Abstract
Purpose
The purpose of this study was to evaluate the effect of chemotherapy and trastuzumab on invasive disease-free survival (iDFS) and overall survival (OS) in patients with human epidermal growth factor receptor 2 (HER2) positive, T1abN0 breast cancer.
Methods
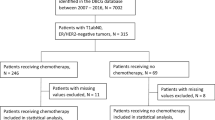
In the Danish Breast Cancer Group database, patients with HER2-positive, T1abN0 tumors diagnosed between 2007 and 2016 were identified. Cox proportional hazards analysis was performed to analyze the association between adjuvant chemotherapy and trastuzumab and iDFS and OS.
Results
Of 605 patients included in the analyses, 465 patients received chemotherapy and trastuzumab and 140 patients did not. Chemotherapy and trastuzumab did not improve iDFS or OS significantly in adjusted analyses. 5-year iDFS was 92.3% vs. 89.9%, Hazard ratio (HR) 1.01 (p = 0.98), and 5-year OS was 97.4% vs. 94.3%, HR 0.60 (p = 0.15), chemotherapy and trastuzumab vs. no chemotherapy/trastuzumab. In unadjusted analyses, significant treatment benefit on OS was found in patients with T1b tumors. The largest absolute treatment benefits were found in patients with T1b tumors and estrogen receptor (ER) negative tumors, respectively, whereas treatment effects in patients with T1a tumors and ER-positive tumors, respectively, were limited.
Conclusion
Adjuvant chemotherapy and trastuzumab did not improve OS or iDFS significantly in patients with HER2-positive, T1abN0 breast cancers in adjusted analyses. In unadjusted analyses, significant OS benefit was found in patients with T1b tumors. The largest absolute benefit was observed in patients with T1b tumors and ER-negative tumors, respectively, whereas the effect was limited in patients with T1a tumors and ER-positive tumors, respectively.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to Danish legislation. Access to data from the Danish Breast Cancer Group database can be applied through Danish Clinical Registries (RKKP).
References
The Danish Breast Cancer Group (2020). Kvalitetsindikatorrapport for Brystkræft [Internet]. Available from: https://dbcg.dk/images/PDF/Rapporter/DBCG_årsrapport_2020_Publiceret_FINAL.pdf
Gonzalez-Angulo AM, Litton JK, Broglio KR, Meric-Bernstam F, Rakkhit R, Cardoso F et al (2009) High risk of recurrence for patients with breast cancer who have human epidermal growth factor receptor 2-positive, node-negative tumors 1 cm or smaller. J Clin Oncol 27(34):5700–5706
Harbeck N, Penault-Llorca F, Cortes J, Gnant M, Houssami N, Poortmans P et al (2019) Breast cancer. Nat Rev Dis Prim 5(1):66
Slamon D, Eiermann W, Robert N, Pienkowski T, Martin M, Press M et al (2011) Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med 365(14):1273–1283
Cameron D, Piccart-Gebhart MJ, Gelber RD, Procter M, Goldhirsch A, de Azambuja E et al (2017) 11 years’ follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the HERceptin Adjuvant (HERA) trial. Lancet 389(10075):1195–1205
Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Davidson NE et al (2005) Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 353(16):1673–1684
Bradley R, Braybrooke J, Gray R, Hills R, Liu Z, Peto R et al (2021) Trastuzumab for early-stage, HER2-positive breast cancer: a meta-analysis of 13,864 women in seven randomised trials. Lancet Oncol 22(8):1139–1150
Dumas A, Vaz Luis I, Bovagnet T, El Mouhebb M, Di Meglio A, Pinto S et al (2020) Impact of breast cancer treatment on employment: results of a multicenter prospective cohort study (CANTO). J Clin Oncol 38(7):734–743
National comprehensive cancer network clinical practice guidelines oncology [Internet]. (2021) [cited 2021 Nov 9]. Available from: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1419
Burstein HJ, Curigliano G, Thürlimann B, Weber WP, Poortmans P, Regan MM et al (2021) Customizing local and systemic therapies for women with early breast cancer: the St. Gallen international consensus guidelines for treatment of early breast cancer 2021. Ann Oncol 32(10):1216–1235
Jensen M-B, Laenkholm A-V, Offersen BV, Christiansen P, Kroman N, Mouridsen HT et al (2018) The clinical database and implementation of treatment guidelines by the danish breast cancer cooperative group in 2007–2016. Acta Oncol 57(1):13–18. https://doi.org/10.1080/0284186X.2017.1404638
Hudis CA, Barlow WE, Costantino JP, Gray RJ, Pritchard KI, Chapman JAW et al (2007) Proposal for standardized definitions for efficacy end points in adjuvant breast cancer trials: the STEEP system. J Clin Oncol 25(15):2127–2132
Wolff AC, Hammond MEH, Schwartz JN, Hagerty KL, Allred DC, Cote RJ et al (2007) American society of clinical oncology/college of American pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25(1):118–145
Wolff AC, Hammond MEH, Hicks DG, Dowsett M, McShane LM, Allison KH et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American society of clinical oncology/college of American pathologists clinical practice guideline update. J Clin Oncol 31(31):3997–4013
Danish Breast Cancer Group (2021). Patologiprocedurer og molekylærpatologiske analyser ved brystkræft
