Abstract
Purpose
SOLAR-1 investigated alpelisib-fulvestrant (ALP + FLV) in patients with HR + /HER2-, PIK3CA-mutated advanced breast cancer and demonstrated a clinically significant increase in all-grade and grade (G) 3-4 hyperglycemia (HG) compared to placebo-fulvestrant. Given high rates of HG, a preventative protocol and identification of associated risk factors was implemented.
Methods
This single-center, retrospective study included patients receiving ALP + FLV. One week before ALP initiation, patients started an insulin-sensitizer. Patients had fasting plasma glucose (FPG) levels drawn day 8, 15, 28, then monthly. Primary outcome was incidence of G2-4 HG by day 28. Risk factors assessed included age, BMI, FPG, and HbA1c. Number of risk factors were compared between patients with and without HG.
Results
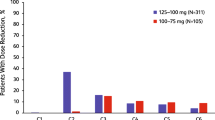
Sixteen women were included with median age of 59 years. The cohort was 69% White, 25% Black, 75% with BMI ≥ 25 kg/m2, and 50% with history of diabetes. By day 28, 9 patients (56%) had G2-4 HG, with only 3 (19%) G3 and zero G4. Patients with G2-4 HG had a median of 2 risk factors compared to only 1 if no HG (p = 0.03). 5 patients (31%) required a temporary hold of ALP and 3 (19%) required dose reduction due to HG. 13 patients permanently discontinued ALP—9 due to disease progression and 4 from an adverse event (only 1 HG).
Conclusion
Implementation of a HG prophylaxis protocol with ALP in a single-center study demonstrated fewer G3-4 HG events compared to that seen in SOLAR-1 (19% vs 36.6%). An increase in HG-associated risk factors correlated with a higher incidence of G2-4 HG.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ADA:
-
American Diabetes Association
- ALP:
-
Alpelisib
- CTCAE:
-
Common Terminology Criteria for Adverse Events
- DBOC:
-
Duke Breast Oncology Clinic
- FLV:
-
Fulvestrant
- FBG:
-
Fasting blood glucose
- FPG:
-
Fasting plasma glucose
- G:
-
Grade
- HbA1c:
-
Hemoglobin A1c
- HER2:
-
Human epidermal growth factor 2
- HG:
-
Hyperglycemia
- HR:
-
Hormone receptor
- P:
-
Placebo
- PI3K:
-
Phosphatidylinositol-3-kinase
- REDCap:
-
Research electronic data capture
- SGLT-2:
-
Sodium glucose cotransporter-2
- T2DM:
-
Type 2 diabetes mellitus
- XR:
-
Extended release
References
Siegel RL, Miller KD, Jemal A (2020) Cancer Statistics, 2020. CA Cancer J Clin 70:7–30
American Cancer Society (2019) Breast cancer facts & figures 2019–2020. American Cancer Society, Inc., Atlanta
National Comprehensive Cancer Network. Breast Cancer (Version 5.2020). https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed 26 July 2020
Network CGA (2012) Comprehensive molecular portraits of human breast tumors. Nature 490(7418):61–70. https://doi.org/10.1038/nature11412
Ellis H, Ma CX (2019) PI3K inhibitors in breast cancer therapy. Curr Oncol Rep 21(110):1–9. https://doi.org/10.1007/s11912-019-0846-7
Piqray (alpelisib) (2019) [prescribing information]. Novartis Pharmaceuticals Corporation, East Hanover
Andre F, Ciruelos E, Rubovszky G et al (2019) Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. N Engl J Med 380(20):1929–1940. https://doi.org/10.1056/NEJMoa1813904
Dullinsky PR, Hurvitz SA (2020) Mechanistic basis for PI3K inhibitor antitumor activity and adverse reactions in advanced breast cancer. Breast Cancer Res Treat 181:233–248. https://doi.org/10.1007/s10549-020-05618-1
Rugo HS, Andre F, Yamashita T et al (2020) Time course and management of key adverse events during the randomized phase III SOLAR-1 study of PI3K inhibitor alpelisib plus fulvestrant in patients with HR-positive advanced breast cancer. Ann Oncol 31(8):1001–1010. https://doi.org/10.1016/j.annonc.2020.05.001
Hopkins BD, Pauli C, Du X et al (2018) Suppression of insulin feedback enhances the efficacy of PI3K inhibitors. Nature 560:499–503. https://doi.org/10.1038/s41586-018-0343-4
American Diabetes Association (2021) Classification and diagnosis of diabetes: Standards of medical care in diabetes—2021. Diabetes Care 44(Supplement 1):S15–S33. https://doi.org/10.2337/dc21-S002
Harris PA, Taylor R, Minor BL et al (2019) The REDCap consortium: Building an international community of software platform partners. J Biomed Inform 95:103208. https://doi.org/10.1016/j.jbi.2019.103208
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG (2009) Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42(2):377–381. https://doi.org/10.1016/j.jbi.2008.08.010
Common Terminology Criteria for Adverse Events (CTCAE) Version 4.03. Published: May 28, 2009. US Department of Health and Human Services, National Institutes of Health, National Cancer Institute
Rugo HS, Lerebours F, Ciruelos E et al (2021) Aplelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): One cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncol 22(4):489–498. https://doi.org/10.1016/S1470-2045(21)00034-6
Kawahito S, Kitahata H, Oshita S (2009) Problems associated with glucose toxicity: role of hyperglycemia-induced oxidative stress. World J Gastroenterol 15:4137–4142. https://doi.org/10.3748/wjg.15.4137
American Diabetes Association (2021) Glycemic targets: Standards of medical care in diabetes—2021. Diabetes Care 44(Supplement 1):S73–S84. https://doi.org/10.2337/dc21-S006
American Diabetes Association (2021) Pharmacologic approaches to glycemic treatment: Standards of medical care in diabetes—2021. Diabetes Care 44(Supplement 1):S111–S124. https://doi.org/10.2337/dc21-S009
Study of Safety and Efficacy of Dapagliflozin + Metformin XR Versus Metformin XR in Participants With HR+, HER2-, Advanced Breast Cancer While on Treatment With Alpelisib and Fulvestrant (EPIK-B4). ClinicalTrials.gov identifier: NCT04899349. Updated December 7, 2021. Accessed 28 Jan 2022. https://clinicaltrials.gov/ct2/show/NCT04899349.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
SB, HM, JF, and KW were responsible for concept and design of the study. SB contributed to collection of the data. EP and H-JL conducted statistical analysis of the data. All authors contributed to analysis and interpretation of the data. SB, with supervision from HM, drafted the manuscript. All authors critically reviewed the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
H Moore is an active consultant for Novartis Pharmaceuticals and serves on the EPIK-B4 steering committee and is active PI of EPIK-B4 at Duke University Hospital. S Burnette, HJ Lee, and E Poehlein, J Force, and K Westbrook have no conflicts of interest to report.
Ethical approval and consent to participate
All study protocols were reviewed and approved by Duke University Health System’s Institutional Review Board.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Burnette, S.E., Poehlein, E., Lee, HJ. et al. Evaluation of alpelisib-induced hyperglycemia prophylaxis and associated risk factors in PIK3CA-mutated hormone-receptor positive, human epidermal growth factor-2 negative advanced breast cancer. Breast Cancer Res Treat 197, 369–376 (2023). https://doi.org/10.1007/s10549-022-06798-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06798-8