Abstract
Purpose
In rodents, a single exercise bout performed 24 h prior to a single doxorubicin treatment provides cardio-protection. This study investigated whether performing this intervention prior to every doxorubicin treatment for breast cancer reduced subclinical cardiotoxicity and treatment symptoms.
Methods
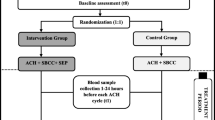
Twenty-four women with early stage breast cancer were randomly assigned to perform a 30-min, vigorous-intensity treadmill bout 24 h prior to each of four doxorubicin-containing chemotherapy treatments or to usual care. Established echocardiographic and circulating biomarkers of subclinical cardiotoxicity, as well as blood pressure and body weight were measured before the first and 7–14 days after the last treatment. The Rotterdam symptom checklist was used to assess patient-reported symptoms.
Results
The exercise and usual care groups did not differ in the doxorubicin-related change in longitudinal strain, twist, or cardiac troponin. However, the four total exercise bouts prevented changes in hemodynamics (increased cardiac output, resting heart rate, decreased systemic vascular resistance, p < 0.01) and reduced body weight gain, prevalence of depressed mood, sore muscles, and low back pain after the last treatment (p < 0.05) relative to the usual care group. No adverse events occurred.
Conclusions
An exercise bout performed 24 h prior to every doxorubicin treatment did not have an effect on markers of subclinical cardiotoxicity, but had a positive systemic effect on hemodynamics, musculoskeletal symptoms, mood, and body weight in women with breast cancer. A single exercise bout prior to chemotherapy treatments may be a simple clinical modality to reduce symptoms and weight gain among women with breast cancer.

Similar content being viewed by others
References
Crozier JA, Swaika A, Moreno-Aspitia A (2014) Adjuvant chemotherapy in breast cancer: to use or not to use, the anthracyclines. World J Clin Oncol 5:529–538. https://doi.org/10.5306/wjco.v5.i3.529
Plana JC, Galderisi M, Barac A et al (2014) Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 27:911–939. https://doi.org/10.1016/j.echo.2014.07.012
Henderson IC (2011) Can we abandon anthracyclines for early breast cancer patients? Oncology 25:115–124
Thavendiranathan P, Poulin F, Lim K-D et al (2014) Use of myocardial strain imaging by echocardiography for the early detection of cardiotoxicity in patients during and after cancer chemotherapy: a systematic review. J Am Coll Cardiol 63:2751–2768. https://doi.org/10.1016/j.jacc.2014.01.073
Dolci A, Dominici R, Cardinale D et al (2008) Biochemical markers for prediction of chemotherapy-induced cardiotoxicity: systematic review of the literature and recommendations for use. Am J Clin Pathol 130:688–695. https://doi.org/10.1309/AJCPB66LRIIVMQDR
Patnaik JL, Byers T, DiGuiseppi C et al (2011) Cardiovascular disease competes with breast cancer as the leading cause of death for older females diagnosed with breast cancer: a retrospective cohort study. Breast Cancer Res 13:R64. https://doi.org/10.1186/bcr2901
Kirkham AA, Davis MK (2015) Exercise prevention of cardiovascular disease in breast cancer survivors. J Oncol. https://doi.org/10.1155/2015/917606
Ascensão A, Lumini Oliveira J, Machado NG et al (2011) Acute exercise protects against calcium-induced cardiac mitochondrial permeability transition pore opening in doxorubicin-treated rats. Clin Sci 120:37–49. https://doi.org/10.1038/35037710
Wonders KY, Hydock DS, Schneider CM, Hayward R (2008) Acute exercise protects against doxorubicin cardiotoxicity. Integr Cancer Ther 7:147–154. https://doi.org/10.1177/1534735408322848
Kirkham AA, Shave RE, Bland KA et al (2017) Protective effects of acute exercise prior to doxorubicin on cardiac function of breast cancer patients: a proof-of-concept RCT. Int J Cardiol. https://doi.org/10.1016/j.ijcard.2017.07.037
Taylor HL, Jacobs DR, Schucker B et al (1978) A questionnaire for the assessment of leisure time physical activities. J Chron Dis 31:741–755
