Abstract
Background
The role of G-protein-coupled estrogen receptor 1 (GPER-1) in the development of tamoxifen resistance in breast cancer is a highly controversial issue. The aim of this study was to determine the expression of GPER-1 in the clinical routine under conditions of endocrine treatment.
Patients and methods
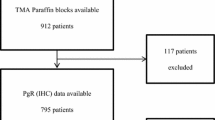
GPER-1 expression was analyzed in 442 patients with primary invasive breast cancer. GPER-1 score of > 3 was determined as positive. Expression data were correlated with clinical and pathological characteristics and patient survival.
Results
GPER-1 expression was observed in 352 (80.9%) cases, and positively correlated with estrogen and progesterone receptor status (p = 0.0001). GPER-1 positivity was associated with an increased grade of differentiation (p = 0.0001) and with a low level of Ki-67 expression (p = 0.0001). High GPER-1 expression was associated with a decreased level upon systemic treatment (p = 0.011). In the whole cohort, GPER-1 expression was associated with prolonged disease-free survival (DFS). DFS between tamoxifen- and aromatase inhibitor-treated GPER-1-positive patients was similar (p = 0.090). Notably, after matching the analysis for the most important prognostic factors, DFS for tamoxifen-treated GPER-1-positive patients was 69.1%, which is a percentage that is significantly lower compared to DFS for GPER-1-positive patients treated with aromatase inhibitors (92.7%) (p = 0.005).
Conclusion
GPER-1 expression is a favorable prognostic factor in breast cancer patients. Its predictive role for poor benefit form tamoxifen treatment should be investigated in further studies.



Similar content being viewed by others
References
Molina L, Figueroa CD, Bhoola KD, Ehrenfeld P (2017) GPER-1/GPR30 a novel estrogen receptor sited in the cell membrane: therapeutic coupling to breast cancer. Expert Opin Ther Targets 21:755–766
Ignatov A, Ignatov T, Weissenborn C et al (2011) G-protein-coupled estrogen receptor GPR30 and tamoxifen resistance in breast cancer. Breast Cancer Res Treat 128:457–466
Sjostrom M, Hartman L, Grabau D et al (2014) Lack of G protein-coupled estrogen receptor (GPER) in the plasma membrane is associated with excellent long-term prognosis in breast cancer. Breast Cancer Res Treat 145:61–71
Ignatov A, Ignatov T, Roessner A, Costa SD, Kalinski T (2010) Role of GPR30 in the mechanisms of tamoxifen resistance in breast cancer MCF-7 cells. Breast Cancer Res Treat 123:87–96
Mo Z, Liu M, Yang F et al (2013) GPR30 as an initiator of tamoxifen resistance in hormone-dependent breast cancer. Breast Cancer Res 15:R114
Ignatov T, Weissenborn C, Poehlmann A et al (2013) GPER-1 expression decreases during breast cancer tumorigenesis. Cancer Invest 31:309–315
Weissenborn C, Ignatov T, Nass N et al (2017) GPER promoter methylation controls GPER expression in breast cancer patients. Cancer Invest 35:100–107
von Elm E, Altman DG, Egger M et al (2007) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 370:1453–1457
Broselid S, Cheng B, Sjostrom M et al (2013) G protein-coupled estrogen receptor is apoptotic and correlates with increased distant disease-free survival of estrogen receptor-positive breast cancer patients. Clin Cancer Res 19:1681–1692
Samartzis EP, Noske A, Meisel A, Varga Z, Fink D, Imesch P (2014) The G protein-coupled estrogen receptor (GPER) is expressed in two different subcellular localizations reflecting distinct tumor properties in breast cancer. PLoS ONE 9:e83296
Ariazi EA, Brailoiu E, Yerrum S et al (2010) The G protein-coupled receptor GPR30 inhibits proliferation of estrogen receptor-positive breast cancer cells. Cancer Res 70:1184–1194
Ignatov T, Modl S, Thulig M et al (2013) GPER-1 acts as a tumor suppressor in ovarian cancer. J Ovarian Res 6:51
Liu Q, Chen Z, Jiang G et al (2017) Epigenetic down regulation of G protein-coupled estrogen receptor (GPER) functions as a tumor suppressor in colorectal cancer. Mol Cancer 16:87
Weissenborn C, Ignatov T, Ochel HJ et al (2014) GPER functions as a tumor suppressor in triple-negative breast cancer cells. J Cancer Res Clin Oncol 140:713–723
Weissenborn C, Ignatov T, Poehlmann A et al (2014) GPER functions as a tumor suppressor in MCF-7 and SK-BR-3 breast cancer cells. J Cancer Res Clin Oncol 140:663–671
Chan QK, Lam HM, Ng CF et al (2010) Activation of GPR30 inhibits the growth of prostate cancer cells through sustained activation of Erk1/2, c-jun/c-fos-dependent upregulation of p21, and induction of G(2) cell-cycle arrest. Cell Death Differ 17:1511–1523
Ribeiro MPC, Santos AE, Custodio JBA (2017) The activation of the G protein-coupled estrogen receptor (GPER) inhibits the proliferation of mouse melanoma K1735-M2 cells. Chem Biol Interact 277:176–184
Ignatov T, Eggemann H, Semczuk A et al (2010) Role of GPR30 in endometrial pathology after tamoxifen for breast cancer. Am J Obstet Gynecol 203:595 e9–e16
Vivacqua A, Bonofiglio D, Recchia AG et al (2006) The G protein-coupled receptor GPR30 mediates the proliferative effects induced by 17beta-estradiol and hydroxytamoxifen in endometrial cancer cells. Mol Endocrinol 20:631–646
Funding
This research did not receive any specific Grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Ethical standards
The experiments comply with the current laws of Germany and were performed according to the good clinical practice (GCP) guidelines.
Rights and permissions
About this article
Cite this article
Ignatov, T., Claus, M., Nass, N. et al. G-protein-coupled estrogen receptor GPER-1 expression in hormone receptor-positive breast cancer is associated with poor benefit of tamoxifen. Breast Cancer Res Treat 174, 121–127 (2019). https://doi.org/10.1007/s10549-018-5064-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-5064-8