Abstract
Purpose
Older cancer patients are susceptible to long-term effects of chemotherapy, including cancer-related cognitive decline and impairments to quality of life. Taxane-based chemotherapies are associated with physical declines among older women and may negatively impact cognitive performance. We sought to examine whether changes in objective and subjective measures of cognitive performance and well-being differ among older breast cancer survivors as a function of taxane-based chemotherapy treatment regimens.
Methods
Individual-level data were pooled and harmonized from two large prospective studies of older (greater than 60 years) breast cancer survivors. Assessments were conducted prior to systemic therapy and up to 36 months after. Cognitive performance was assessed with objective (working memory, processing speed, and executive functions) and subjective tests and physical, emotional, and functional well-being were also assessed.
Results
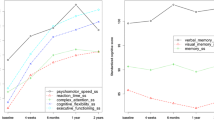
One hundred and sixty-seven (M age = 67.3 years) women with 116 receiving chemotherapy with taxanes and 51 without taxanes contributed data. Declines in subjective cognition for both groups were significant between pre-treatment and 12-month follow-up. Significant improvements were seen on a measure of objective cognition (working memory) from 12 to 36 months. Measures of well-being improved from prior to systemic therapy to 12 months. Longitudinal changes across all measures did not vary as a function of receipt of taxane-based treatment.
Conclusion
Older women who received treatment with taxanes did not have greater declines in cognitive performance or well-being than women receiving other chemotherapy regimens. Despite older cancer survivors being at greater risk for negative outcomes, treatment with taxane-based chemotherapies does not appear to exacerbate these health consequences.



Similar content being viewed by others
Data availability
Data are made available upon request.
Code availability
NA.
References
Plevritis SK, Munoz D, Kurian AW, Stout NK, Alagoz O, Near AM et al (2018) Association of screening and treatment with breast cancer mortality by molecular subtype in US women, 2000–2012. JAMA 319:154–164
Bluethmann S, Mariotto A, Rowland J (2016) Anticipating the “Silver Tsunami”: Prevalence trajectories and comorbidity burden among older cancer survivors in the United States. Cancer Epidemiol Biomark Prev 25:1029–1036
Rowland JH, Bellizzi KM (2014) Cancer survivorship issues: life after treatment and implications for an aging population. J Clin Oncol 32:2662–2668
Mandelblatt JS, Zhai W, Ahn J, Small BJ, Ahles TA, Carroll JE et al (2020) Symptom burden among older breast cancer survivors: the thinking and living with cancer (TLC) study. Cancer 126:1183–1192
Mandelblatt JS, Small BJ, Luta G, Hurria A, Jim H, McDonald BC et al (2018) Cancer-related cognitive outcomes among older breast cancer survivors in the thinking and living with cancer study. J Clin Oncol. https://doi.org/10.1200/JCO.18.00140
Ahles TA, Root JC, Ryan EL (2012) Cancer- and cancer treatment-associated cognitive change: an update on the state of the science. J Clin Oncol 30:3675–3686
Janelsins MC, Heckler CE, Peppone LJ, Kamen C, Mustian KM, Mohile SG et al (2017) Cognitive complaints in survivors of breast cancer after chemotherapy compared with age-matched controls: an analysis from a nationwide, multicenter. Prospect Longitud Study J Clin Oncol 35:506–514
Lange M, Rigal O, Clarisse B, Giffard B, Sevin E, Barillet M et al (2014) Cognitive dysfunctions in elderly cancer patients: a new challenge for oncologists. Cancer Treat Rev 40:810–817
Ahles TA, Li Y, McDonald BC, Schwartz GN, Kaufman PA, Tsongalis GJ et al (2014) Longitudinal assessment of cognitive changes associated with adjuvant treatment for breast cancer: the impact of APOE and smoking. Psychooncology 23:1382–1390
Mandelblatt JS, Stern RA, Luta G, McGuckin M, Clapp JD, Hurria A et al (2014) Cognitive impairment in older patients with breast cancer before systemic therapy: is there an interaction between cancer and comorbidity? J Clin Oncol 32:1909–1918
Ahles TA, Saykin AJ, McDonald BC, Li Y, Furstenberg CT, Hanscom BS et al (2010) Longitudinal assessment of cognitive changes associated with adjuvant treatment for breast cancer: impact of age and cognitive reserve. JClinOncol 28:4434–4440