Gulati M, Shaw LJ, Thisted RA et al (2010) Heart rate response to exercise stress testing in asymptomatic women: the St. James Women Take Heart Project. Circulation 122:130–137. https://doi.org/10.1161/CIRCULATIONAHA.110.939249
Schmitz KH, Courneya K, Matthews C et al (2010) American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc 42:1409–1426. https://doi.org/10.1249/MSS.0b013e3181e0c112
Godin G (2011) The godin-shephard leisure-time physical activity questionnaire. Health Fit J Can 4:18–22
Lang RM, Badano LP, Mor-Avi V et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28(1–39):e14. https://doi.org/10.1016/j.echo.2014.10.003
van Dalen BM, Vletter WB, Soliman OII et al (2008) Importance of transducer position in the assessment of apical rotation by speckle tracking echocardiography. J Am Soc Echocardiogr 21:895–898. https://doi.org/10.1016/j.echo.2008.02.001
Stöhr EJ, González-Alonso J, Pearson J et al (2010) Effects of graded heat stress on global left ventricular function and twist mechanics at rest and during exercise in healthy humans. Exp Physiol 96:114–124. https://doi.org/10.1113/expphysiol.2010.055137
De Haes JC, Van Knippenberg FC, Neijt JP (1990) Measuring psychological and physical distress in cancer patients: structure and application of the Rotterdam Symptom Checklist. Br J Cancer 62:1034–1038
Bate ST, Clark RA (2014) The Design and Statistical Analysis of Animal Experiments. Cambridge University Press, New York
Sawaya H, Sebag IA, Plana JC et al (2012) Assessment of echocardiography and biomarkers for the extended prediction of cardiotoxicity in patients treated with anthracyclines, taxanes, and trastuzumab. Circ Cardiovasc Imaging 5:596–603. https://doi.org/10.1161/CIRCIMAGING.112.973321
Stoodley PW, Richards DAB, Boyd A et al (2013) Altered left ventricular longitudinal diastolic function correlates with reduced systolic function immediately after anthracycline chemotherapy. Eur Heart J Cardiovasc Imaging 14:228–234. https://doi.org/10.1093/ehjci/jes139
Stoodley PW, Richards DAB, Hui R et al (2011) Two-dimensional myocardial strain imaging detects changes in left ventricular systolic function immediately after anthracycline chemotherapy. Eur J Echocardiogr 12:945–952. https://doi.org/10.1093/ejechocard/jer187
Florescu M, Magda LS, Enescu OA et al (2014) Early detection of epirubicin-induced cardiotoxicity in patients with breast cancer. J Am Soc Echocardiogr 27:83–92. https://doi.org/10.1016/j.echo.2013.10.008
Lange SA, Jung J, Jaeck A et al (2015) Subclinical myocardial impairment occurred in septaland anterior LV wall segments after anthracycline-embedded chemotherapy and did not worsen during adjuvant trastuzumab treatment in breast cancer patients. Cardiovasc Toxicol. https://doi.org/10.1007/s12012-015-9328-9
Tan TC, Bouras S, Sawaya H et al (2015) Time trends of left ventricular ejection fraction and myocardial deformation indices in a cohort of women with breast cancer treated with anthracyclines, taxanes and trastuzumab. J Am Soc Echocardiogr 28:509–514. https://doi.org/10.1016/j.echo.2015.02.001
Mornoş C, Ruşinaru D, Ionac A et al (2011) Additive value of torsion to global longitudinal left ventricular strain in patients with reduced ejection fraction. Acta Cardiol 66:565. https://doi.org/10.2143/AC.66.5.2131080
Motoki H, Koyama J, Nakazawa H et al (2012) Torsion analysis in the early detection of anthracycline-mediated cardiomyopathy. Eur Heart J Cardiovasc Imaging 13:95–103. https://doi.org/10.1093/ehjci/jer172
Burns AT, La Gerche A, D’hooge J et al (2010) Left ventricular strain and strain rate: characterization of the effect of load in human subjects. Eur J Echocardiogr 11:283–289. https://doi.org/10.1093/ejechocard/jep214
Burns AT, Gerche AL, Prior DL, MacIsaac AI (2010) Left ventricular torsion parameters are affected by acute changes in load. Echocardiography 27:407–414. https://doi.org/10.1111/j.1540-8175.2009.01037.x
