Abstract
Purpose
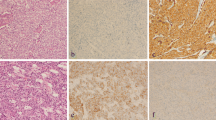
Basal marker expression in triple-negative breast cancers identifies basal-like tumours, and thus separates the TN group into two prognostic groups. However, the expression and prognostic significance of basal markers in luminal breast cancers are poorly described. The aim of this study was to investigate the expression and prognostic value of basal markers (CK5, CK14 and EGFR) in luminal breast cancer.
Methods
A total of 1423 formalin-fixed, paraffin-embedded breast cancer tumours from a well-characterized cohort of Norwegian women, previously reclassified into molecular subtypes using IHC and ISH, were included in the study. For the present study, tumours expressing at least one of the basal markers CK5, CK14 or EGFR were defined as basal marker positive. Cumulative incidence of death from breast cancer and hazard ratio analyses were used to assess prognosis according to basal marker expression.
Results and conclusion
In total, 470 cases (33.0%) were basal marker positive. A higher proportion of the basal marker-positive tumours were of histopathological grade 3 compared to basal marker negative. For hormone receptor-positive, HER2-negative cases, we found better prognosis for basal marker-positive breast cancer compared to basal marker negative. For all subtypes combined, poorer prognosis for basal marker-negative cases was found in histopathological grade 2 tumours but not among grade 1 and 3.





Similar content being viewed by others
References
Alshareeda AT, Soria D, Garibaldi JM, Rakha E, Nolan C, Ellis IO, Green AR (2013) Characteristics of basal cytokeratin expression in breast cancer. Breast Cancer Res Treat 139:23–37. doi:10.1007/s10549-013-2518-x
Asleh-Aburaya K, Sheffield BS, Kos Z, Won JR, Wang XQ, Gao D, Wolber R, Gilks CB, Bernard PS, Chia SK, Nielsen TO (2017) Basal biomarkers nestin and INPP4b identify intrinsic subtypes accurately in breast cancers that are weakly positive for oestrogen receptor. Histopathology 70:185–194. doi:10.1111/his.13038
Badve S, Dabbs DJ, Schnitt SJ, Baehner FL, Decker T, Eusebi V, Fox SB, Ichihara S, Jacquemier J, Lakhani SR, Palacios J, Rakha EA, Richardson AL, Schmitt FC, Tan PH, Tse GM, Weigelt B, Ellis IO, Reis-Filho JS (2011) Basal-like and triple-negative breast cancers: a critical review with an emphasis on the implications for pathologists and oncologists. Mod Pathol 24:157–167. doi:10.1038/modpathol.2010.200
Bhargava R, Beriwal S, McManus K, Dabbs DJ (2008) CK5 is more sensitive than CK5/6 in identifying the “basal-like” phenotype of breast carcinoma. Am J Clin Pathol 130:724–730. doi:10.1309/ajcp3kff1ltywqiy
Blows FM, Driver KE, Schmidt MK, Broeks A, van Leeuwen FE, Wesseling J, Cheang MC, Gelmon K, Nielsen TO, Blomqvist C, Heikkila P, Heikkinen T, Nevanlinna H, Akslen LA, Begin LR, Foulkes WD, Couch FJ, Wang X, Cafourek V, Olson JE, Baglietto L, Giles GG, Severi G, McLean CA, Southey MC, Rakha E, Green AR, Ellis IO, Sherman ME, Lissowska J, Anderson WF, Cox A, Cross SS, Reed MW, Provenzano E, Dawson SJ, Dunning AM, Humphreys M, Easton DF, Garcia-Closas M, Caldas C, Pharoah PD, Huntsman D (2010) Subtyping of breast cancer by immunohistochemistry to investigate a relationship between subtype and short and long term survival: a collaborative analysis of data for 10,159 cases from 12 studies. PLoS Med 7:e1000279. doi:10.1371/journal.pmed.1000279
Callagy G, Cattaneo E, Daigo Y, Happerfield L, Bobrow LG, Pharoah PD, Caldas C (2003) Molecular classification of breast carcinomas using tissue microarrays. Diagn Mol Pathol 12:27–34
Cheang MC, Voduc D, Bajdik C, Leung S, McKinney S, Chia SK, Perou CM, Nielsen TO (2008) Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res 14:1368–1376. doi:10.1158/1078-0432.ccr-07-1658
