. Mol Oncol 13:1874–1886
Wang J, Ji H, Niu X et al (2020) Sodium-dependent glucose transporter 1 (SGLT1) stabled by HER2 promotes breast cancer cell proliferation by activation of the PI3K/Akt/mTOR signaling pathway in HER2+ breast cancer. Dis Markers. https://doi.org/10.1155/2020/6103542
Niu X, Ma J, Li J et al (2021) Sodium/glucose cotransporter 1-dependent metabolic alterations induce tamoxifen resistance in breast cancer by promoting macrophage M2 polarization. Cell Death Dis 12:509
Zuguchi M, Miki Y, Onodera Y et al (2012) Estrogen receptor α and β in esophageal squamous cell carcinoma. Cancer Sci 103:1348–1355
Shi M, Wang C, Ji J et al (2022) CRISPR/Cas9-mediated knockout of SGLT1 inhibits proliferation and alters metabolism of gastric cancer cells. Cell Signal 90:110192
Lai B, Xiao Y, Pu H et al (2012) Overexpression of SGLT1 is correlated with tumor development and poor prognosis of ovarian carcinoma. Arch Gynecol Obstet 285:1455–1461
Brock EJ, Ji K, Shah S et al (2019) In vitro models for studying invasive transitions of ductal carcinoma in situ. J Mammary Gland Biol Neoplasia 24:1–15
Petridis C, Brook MN, Shah V et al (2016) Genetic predisposition to ductal carcinoma in situ of the breast. Breast Cancer Res 18:22
Schuetz CS, Bonin M, Clare SE et al (2006) Progression-specific genes identified by expression profiling of matched ductal carcinomas in situ and invasive breast tumors, combining laser capture microdissection and oligonucleotide microarray analysis. Cancer Res 66:5278–5286
Shah S, Brock EJ, Jackson RM et al (2018) Downregulation of Rap1Gap: a switch from DCIS to invasive breast carcinoma via ERK/MAPK activation. Neoplasia 20:951–963
Blessing Xu, Gao, et al (2012) Sodium/glucose co-transporter 1 expression increases in human diseased prostate. J Cancer Sci Ther. https://doi.org/10.4172/1948-5956.1000159
de Miguel-Luken M-J, Chaves-Conde M, de Miguel-Luken V et al (2015) MAP17 (PDZKIP1) as a novel prognostic biomarker for laryngeal cancer. Oncotarget 6:12625–12636
Perez M, Praena-Fernandez JM, Felipe-Abrio B et al (2013) MAP17 and SGLT1 protein expression levels as prognostic markers for cervical tumor patient survival. PLoS ONE 8:e56169
Obermeier S, Hüselweh B, Tinel H et al (2000) Expression of glucose transporters in lactating human mammary gland epithelial cells. Eur J Nutr 39:194–200
Inoue T, Takemura M, Fushimi N et al (2017) Mizagliflozin, a novel selective SGLT1 inhibitor, exhibits potential in the amelioration of chronic constipation. Eur J Pharmacol 806:25–31
Shibazaki T, Tomae M, Ishikawa-Takemura Y et al (2012) KGA-2727, a novel selective inhibitor of a high-affinity sodium glucose cotransporter (SGLT1), exhibits antidiabetic efficacy in rodent models. J Pharmacol Exp Ther 342:288–296
Carmeliet P (2005) VEGF as a key mediator of angiogenesis in cancer. Oncology 69(Suppl 3):4–10
Bachelder RE, Crago A, Chung J et al (2001) Vascular endothelial growth factor is an autocrine survival factor for neuropilin-expressing breast carcinoma cells. Cancer Res 61:5736–5740
Aesoy R, Sanchez BC, Norum JH et al (2008) An autocrine VEGF/VEGFR2 and p38 signaling loop confers resistance to 4-hydroxytamoxifen in MCF-7 breast cancer cells. Mol Cancer Res 6:1630–1638
Moon SY, Lee H, Kim S et al (2021) Inhibition of STAT3 enhances sensitivity to tamoxifen in tamoxifen-resistant breast cancer cells. BMC Cancer 21:931
Lewis JS, Landers RJ, Underwood JC et al (2000) Expression of vascular endothelial growth factor by macrophages is up-regulated in poorly vascularized areas of breast carcinomas. J Pathol 192:150–158
Wang X, Bove AM, Simone G, Ma B (2020) Molecular bases of VEGFR-2-mediated physiological function and pathological role. Front Cell Dev Biol 8:599281
Takahashi T, Shibuya M (1997) The 230 kDa mature form of KDR/Flk-1 (VEGF receptor-2) activates the PLC-gamma pathway and partially induces mitotic signals in NIH3T3 fibroblasts. Oncogene 14:2079–2089
Gao HF, Chen LY, Cheng CS et al (2019) SLC5A1 promotes growth and proliferation of pancreatic carcinoma via glucose-dependent AMPK/mTOR signaling. Cancer Manag Res 11:3171–3185
Zelniker TA, Wiviott SD, Raz I et al (2019) SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet 393:31–39
Dobbins RL, Greenway FL, Chen L et al (2015) Selective sodium-dependent glucose transporter 1 inhibitors block glucose absorption and impair glucose-dependent insulinotropic peptide release. Am J Physiol Gastrointest Liver Physiol 308:G946–G954
Hui R, Pearson A, Cortes J et al (2020) Lucitanib for the treatment of HR+/HER2- metastatic breast cancer: results from the multicohort phase II FINESSE study. Clin Cancer Res 26:354–363
Acknowledgements
We thank Akiko Morohashi and Katsuhiko Ono for the excellent technical assistance.
Funding
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Contributions
ST and YO performed the experiments. EI and YM designed the study. ST, AK, and TI interpreted the clinical data. ST wrote the manuscript. TS and HS reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was approved by the Medical Ethics Committee of Tohoku University Graduate School of Medicine (Reception No. 2020–1-942).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsunokake, S., Iwabuchi, E., Miki, Y. et al. SGLT1 as an adverse prognostic factor in invasive ductal carcinoma of the breast. Breast Cancer Res Treat 201, 499–513 (2023). https://doi.org/10.1007/s10549-023-07024-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-07024-9