Abstract
Purpose
A positive resection margin after breast conserving surgery (BCS) is the most important risk factor for tumor recurrence. In 2012, Seoul National University Hospital (SNUH) breast surgery team developed a nomogram for predicting positive resection margins before BCS to provide individual surgical plans that could reduce local recurrence without increasing re-excision rates. The purpose of this study was to validate this nomogram using an external cohort and to test if addition of surgeon-related factor could improve its use as a predictive model.
Methods
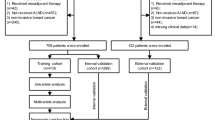
A total of 419 patients with breast cancer who underwent BCS from January to December 2018 were retrospectively reviewed. Using the SNUH BCS nomogram, risk score for positive resection margins was calculated for 343 patients. The predictive accuracy of the nomogram was assessed, and multivariable logistic regression analyses were performed to evaluate the nomogram’s predictive variables.
Results
The positive resection margin rate of the current external validation cohort was 13.5% (46 out of 343), compared to 14.6% (151 out of 1034) of the original study. The discrimination power of the SNUH BCS nomogram as measure by area under the receiver operating characteristics curve (AUC) was 0.656 [95% confidence interval (CI) 0.576–0.735]. This result is lower than expected value of 0.823 [95% CI 0.785–0.862], the AUC of the original study. Multivariable logistic regression analysis showed that, among the five nomogram variables, presence of tumor size discrepancy greater than 0.5 cm between MRI and ultrasonography (OR 2.445, p = 0.019) and presence of ductal carcinoma in situ on needle biopsy (OR 2.066, p = 0.048) were significantly associated with positive resection margins. Finally, the nomogram score was re-calculated by adding each surgeon’s resection margin positive rate as odds ratio and the AUC was increased to 0.733.
Conclusions
Validation of the SNUH BCS nomogram was not successful in the current study as much as its original publication. However, we could improve its predictive power by including surgeon-related factor. Before applying a published nomogram as a preoperative predictive model, we suggest each institution to validate the model and adjust it with surgeon-related factor. Addition of new factors to currently available nomograms holds promise for improving its applicability for breast cancer patients at the actual clinical level.



Similar content being viewed by others
References
Fisher B, Anderson S, Bryant J et al (2002) Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med 347:1233–1241. https://doi.org/10.1056/NEJMoa022152
Veronesi U, Cascinelli N, Mariani L et al (2002) Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med 347:1227–1232. https://doi.org/10.1056/NEJMoa020989
Park CC, Mitsumori M, Nixon A et al (2000) Outcome at 8 years after breast-conserving surgery and radiation therapy for invasive breast cancer: influence of margin status and systemic therapy on local recurrence. J Clin Oncol 18:1668–1675. https://doi.org/10.1200/JCO.2000.18.8.1668
Houssami N, MacAskill P, Marinovich ML et al (2010) Meta-analysis of the impact of surgical margins on local recurrence in women with early-stage invasive breast cancer treated with breast-conserving therapy. Eur J Cancer 46:3219–3232. https://doi.org/10.1016/j.ejca.2010.07.043
Zavagno G, Goldin E, Mencarelli R et al (2008) Role of resection margins in patients treated with breast conservation surgery. Cancer 112:1923–1931. https://doi.org/10.1002/cncr.23383
Patten CR, Walsh K, Sarantou T et al (2017) Changes in margin re-excision rates: Experience incorporating the “no ink on tumor” guideline into practice. J Surg Oncol 116:1040–1045. https://doi.org/10.1002/jso.24770
Van Zee KJ, Manasseh DME, Bevilacqua JLB et al (2003) A nomogram for predicting the likelihood of additional nodal metastases in breast cancer patients with a positive sentinel node biopsy. Ann Surg Oncol 10:1140–1151. https://doi.org/10.1245/ASO.2003.03.015
Poirier E, Sideris L, Dube P et al (2008) Analysis of clinical applicability of the breast cancer nomogram for positive sentinel lymph node: the Canadian experience. Ann Surg Oncol 15:2562–2567
Alran S, De Rycke Y, Fourchotte V et al (2007) Validation and limitations of use of a breast cancer nomogram predicting the likelihood of non-sentinel node involvement after positive sentinel node biopsy. Ann Surg Oncol 14:2195–2201. https://doi.org/10.1245/s10434-006-9331-2
Lambert LA, Ayers GD, Hwang RF et al (2006) Validation of a breast cancer nomogram for predicting nonsentinel lymph node metastases after a positive sentinel node biopsy. Ann Surg Oncol 13:310–320
Shin HC, Han W, Moon HG et al (2012) Nomogram for predicting positive resection margins after breast-conserving surgery. Breast Cancer Res Treat 134:1115–1123. https://doi.org/10.1007/s10549-012-2124-3
Agostinho JL, Zhao X, Sun W et al (2015) Prediction of positive margins following breast conserving surgery. Breast 24:46–50. https://doi.org/10.1016/j.breast.2014.11.001
