Abstract
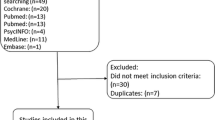
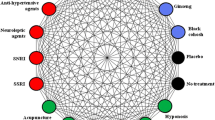
Patient-centered decision making about hot flash treatments often incorporates a balance of efficacy and side effects in addition to patient preference. This systematic review examines randomized controlled trials (RCTs) comparing at least two non-hormonal hot flash treatments in breast cancer survivors. In July 2015, PubMed, SCOPUS, CINAHL, Cochrane, and Web of Science databases were searched for RCTs comparing active, non-hormonal hot flash treatments in female breast cancer survivors. Thirteen trials were included after identifying 906 potential studies. Four trials were dose comparison studies of pharmacologic treatments citalopram, venlafaxine, gabapentin, and paroxetine. Hot flash reduction did not differ by tamoxifen or aromatase inhibitor use. Citalopram 10, 20, and 30 mg daily had comparable outcomes. Venlafaxine 75 mg daily improved hot flashes without additional side effects from higher dosing. Gabapentin 900 mg daily improved hot flashes more than 300 mg. Paroxetine 10 mg daily had fewer side effects than 20 mg. Among four trials comparing different pharmacologic treatments, venlafaxine alleviated hot flash symptoms faster than clonidine; participants preferred venlafaxine over gabapentin. Five trials compared pharmacologic to non-pharmacologic treatments. Acupuncture had similar efficacy to venlafaxine and gabapentin but may have longer durability after completing treatment and fewer side effects. We could not perform a pooled meta-analysis because outcomes were not reported in comparable formats. Clinical trial data on non-hormonal hot flash treatments provide comparisons of hot flash efficacy and other patient important outcomes to guide clinical management. Clinicians can use the information to help patients select hot flash interventions.


Similar content being viewed by others
References
Couzi RJ, Helzlsouer KJ, Fetting JH (1995) Prevalence of menopausal symptoms among women with a history of breast cancer and attitudes toward estrogen replacement therapy. J Clin Oncol 13(11):2737–2744
Bernhard J, Luo W, Ribi K, Colleoni M, Burstein HJ, Tondini C, Pinotti G, Spazzapan S, Ruhstaller T, Puglisi F, Pavesi L, Parmar V, Regan MM, Pagani O, Fleming GF, Francis PA, Price KN, Coates AS, Gelber RD, Goldhirsch A, Walley BA (2015) Patient-reported outcomes with adjuvant exemestane versus tamoxifen in premenopausal women with early breast cancer undergoing ovarian suppression (TEXT and SOFT): a combined analysis of two phase 3 randomised trials. Lancet Oncol 16(7):848–858. doi:10.1016/s1470-2045(15)00049-2
Day R, Ganz PA, Costantino JP, Cronin WM, Wickerham DL, Fisher B (1999) Health-related quality of life and tamoxifen in breast cancer prevention: a report from the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Clin Oncol 17(9):2659–2669
Leining MG, Gelber S, Rosenberg R, Przypyszny M, Winer EP, Partridge AH (2006) Menopausal-type symptoms in young breast cancer survivors. Ann Oncol 17(12):1777–1782. doi:10.1093/annonc/mdl299
Yoo C, Yun MR, Ahn JH, Jung KH, Kim HJ, Kim JE, Park JY, Park KO, Yoon DH, Kim SB (2013) Chemotherapy-induced amenorrhea, menopause-specific quality of life, and endocrine profiles in premenopausal women with breast cancer who received adjuvant anthracycline-based chemotherapy: a prospective cohort study. Cancer Chemother Pharmacol 72(3):565–575. doi:10.1007/s00280-013-2227-5