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19(5):403–410
Danish Breast Cancer Group (2019). Medicinsk behandling [Internet]. [cited 2022 Feb 21]. Available from: https://dbcg.dk/images/PDF/Retningslinier/Kap_6_Medicinsk_behandling-21.02.2019.pdf
Vaz-Luis I, Ottesen RA, Hughes ME, Mamet R, Burstein HJ, Edge SB et al (2014) Outcomes by tumor subtype and treatment pattern in women with small, node-negative breast cancer: a multi-institutional study. J Clin Oncol 32(20):2142–2150
van Ramshorst MS, van der Heiden M, Dackus GMHE, Linn SC, Sonke GS (2016) The effect of trastuzumab-based chemotherapy in small node-negative HER2-positive breast cancer. Breast Cancer Res Treat 158(2):361–371
He X, Ji J, Tian M, Esteva FJ, Hortobagyi GN, Yeung SCJ (2019) Long-term survival analysis of adjuvant chemotherapy with or without trastuzumab in patients with T1, node-negative HER2-positive breast cancer. Clin Cancer Res 25(24):7388–7395
Rodrigues MJ, Peron J, Frénel JS, Vano YA, Wassermann J, Debled M et al (2013) Benefit of adjuvant trastuzumab-based chemotherapy in T1ab node-negative HER2-overexpressing breast carcinomas: a multicenter retrospective series. Ann Oncol 24(4):916–924
de Nonneville A, Gonçalves A, Zemmour C, Classe JM, Cohen M, Lambaudie E et al (2017) Benefit of adjuvant chemotherapy with or without trastuzumab in pT1ab node-negative human epidermal growth factor receptor 2-positive breast carcinomas: results of a national multi-institutional study. Breast Cancer Res Treat 162(2):307–316
Ali S, Hendry J, Le D, Mondal PK, Sami A, Chalchal H et al (2022) Efficacy of adjuvant trastuzumab in women with HER2-positive T1a or bN0M0 breast cancer: a population-based cohort study. Sci Rep 12(1):1068
McArthur HL, Mahoney KM, Morris PG, Patil S, Jacks LM, Howard J et al (2011) Adjuvant trastuzumab with chemotherapy is effective in women with small, node-negative, HER2-positive breast cancer. Cancer 117(24):5461–5468
Gori S, Inno A, Fiorio E, Foglietta J, Ferro A, Gulisano M et al (2015) The promher study: an observational Italian study on adjuvant therapy for HER2-positive, pT1a-b pN0 breast cancer. PLoS ONE 10(9):e0136731
Tolaney SM, Guo H, Pernas S, Barry WT, Dillon DA, Ritterhouse L et al (2019) Seven-year follow-up analysis of adjuvant paclitaxel and trastuzumab trial for node-negative, human epidermal growth factor receptor 2–positive breast cancer. J Clin Oncol 37(22):1868–1875
Thygesen LC, Ersbøll AK (2014) When the entire population is the sample: strengths and limitations in register-based epidemiology. Eur J Epidemiol 29(8):551–558
Colleoni M, Sun Z, Price KN, Karlsson P, Forbes JF, Thürlimann B et al (2016) Annual hazard rates of recurrence for breast cancer during 24 years of follow-up: results from the international breast cancer study group trials I to V. J Clin Oncol 34(9):927–935
Funding
This study is part of a PhD-project supported by the Danish Cancer Society, Denmark (R246-A14574-19-S73); Interne Forskningsmidler, Herlev and Gentofte Hospital, Denmark; Tømrermester Jørgen Holm og Hustru Elisa F. Hansens Mindelegat, Denmark (20006-1846); Dansk Kræftforskningsfond, Denmark; Region Sjællands Sundhedsvidenskabelige Forskningsfond, Denmark (R19A283B198); Dansk Brystkirurgisk Selskabs legat, Denmark; Helsefonden, Denmark (20-B-0003). The funding sources had no role in the study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. All authors were involved in material preparation, data collection, analysis, or interpretation of the analyses. The first draft of the manuscript was written by CH and TT. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Ann Søegaard Knoop reports personal fees from Novartis, Roche, Merck, Daiichi Sankyo, Gilead Sciences, Pfizer, Eli Lilly. Furthermore, she has been national or site investigator for Daiichi Sankyo and Sanofi. Tove Tvedskov reports speaker honorariums from Roche, MSD and Pfizer. Anne-Vibeke Lænkholm reports institutional research grants from Novartis and Astra Zeneca (AZ). Anne-Vibeke Lænkholm reports personal fees for participation in advisory boards, Novartis and AZ and for a research collaboration with Veracyte. No other potential conflicts of interest were reported.
Ethical approval
This study was approved by the Regional Research Ethics Committee of the Capital Region of Denmark (H-18045702) and by the Regional Data Protection Agency (P-2019-124).
Informed consent
Exception for obtaining informed consent was given by the Regional Research Ethics Committee of the Capital Region of Denmark as the present study implied no health-related risks or discomfort for the involved patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hassing, C.M.S., Mejdahl, M.K., Lænkholm, AV. et al. Benefit of adjuvant chemotherapy and trastuzumab in patients with HER2-positive, node-negative breast tumors ≤ 10 mm: a nationwide study. Breast Cancer Res Treat 196, 197–206 (2022). https://doi.org/10.1007/s10549-022-06724-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06724-y