Wagner LI, Gray RJ, Sparano JA, Whelan TJ, Garcia SF, Yanez B et al (2020) Patient-reported cognitive impairment among women with early breast cancer randomly assigned to endocrine therapy alone versus chemoendocrine therapy: results from TAILORx. J Clin Oncol 38:1875–1886
Denduluri N, Somerfield MR, Eisen A, Holloway JN, Hurria A, King TA et al (2016) Selection of optimal adjuvant chemotherapy regimens for human epidermal growth factor receptor 2 (HER2) -negative and adjuvant targeted therapy for HER2-positive breast cancers: an American society of clinical oncology guideline adaptation of the cancer care Ontario clinical practice guideline. J Clin Oncol 34:2416–2427
Lange M, Heutte N, Rigal O, Noal S, Kurtz JE, Levy C et al (2016) Decline in cognitive function in older adults with early-stage breast cancer after adjuvant treatment. Oncologist 21:1337–1348
Hershman DL, Till C, Wright JD, Awad D, Ramsey SD, Barlow WE et al (2016) Comorbidities and risk of chemotherapy-induced peripheral neuropathy among participants 65 years or older in southwest oncology group clinical trials. J Clin Oncol 34:3014–3022
da Costa R, Passos GF, Quintão NLM, Fernandes ES, Maia J, Campos MM et al (2020) Taxane-induced neurotoxicity: pathophysiology and therapeutic perspectives. Br J Pharmacol 177:3127–3146
Lange M, Giffard B, Noal S, Rigal O, Kurtz JE, Heutte N et al (2014) Baseline cognitive functions among elderly patients with localised breast cancer. Eur J Cancer 50:2181–2189
Wechsler D (1999) Wechsler adult intelligence scale-III (WAIS-III). The Psychological Corporation, San Antonio, TX
Reitan RM, Wolfson D (1993) The Halstead-Reitan neuropsychological test battery: theory and clinical interpretation. Neuropsychology Press, Tucson, AZ
Godefroy O. Réflexion pour l’évaluation des Fonction sEXécutives (2008) Executive Functions and neurological and psychiatric diseases: assessment in clinical practice. Marseille, France: France:Solal.
Wechsler D (1997) WAIS-III administration and scoring manual. Psychological Corporation, San Antonio, TX
Tombaugh T, McIntyre N (1992) The mini-mental state examination: a comprehensive review. J Am Geriatr Soc 40:922–935
Stern RA, White T (2003) Neuropsychological assessment battery (NAB). Psychological Assessment Resources, Lutz, FL
Wagner LI, Sweet J, Butt Z, Lai J-s, Cella D (2009) Measuring patient self-reported cognitive function: development of the functional assessment of cancer therapy-cognitive function instrument. J Support Oncol 7:W32–W9
Joly F, Lange M, Rigal O, Correia H, Giffard B, Beaumont JL et al (2012) French version of the functional assessment of cancer therapy-cognitive function (FACT-Cog) version 3. Support Care Cancer 20:3297–3305
Brady MJ, Cella DF, Mo F, Bonomi AE, Tulsky DS, Lloyd SR et al (1997) Reliability and validity of the functional assessment of cancer therapy-breast quality-of-life instrument. J Clin Oncol 15:974–986
Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW (1963) Studies of illness in the aged. The index of Adl: a standardized measure of biological and psychosocial function. JAMA 185:914–919
Hoffman L (2015) Longitudinal analysis: modeling within-person fluctuation and change. Routledge, New York, NY
Bell ML, Dhillon HM, Bray VJ, Vardy JL (2018) Important differences and meaningful changes for the functional assessment of cancer therapy-cognitive function (FACT-Cog). J Patient-Rep Outcomes 2:48
Myers JS, Wick JA, Klemp J (2015) Potential factors associated with perceived cognitive impairment in breast cancer survivors. Support Care Cancer 23:3219–3228
Liou KT, Ahles TA, Garland SN, Li QS, Bao T, Li Y et al (2019) The relationship between insomnia and cognitive impairment in breast cancer survivors. JNCI Cancer Spectr. https://doi.org/10.1093/jncics/pkz041
Jansen CE, Cooper BA, Dodd MJ, Miaskowski CA (2011) A prospective longitudinal study of chemotherapy-induced cognitive changes in breast cancer patients. Support Care Cancer 19:1647–1656
Cerulla N, Arcusa À, Navarro JB, Garolera M, Enero C, Chico G et al (2017) Role of taxanes in chemotherapy-related cognitive impairment: a prospective longitudinal study. Breast Cancer Res Treat 164:179–187