Gupta P, Sturdee DW, Palin SL, Majumder K, Fear R, Marshall T, Paterson I (2006) Menopausal symptoms in women treated for breast cancer: the prevalence and severity of symptoms and their perceived effects on quality of life. Climacteric 9(1):49–58. doi:10.1080/13697130500487224
Marino JL, Saunders CM, Emery LI, Green H, Doherty DA, Hickey M (2014) Nature and severity of menopausal symptoms and their impact on quality of life and sexual function in cancer survivors compared with women without a cancer history. Menopause 21(3):267–274. doi:10.1097/GME.0b013e3182976f46
Stein KD, Jacobsen PB, Hann DM, Greenberg H, Lyman G (2000) Impact of hot flashes on quality of life among postmenopausal women being treated for breast cancer. J Pain Symptom Manage 19(6):436–445
Barnabei VM, Cochrane BB, Aragaki AK, Nygaard I, Williams RS, McGovern PG, Young RL, Wells EC, O’Sullivan MJ, Chen B, Schenken R, Johnson SR (2005) Menopausal symptoms and treatment-related effects of estrogen and progestin in the Women’s Health Initiative. Obstet Gynecol 105(5 Pt 1):1063–1073. doi:10.1097/01.aog.0000158120.47542.18
Holmberg L, Iversen OE, Rudenstam CM, Hammar M, Kumpulainen E, Jaskiewicz J, Jassem J, Dobaczewska D, Fjosne HE, Peralta O, Arriagada R, Holmqvist M, Maenpaa J (2008) Increased risk of recurrence after hormone replacement therapy in breast cancer survivors. J Natl Cancer Inst 100(7):475–482. doi:10.1093/jnci/djn058
von Schoultz E, Rutqvist LE (2005) Menopausal hormone therapy after breast cancer: the Stockholm randomized trial. J Natl Cancer Inst 97(7):533–535. doi:10.1093/jnci/dji071
Barton DL, LaVasseur BI, Sloan JA, Stawis AN, Flynn KA, Dyar M, Johnson DB, Atherton PJ, Diekmann B, Loprinzi CL (2010) Phase III, placebo-controlled trial of three doses of citalopram for the treatment of hot flashes: nCCTG trial N05C9. J Clin Oncol 28(20):3278–3283. doi:10.1200/jco.2009.26.6379
Goldberg RM, Loprinzi CL, O’Fallon JR, Veeder MH, Miser AW, Mailliard JA, Michalak JC, Dose AM, Rowland KM, Burnham NL (1994) Transdermal clonidine for ameliorating tamoxifen-induced hot flashes. J Clin Oncol 12(1):155–158
Loprinzi CL, Kugler JW, Sloan JA, Mailliard JA, LaVasseur BI, Barton DL, Novotny PJ, Dakhil SR, Rodger K, Rummans TA, Christensen BJ (2000) Venlafaxine in management of hot flashes in survivors of breast cancer: a randomised controlled trial. Lancet 356(9247):2059–2063. doi:10.1016/s0140-6736(00)03403-6
Loprinzi CL, Qin R, Balcueva EP, Baclueva EP, Flynn KA, Rowland KM, Graham DL, Erwin NK, Dakhil SR, Jurgens DJ, Burger KN (2010) Phase III, randomized, double-blind, placebo-controlled evaluation of pregabalin for alleviating hot flashes, N07C1. J Clin Oncol 28(4):641–647. doi:10.1200/jco.2009.24.5647
Pandya KJ, Morrow GR, Roscoe JA, Zhao H, Hickok JT, Pajon E, Sweeney TJ, Banerjee TK, Flynn PJ (2005) Gabapentin for hot flashes in 420 women with breast cancer: a randomised double-blind placebo-controlled trial. Lancet 366(9488):818–824. doi:10.1016/s0140-6736(05)67215-7
Stearns V, Slack R, Greep N, Henry-Tilman R, Osborne M, Bunnell C, Ullmer L, Gallagher A, Cullen J, Gehan E, Hayes DF, Isaacs C (2005) Paroxetine is an effective treatment for hot flashes: results from a prospective randomized clinical trial. J Clin Oncol 23(28):6919–6930. doi:10.1200/jco.2005.10.081
