Abstract
Purpose
To update and expand the Rosner–Colditz breast cancer incidence model by evaluating the contributions of more recently identified risk factors as well as predicted percent mammographic density (MD) to breast cancer risk.
Methods
Using data from the Nurses’ Health Study (NHS) and NHSII, we added adolescent somatotype (9 unit scale), vegetable intake (servings/day), breastfeeding (months), physical activity (MET-h/week), and predicted percent MD to the Rosner–Colditz model to determine whether these variables improved model discrimination. We evaluated all invasive as well as ER+/PR+, ER+/PR−, and ER−/PR− breast cancer.
Results
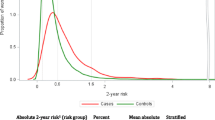
In the NHS/NHSII, we accrued over 5200 cases of invasive breast cancer over more than 20 years of follow-up with complete data on the risk factors. Adolescent somatotype and predicted percent MD significantly improved the original Rosner–Colditz model for all invasive breast cancer (change in age-adjusted AUC = 0.020, p < 0.001). The relative risk (RR) of invasive breast cancer for a 4-unit increase in adolescent somatotype was 0.62 (95% CI 0.56, 0.70), whereas the RR for a 20-unit increase in predicted percent MD was 1.32 (95% CI 1.28, 1.36). Adolescent somatotype and predicted percent MD also significantly improved the ER+/PR+model (change in age-adjusted AUC = 0.020, p < 0.001) as well as the ER+/PR− model (change in age-adjusted AUC = 0.012, p = 0.007). Adolescent somatotype, predicted percent MD, breastfeeding, and vegetable intake improved the ER−/PR− model (change in AUC = 0.031, p < 0.0001). The RR of ER−/PR− disease for 5 vegetable servings/day increase was 0.83 (95% CI 0.70, 0.99), while the RR for every 12 months of breastfeeding was 0.88 (95% CI 0.77, 1.01). Physical activity did not improve risk classification in any model.
Conclusion
Adolescent somatotype and predicted percent MD significantly improved breast cancer risk classification using the Rosner–Colditz model. Further, risk factors specific to ER− disease, such as breastfeeding and vegetable intake, may also help improve risk prediction of this aggressive subtype.

Similar content being viewed by others
Abbreviations
- NHS:
-
Nurses’ Health Study
- BMI:
-
Body mass index
- BBD:
-
Benign breast disease
- HT:
-
Hormone therapy
- MD:
-
Mammographic density
- ER:
-
Estrogen receptor
- PR:
-
Progesterone receptor
References
Rosner B, Colditz GA (1996) Nurses’ health study: log-incidence mathematical model of breast cancer incidence. J Natl Cancer Inst 88(6):359–364
Colditz GA, Rosner B (2000) Cumulative risk of breast cancer to age 70 years according to risk factor status: data from the Nurses’ Health Study. Am J Epidemiol 152(10):950–964
Li J et al (2010) Effects of childhood body size on breast cancer tumour characteristics. Breast Cancer Res 12(2):R23
Ahlgren M et al (2004) Growth patterns and the risk of breast cancer in women. N Engl J Med 351(16):1619–1626
Bardia A et al (2008) Relative weight at age 12 and risk of postmenopausal breast cancer. Cancer Epidemiol Biomarkers Prev 17(2):374–378
Hilakivi-Clarke L et al (2001) Tallness and overweight during childhood have opposing effects on breast cancer risk. Br J Cancer 85(11):1680–1684
Baer HJ et al (2005) Body fatness during childhood and adolescence and incidence of breast cancer in premenopausal women: a prospective cohort study. Breast Cancer Res 7(3):R314–R325
Baer HJ et al (2010) Body fatness at young ages and risk of breast cancer throughout life. Am J Epidemiol 171(11):1183–1194
Eliassen AH et al (2010) Physical activity and risk of breast cancer among postmenopausal women. Arch Intern Med 170(19):1758–1764
Peters TM et al (2009) Physical activity and postmenopausal breast cancer risk in the NIH-AARP diet and health study. Cancer Epidemiol Biomarkers Prev 18(1):289–296
Dallal CM et al (2007) Long-term recreational physical activity and risk of invasive and in situ breast cancer: the California teachers study. Arch Intern Med 167(4):408–415
Steindorf K et al (2013) Physical activity and risk of breast cancer overall and by hormone receptor status: the European prospective investigation into cancer and nutrition. Int J Cancer 132(7):1667–1678
Wu Y, Zhang D, Kang S (2013) Physical activity and risk of breast cancer: a meta-analysis of prospective studies. Breast Cancer Res Treat 137(3):869–882
Neilson HK et al (2017) Moderate-vigorous recreational physical activity and breast cancer risk, stratified by menopause status: a systematic review and meta-analysis. Menopause 24(3):322–344
Fung TT et al (2005) Dietary patterns and the risk of postmenopausal breast cancer. Int J Cancer 116(1):116–121
Fung TT et al (2013) Intake of specific fruits and vegetables in relation to risk of estrogen receptor-negative breast cancer among postmenopausal women. Breast Cancer Res Treat 138(3):925–930