Kobayashi LC, Cohen HJ, Zhai W, Zhou X, Small BJ, Luta G et al (2020) Cognitive function prior to systemic therapy and subsequent well-being in older breast cancer survivors: longitudinal findings from the thinking and living with cancer study. Psychooncology 29:1051–1059
Hurria A, Soto-Perez-de-Celis E, Allred JB, Cohen HJ, Arsenyan A, Ballman K et al (2019) Functional decline and resilience in older women receiving adjuvant chemotherapy for breast cancer. J Am Geriatr Soc 67:920–927
McArdle JJ, Grimm KJ, Hamagami F, Bowles RP, Meredith W (2009) Modeling life-span growth curves of cognition using longitudinal data with multiple samples and changing scales of measurement. Psychol Methods 14:126–149
Duggan EC, Piccinin AM, Clouston S, Koval AV, Robitaille A, Zammit AR et al (2019) A multi-study coordinated meta-analysis of pulmonary function and cognition in aging. J Gerontol A Biol Sci Med Sci 74:1793–1804
Gross AL, Sherva R, Mukherjee S, Newhouse S, Kauwe JS, Munsie LM et al (2014) Calibrating longitudinal cognition in Alzheimer’s disease across diverse test batteries and datasets. Neuroepidemiology 43:194–205
Rabin LA, Smart CM, Crane PK, Amariglio RE, Berman LM, Boada M et al (2015) Subjective cognitive decline in older adults: an overview of self-report measures used across 19 international research studies. J Alzheimer’s Dis 48(Suppl 1):S63-86
Wefel JS, Kesler SR, Noll KR, Schagen SB (2015) Clinical characteristics, pathophysiology, and management of noncentral nervous system cancer-related cognitive impairment in adults. CA 65:123–38
Funding
This research was supported by the National Cancer Institute at the National Institutes of Health Grants R01CA129769 and R35CA197289 to JM. This work was supported in part by the National Institute of Aging at the National Institutes of Health Grant R01 AG068193 to JM. As a member of the Cancer and Aging Research Group (CARG), JM’s work on this project was also supported in part by grant R33AG059206 (MPI: W Dale, S Mohile, H Klepin) from the National Institute of Aging at the National Institutes of Health. This study was also supported in part by the National Cancer Institute at the National Institutes of Health Grant P30CA51008 to Georgetown Lombardi Comprehensive Cancer Center for support of the Biostatistics and Bioinformatics Resource and the Non-Therapeutic Shared Resource. The work of AJS was supported in part by the National Institute of Aging at the National Institutes of Health Grants P30AG10133, R01AG19771 and R01LM01136. TAA was supported in part by National Cancer Institute at the National Institutes of Health Grants R01CA172119, U54 CA137788, and P30CA008748. JR was supported in part by National Cancer Institute at the National Institutes of Health grant R01CA172119. BCM was supported in part by National Cancer Institute at the National Institutes of Health Grant R01CA244673. KVD was supported in part by the National Cancer Institute at the National Institutes of Health Grant K08CA241337. The work of JEC was supported in part by the American Cancer Society Research Scholars Grant 128660-RSG-15-187-01-PCSM and the National Cancer Institute at the National Institutes of Health Grant R01CA237535. HJC was supported in part by the National Institute of Aging at the National Institutes of Health Grant P30AG028716 for the Duke Pepper Center. SKP was supported in part by the American Cancer Society Research Scholars Grant RSG-17-023-01-CPPB. The CogAge study was supported by a National Grant (Inca Programme Hospitalier de Recherche Clinique, Grant APN 2008 n°06-08) and Sanofi.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to report.
Ethical approval
Ethical approval was obtained from relevant organizations.
Consent to participate
All participants provided informed consent.
Consent for publication
All authors consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Brent J. Small, Marie Lange, Florence Joly, and Jeanne Mandelblatt were the writing committee for this manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Small, B.J., Lange, M., Zhai, W. et al. Impact of taxane-based chemotherapy among older women with breast cancer on cognition and quality of life: a longitudinal pooled analysis. Breast Cancer Res Treat 191, 459–469 (2022). https://doi.org/10.1007/s10549-021-06455-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-021-06455-6