Bosch X, Rovira M, Sitges M et al (2013) Enalapril and carvedilol for preventing chemotherapy-induced left ventricular systolic dysfunction in patients with malignant hemopathies: the overcome trial (prevention of left ventricular dysfunction with enalapril and carvedilol in patients submitted to intensive Chemotherapy for the treatment of malignant hemopathies). J Am Coll Cardiol 61:2355–2362
Gulati G, Heck SL, Ree AH et al (2016) Prevention of cardiac dysfunction during adjuvant breast cancer therapy (PRADA): a 2 × 2 factorial, randomized, placebo-controlled, double-blind clinical trial of candesartan and metoprolol. Eur Heart J 37:1671–1680. https://doi.org/10.1093/eurheartj/ehw022
Sandri MT, Salvatici M, Cardinale D et al (2005) N-terminal pro-B-type natriuretic peptide after high-dose chemotherapy: a marker predictive of cardiac dysfunction? Clin Chem 51:1405–1410. https://doi.org/10.1373/clinchem.2005.050153
Romano S, Fratini S, Ricevuto E et al (2011) Serial measurements of NT-proBNP are predictive of not-high-dose anthracycline cardiotoxicity in breast cancer patients. Br J Cancer 105:1663–1668. https://doi.org/10.1038/bjc.2011.439
Ky B, Putt M, Sawaya H et al (2014) Early increases in multiple biomarkers predict subsequent cardiotoxicity in patients with breast cancer treated with doxorubicin, taxanes, and trastuzumab. J Am Coll Cardiol 63:809–816. https://doi.org/10.1016/j.jacc.2013.10.061
Sawaya H, Sebag IA, Plana JC et al (2011) Early detection and prediction of cardiotoxicity in chemotherapy-treated patients. Am J Cardiol 107:1375–1380. https://doi.org/10.1016/j.amjcard.2011.01.006
Metivier F, Marchais SJ, Guerin AP et al (2000) Pathophysiology of anaemia: focus on the heart and blood vessels. Nephrol Dial Transplant 15:14–18. https://doi.org/10.1093/oxfordjournals.ndt.a027970
Campbell KL, Lane K, Martin A et al (2007) Resting energy expenditure and body mass changes in women during adjuvant chemotherapy for breast cancer. Cancer Nurs 30:95–100
Lakoski SG, Eves ND, Douglas PS, Jones LW (2012) Exercise rehabilitation in patients with cancer. Nat Rev Clin Oncol 9:288–296. https://doi.org/10.1038/nrclinonc.2012.27
Kelley DE, Goodpaster B, Wing RR, Simoneau JA (1999) Skeletal muscle fatty acid metabolism in association with insulin resistance, obesity, and weight loss. Am J Physiol 277:E1130–E1141
Filler K, Lyon D, Bennett J et al (2014) Association of mitochondrial dysfunction and fatigue: a review of the literature. BBA Clin 1:12–23. https://doi.org/10.1016/j.bbacli.2014.04.001
Jones LW, Haykowsky M, Pituskin EN et al (2007) Cardiovascular reserve and risk profile of postmenopausal women after chemoendocrine therapy for hormone receptor positive operable breast cancer. Oncologist 12:1156–1164. https://doi.org/10.1634/theoncologist.12-10-1156
Thayer JF, Lane RD (2007) The role of vagal function in the risk for cardiovascular disease and mortality. Biol Psychol 74:224–242. https://doi.org/10.1016/j.biopsycho.2005.11.013
Hornsby WE, Douglas PS, West MJ et al (2014) Safety and efficacy of aerobic training in operable breast cancer patients receiving neoadjuvant chemotherapy: a phase II randomized trial. Acta Oncol 53:65–74. https://doi.org/10.3109/0284186X.2013.781673
Ghose A, Kundu R, Toumeh A et al (2015) A review of obesity, insulin resistance, and the role of exercise in breast cancer patients. Nutr Cancer 67:197–202. https://doi.org/10.1080/01635581.2015.990569
Schubert MM, Sabapathy S, Leveritt M, Desbrow B (2013) Acute exercise and hormones related to appetite regulation: a meta-analysis. Sports Med 44:387–403. https://doi.org/10.1007/s40279-013-0120-3
Financial support
This work was supported by an Anita Cochrane Memorial Fund Award from the British Columbia Cancer Foundation. AK was supported by the Canadian Institutes of Health Research. We also acknowledge the support of GE Healthcare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Kirkham, A.A., Eves, N.D., Shave, R.E. et al. The effect of an aerobic exercise bout 24 h prior to each doxorubicin treatment for breast cancer on markers of cardiotoxicity and treatment symptoms: a RCT. Breast Cancer Res Treat 167, 719–729 (2018). https://doi.org/10.1007/s10549-017-4554-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4554-4