Jung S et al (2013) Fruit and vegetable intake and risk of breast cancer by hormone receptor status. J Natl Cancer Inst 105(3):219–236
Sisti JS et al (2015) Reproductive factors, tumor estrogen receptor status and contralateral breast cancer risk: results from the WECARE study. Springerplus 4:825
Sisti JS et al (2015) Reproductive risk factors in relation to molecular subtypes of breast cancer: results from the Nurses’ Health Studies. Int J Cancer 18(10):29968
Boyd NF et al (2009) Mammographic density. Breast Cancer Res 11(Suppl 3):S4
Warwick J et al (2014) Mammographic breast density refines Tyrer-Cuzick estimates of breast cancer risk in high-risk women: findings from the placebo arm of the International Breast Cancer Intervention Study I. Breast Cancer Res 16(5):451
Brentnall AR et al (2015) Mammographic density adds accuracy to both the Tyrer-Cuzick and Gail breast cancer risk models in a prospective UK screening cohort. Breast Cancer Res 17(1):147
Rice MS, Rosner BA, Tamimi RM (2017) Percent mammographic density prediction: development of a model in the Nurses’ Health Studies. Cancer Causes Control 28(7):677–684
Harris HR et al (2011) Body size across the life course, mammographic density, and risk of breast cancer. Am J Epidemiol 174(8):909–918
Colditz GA et al (2003) Physical activity and risk of breast cancer in premenopausal women. Br J Cancer 89(5):847–851
Rice MS et al (2016) Mammographic density and breast cancer risk: a mediation analysis. Breast Cancer Res 18(1):94
Yaghjyan L et al (2015) Postmenopausal mammographic breast density and subsequent breast cancer risk according to selected tissue markers. Br J Cancer 113(7):1104–1113
Tamimi RM et al (2012) Traditional breast cancer risk factors in relation to molecular subtypes of breast cancer. Breast Cancer Res Treat 131(1):159–167
Rosner B, Glynn RJ (2009) Power and sample size estimation for the Wilcoxon rank sum test with application to comparisons of C statistics from alternative prediction models. Biometrics 65(1):188–197
DerSimonian R (1996) Meta-analysis in the design and monitoring of clinical trials. Stat Med 15(12):1237–1248; discussion 1249–1252
Rosner B et al (2008) Risk prediction models with incomplete data with application to prediction of estrogen receptor-positive breast cancer: prospective data from the Nurses’ Health Study. Breast Cancer Res 10(4):R55
Rosner BA et al (2013) Validation of Rosner–Colditz breast cancer incidence model using an independent data set, the California Teachers Study. Breast Cancer Res Treat 142(1):187–202
Berkey CS et al (1999) Adolescence and breast carcinoma risk. Cancer 85(11):2400–2409
Baer HJ et al (2007) Adiposity and sex hormones in girls. Cancer Epidemiol Biomarkers Prev 16(9):1880–1888
Poole EM et al (2011) Body size in early life and adult levels of insulin-like growth factor 1 and insulin-like growth factor binding protein 3. Am J Epidemiol 174(6):642–651
Samimi G et al (2008) Measures of energy balance and mammographic density in the Nurses’ Health Study. Breast Cancer Res Treat 109(1):113–122
McCormack VA, dos Santos I (2006) Silva, Breast density and parenchymal patterns as markers of breast cancer risk: a meta-analysis. Cancer Epidemiol Biomarkers Prev 15(6):1159–1169
Tice JA et al (2008) Using clinical factors and mammographic breast density to estimate breast cancer risk: development and validation of a new predictive model. Ann Intern Med 148(5):337–347
Vachon CM et al (2015) The contributions of breast density and common genetic variation to breast cancer risk. J Natl Cancer Inst 107(5):dju397
Michailidou K et al (2013) Large-scale genotyping identifies 41 new loci associated with breast cancer risk. Nat Genet 45(4):353–361, 361e1–2
Tworoger SS et al (2014) Inclusion of endogenous hormone levels in risk prediction models of postmenopausal breast cancer. J Clin Oncol 32(28):3111–3117
Acknowledgments
This study was supported by research Grants from the National Cancer Institute, National Institutes of Health, UM1 CA186107, P01 CA87969, UM1 CA176726, R01CA175080, and T32 CA09001. We would like to thank the participants of the Nurses’ Health Study and Nurses’ Health Study II for their continuing contributions. We thank the following state cancer registries for their help: AL, AZ, AR, CA, CO, CT, DE, FL, GA, ID, IL, IN, IA, KY, LA, ME, MD, MA, MI, NE, NH, NJ, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, VA, WA, WY. The authors assume full responsibility for analyses and interpretation of these data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rice, M.S., Tworoger, S.S., Hankinson, S.E. et al. Breast cancer risk prediction: an update to the Rosner–Colditz breast cancer incidence model. Breast Cancer Res Treat 166, 227–240 (2017). https://doi.org/10.1007/s10549-017-4391-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4391-5