Pleijhuis RG, Kwast ABG, Jansen L et al (2013) A validated web-based nomogram for predicting positive surgical margins following breast-conserving surgery as a preoperative tool for clinical decision-making. Breast 22:773–779. https://doi.org/10.1016/j.breast.2013.01.010
Rouzier R, Pusztai L, Garbay JR et al (2006) Development and validation of nomograms for predicting residual tumor size and the probability of successful conservative surgery with neoadjuvant chemotherapy for breast cancer. Cancer 107:1459–1466. https://doi.org/10.1002/cncr.22177
NCCN Guidelines Insights: Breast Cancer, Version 3.2018 in: Journal of the National Comprehensive Cancer Network Volume 17 Issue 2 (2019). https://jnccn.org/view/journals/jnccn/17/2/article-p118.xml?utm_source=TrendMD&utm_medium=cpc&utm_campaign=JNCCN_TrendMD_1. Accessed 10 Jun 2020
Akobeng AK (2007) Understanding diagnostic tests 3: receiver operating characteristic curves. Acta Paediatr Int J Paediatr 96:644–647. https://doi.org/10.1111/j.1651-2227.2006.00178.x
Fischer JE, Bachmann LM, Jaeschke R (2003) A readers’ guide to the interpretation of diagnostic test properties: clinical example of sepsis. Intensive Care Med 29:1043–1051
Peintinger F (2011) Clinical use of nomograms for breast cancer. J Surg Oncol 103:745
Klar M, Jochmann A, Foeldi M et al (2008) The MSKCC nomogram for prediction the likelihood of non-sentinel node involvement in a German breast cancer population. Breast Cancer Res Treat 112:523–531. https://doi.org/10.1007/s10549-007-9884-1
Pal A, Provenzano E, Duffy SW et al (2008) A model for predicting non-sentinel lymph node metastatic disease when the sentinel lymph node is positive. Br J Surg 95:302–309. https://doi.org/10.1002/bjs.5943
Soni NK, Carmalt HL, Gillett DJ, Spillane AJ (2005) Evaluation of a breast cancer nomogram for prediction of non-sentinel lymph node positivity. Eur J Surg Oncol 31:958–964. https://doi.org/10.1016/j.ejso.2005.04.011
Rahman GA (2011) Editorial: breast conserving therapy: a surgical technique where little can mean more. J Surg Tech Case Rep 3:1–4
Sarin R, Somsekhar S, Kumar R et al (2018) Practical consensus recommendations for tumor margins and breast conservative surgery. South Asian J Cancer 7:72. https://doi.org/10.4103/sajc.sajc_105_18
Azu M, Abrahamse P, Katz SJ et al (2010) What is an adequate margin for breast-conserving surgery? Surgeon attitudes and correlates. Ann Surg Oncol 17:558–563. https://doi.org/10.1245/s10434-009-0765-1
Taghian A, Mohiuddin M, Jagsi R et al (2005) Current perceptions regarding surgical margin status after breast-conserving therapy: results of a survey. Ann Surg 241:629–639
Fisher B (2002) Twenty-year follow-up of a randomized trial comparing total for the treatment of invasive breast cancer. English J 347:1233–1241. https://doi.org/10.1056/NEJMoa020128
Arriagada R, Lê MG, Rochard F, Contesso G (1996) Conservative treatment versus mastectomy in early breast cancer: patterns of failure with 15 years of follow-up data. J Clin Oncol 14:1558–1564. https://doi.org/10.1200/JCO.1996.14.5.1558
Moran MS, Schnitt SJ, Giuliano AE et al (2014) Society of surgical oncology-American society for radiation oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages i and II invasive breast cancer. Int J Radiat Oncol Biol Phys 88:553–564. https://doi.org/10.1016/j.ijrobp.2013.11.012
Morrow M, Van Zee KJ, Solin LJ et al (2016) Society of surgical oncology-American society for radiation oncology-American society of clinical oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in ductal carcinoma in situ. J Clin Oncol 34:4040–4046. https://doi.org/10.1200/JCO.2016.68.3573
Association of Breast Surgery at Baso (2009) (2009) Surgical guidelines for the management of breast cancer. Eur J Surg Oncol 35(Suppl 1):1–22. https://doi.org/10.1016/j.ejso.2009.01.008
Bhutiani N, Mercer MK, Bachman KC et al (2018) Evaluating the effect of margin consensus guideline publication on operative patterns and financial impact of breast cancer operation. J Am Coll Surg 227:6–11. https://doi.org/10.1016/j.jamcollsurg.2018.01.050
Schulman AM, Mirrielees JA, Leverson G et al (2017) Reexcision surgery for breast cancer: an analysis of the American Society of Breast Surgeons (ASBrS) MasterySM database following the SSO-ASTRO “no ink on tumor” guidelines. Ann Surg Oncol 24:52–58. https://doi.org/10.1245/s10434-016-5516-5
Brouwer de Koning SG, Vrancken Peeters MJTFD, Jóźwiak K et al (2018) Tumor resection margin definitions in breast-conserving surgery: systematic review and meta-analysis of the current literature. Clin Breast Cancer 18:e595–e600
Acknowledgement
We greatly appreciate the active support of Division of Statistics in Medical Research Collaborating Center at Seoul National University Bundang Hospital for statistical analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has a financial interest or commercial associations to declare in relation to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jung, JJ., Kang, E., Kim, EK. et al. External validation and modification of nomogram for predicting positive resection margins before breast conserving surgery. Breast Cancer Res Treat 183, 373–380 (2020). https://doi.org/10.1007/s10549-020-05779-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05779-z