Chowdhury N (2011) Histopathological and genomic grading provide complementary prognostic information in breast cancer: a study on publicly available datasets. Patholog Res Int 2011:890938. doi:10.4061/2011/890938
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19:403–410
Engstrom MJ, Opdahl S, Hagen AI, Romundstad PR, Akslen LA, Haugen OA, Vatten LJ, Bofin AM (2013) Molecular subtypes, histopathological grade and survival in a historic cohort of breast cancer patients. Breast Cancer Res Treat. doi:10.1007/s10549-013-2647-2
Hennigs A, Riedel F, Gondos A, Sinn P, Schirmacher P, Marme F, Jager D, Kauczor HU, Stieber A, Lindel K, Debus J, Golatta M, Schutz F, Sohn C, Heil J, Schneeweiss A (2016) Prognosis of breast cancer molecular subtypes in routine clinical care: a large prospective cohort study. BMC Cancer 16:734. doi:10.1186/s12885-016-2766-3
Herbst RS (2004) Review of epidermal growth factor receptor biology. Int J Radiat Oncol Biol Phys 59:21–26. doi:10.1016/j.ijrobp.2003.11.041
Hofvind S, Lee CI, Elmore JG (2012) Stage-specific breast cancer incidence rates among participants and non-participants of a population-based mammographic screening program. Breast Cancer Res Treat 135:291–299. doi:10.1007/s10549-012-2162-x
Holmen JMK, Kruger Ø, Langhammer A, Holmen TL, Bratberg GH et al (2003) The Nord-Trøndelag health study 1995–97 (HUNT 2): objectives, contents, methods and participation. Norsk Epidemiologi 13:19–32
Ishitha G, Manipadam MT, Backianathan S, Chacko RT, Abraham DT, Jacob PM (2016) Clinicopathological study of triple negative breast cancers. J Clin Diagn Res. doi:10.7860/JCDR/2016/20475.8539
Kononen J, Bubendorf L, Kallioniemi A, Barlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G, Kallioniemi OP (1998) Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 4:844–847
Korsching E, Packeisen J, Agelopoulos K, Eisenacher M, Voss R, Isola J, van Diest PJ, Brandt B, Boecker W, Buerger H (2002) Cytogenetic alterations and cytokeratin expression patterns in breast cancer: integrating a new model of breast differentiation into cytogenetic pathways of breast carcinogenesis. Lab Invest 82:1525–1533
Kvåle G, Heuch I, Eide G (1987) A prospective study of reproductive factors and breast cancer. Am J Epidemiol 126:831–841
Lakhani SR, Ellis I, Schnitt SJ, Tan PH, Van de Vijver M, World Health Organization (eds) (2012) WHO Classification of Tumours of the Breast. International Agency for Research on Cancer (IARC), Lyon
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2006) REporting recommendations for tumor MARKer prognostic studies (REMARK). Breast Cancer Res Treat 100:229–235. doi:10.1007/s10549-006-9242-8
Moll RFW, Schiller DL (1982) The Catalog of human cytokeratins: patterns of expression in normal epithelia, tumors and cultured cells. Cell 31:11–24
Nielsen TO, Hsu FD, Jensen K, Cheang M, Karaca G, Hu Z, Hernandez-Boussard T, Livasy C, Cowan D, Dressler L, Akslen LA, Ragaz J, Gown AM, Gilks CB, van de Rijn M, Perou CM (2004) Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 10:5367–5374. doi:10.1158/1078-0432.ccr-04-0220
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lonning PE, Borresen-Dale AL, Brown PO, Botstein D (2000) Molecular portraits of human breast tumours. Nature 406:747–752. doi:10.1038/35021093
Pinder SE, Brown JP, Gillett C, Purdie CA, Speirs V, Thompson AM, Shaaban AM (2013) The manufacture and assessment of tissue microarrays: suggestions and criteria for analysis, with breast cancer as an example. J Clin Pathol 66:169–177. doi:10.1136/jclinpath-2012-201091
Rakha EA, Patel A, Powe DG, Benhasouna A, Green AR, Lambros MB, Reis-Filho JS, Ellis IO (2010) Clinical and biological significance of E-cadherin protein expression in invasive lobular carcinoma of the breast. Am J Surg Pathol 34:1472–1479. doi:10.1097/PAS.0b013e3181f01916
Schweizer J, Bowden PE, Coulombe PA, Langbein L, Lane EB, Magin TM, Maltais L, Omary MB, Parry DA, Rogers MA, Wright MW (2006) New consensus nomenclature for mammalian keratins. J Cell Biol 174:169–174. doi:10.1083/jcb.200603161
Sung H, Garcia-Closas M, Chang-Claude J, Blows FM, Ali HR, Figueroa J, Nevanlinna H, Fagerholm R, Heikkila P, Blomqvist C, Giles GG, Milne RL, Southey MC, McLean C, Mannermaa A, Kosma VM, Kataja V, Sironen R, Couch FJ, Olson JE, Hallberg E, Olswold C, Cox A, Cross SS, Kraft P, Tamimi RM, Eliassen AH, Schmidt MK, Bolla MK, Wang Q, Easton D, Howat WJ, Coulson P, Pharoah PD, Sherman ME, Yang XR (2016) Heterogeneity of luminal breast cancer characterised by immunohistochemical expression of basal markers. Br J Cancer 114:298–304. doi:10.1038/bjc.2015.437
Tas F, Bilgin E, Karabulut S, Duranyildiz D (2014) Clinical significance of serum epidermal growth factor receptor (EGFR) levels in patients with breast cancer. Cytokine 71:66–70. doi:10.1016/j.cyto.2014.09.001
Tischkowitz M, Brunet JS, Begin LR, Huntsman DG, Cheang MC, Akslen LA, Nielsen TO, Foulkes WD (2007) Use of immunohistochemical markers can refine prognosis in triple negative breast cancer. BMC Cancer 7:134. doi:10.1186/1471-2407-7-134
Valentin MD, da Silva SD, Privat M, Alaoui-Jamali M, Bignon YJ (2012) Molecular insights on basal-like breast cancer. Breast Cancer Res Treat 134:21–30. doi:10.1007/s10549-011-1934-z
Valla M, Vatten LJ, Engstrom MJ, Haugen OA, Akslen LA, Bjorngaard JH, Hagen AI, Ytterhus B, Bofin AM, Opdahl S (2016) Molecular subtypes of breast cancer: long-term incidence trends and prognostic differences. Cancer Epidemiol Biomarkers Prev. doi:10.1158/1055-9965.epi-16-0427
Valla M, Vatten LJ, Engstrom MJ, Haugen OA, Akslen LA, Bjorngaard JH, Hagen AI, Ytterhus B, Bofin AM, Opdahl S (2016) Molecular subtypes of breast cancer: long-term incidence trends and prognostic differences. Cancer Epidemiol Biomarkers Prev 25:1625–1634. doi:10.1158/1055-9965.EPI-16-0427
Yadav BS, Chanana P, Jhamb S (2015) Biomarkers in triple negative breast cancer: a review. World J Clin Oncol 6:252–263. doi:10.5306/wjco.v6.i6.252
Acknowledgements
The authors thank the Department of Pathology and Medical Genetics at St. Olav’s Hospital, Trondheim University Hospital, Norway for making the archives available for the study; the Cancer Registry of Norway, and the Norwegian Cause of Death Registry for providing the patient data. Biomedical scientists Borgny Ytterhus and Anette Skjervold have made invaluable contributions to the logistical and laboratory aspects of the study. The Cancer Registry of Norway; Research Council of Norway; and the Liaison Committee between the Central Norway Regional Health Authority and the Norwegian University of Science and Technology supported this work.
Authors’ contribution
MJE contributed to conception and design of the study, interpretation of the IHC markers, carried out statistical analyses, interpretation of the results and drafted the manuscript. MV contributed to discussion and interpretation of the results, interpretation of the IHC markers, review and revision of the manuscript. AMB contributed to discussion and interpretation of the results, interpretation of the IHC markers, review and revision of the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Engstrøm, M.J., Valla, M. & Bofin, A.M. Basal markers and prognosis in luminal breast cancer. Breast Cancer Res Treat 163, 207–217 (2017). https://doi.org/10.1007/s10549-017-4182-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4182-z