Bao T, Cai L, Snyder C, Betts K, Tarpinian K, Gould J, Jeter S, Medeiros M, Chumsri S, Bardia A, Tan M, Singh H, Tkaczuk KH, Stearns V (2014) Patient-reported outcomes in women with breast cancer enrolled in a dual-center, double-blind, randomized controlled trial assessing the effect of acupuncture in reducing aromatase inhibitor-induced musculoskeletal symptoms. Cancer 120(3):381–389. doi:10.1002/cncr.28352
Bokmand S, Flyger H (2013) Acupuncture relieves menopausal discomfort in breast cancer patients: a prospective, double blinded, randomized study. Breast 22(3):320–323. doi:10.1016/j.breast.2012.07.015
Carson JW, Carson KM, Porter LS, Keefe FJ, Seewaldt VL (2009) Yoga of awareness program for menopausal symptoms in breast cancer survivors: results from a randomized trial. Support Care Cancer 17(10):1301–1309. doi:10.1007/s00520-009-0587-5
Deng G, Vickers A, Yeung S, D’Andrea GM, Xiao H, Heerdt AS, Sugarman S, Troso-Sandoval T, Seidman AD, Hudis CA, Cassileth B (2007) Randomized, controlled trial of acupuncture for the treatment of hot flashes in breast cancer patients. J Clin Oncol 25(35):5584–5590. doi:10.1200/jco.2007.12.0774
Elkins G, Marcus J, Stearns V, Perfect M, Rajab MH, Ruud C, Palamara L, Keith T (2008) Randomized trial of a hypnosis intervention for treatment of hot flashes among breast cancer survivors. J Clin Oncol 26(31):5022–5026. doi:10.1200/jco.2008.16.6389
Hervik JB, Mjaland O (2011) Acupuncture for the treatment of hot flashes in breast cancer patients, a randomized, controlled trial. Support Care Cancer 19(2 suppl. 1):S102. doi:10.1007/s00520-011-1184-y
Liljegren A, Gunnarsson P, Landgren BM, Robéus N, Johansson H, Rotstein S (2012) Reducing vasomotor symptoms with acupuncture in breast cancer patients treated with adjuvant tamoxifen: a randomized controlled trial. Breast Cancer Res Treat 135(3):791–798. doi:10.1007/s10549-010-1283-3
Mao JJ, Bowman MA, Xie SX, Bruner D, DeMichele A, Farrar JT (2015) Electroacupuncture versus gabapentin for hot flashes among breast cancer survivors: a randomized placebo-controlled trial. J Clin Oncol 33(31):3615–3620. doi:10.1200/jco.2015.60.9412
Dodin S, Blanchet C, Marc I, Ernst E, Wu T, Vaillancourt C, Paquette J, Maunsell E (2013) Acupuncture for menopausal hot flushes. Cochrane Database Syst Rev 7:Cd007410. doi:10.1002/14651858.CD007410.pub2
L’Esperance S, Frenette S, Dionne A, Dionne JY (2013) Pharmacological and non-hormonal treatment of hot flashes in breast cancer survivors: CEPO review and recommendations. Support Care Cancer 21(5):1461–1474. doi:10.1007/s00520-013-1732-8
Loprinzi CL, Sloan J, Stearns V, Slack R, Iyengar M, Diekmann B, Kimmick G, Lovato J, Gordon P, Pandya K, Guttuso T Jr, Barton D, Novotny P (2009) Newer antidepressants and gabapentin for hot flashes: an individual patient pooled analysis. J Clin Oncol 27(17):2831–2837. doi:10.1200/jco.2008.19.6253
Rada G, Capurro D, Pantoja T, Corbalan J, Moreno G, Letelier LM, Vera C (2010) Non-hormonal interventions for hot flushes in women with a history of breast cancer. Cochrane Database Syst Rev. doi:10.1002/14651858.CD004923.pub2
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7):e1000097. doi:10.1371/journal.pmed.1000097
Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, Cochrane Bias Methods Group, Cochrane Statistical Methods Group (2011) The Cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. doi:10.1136/bmj.d5928
Atkins D, Best D, Briss PA, Eccles M, Falck-Ytter Y, Flottorp S, Guyatt GH, Harbour RT, Haugh MC, Henry D, Hill S, Jaeschke R, Leng G, Liberati A, Magrini N, Mason J, Middleton P, Mrukowicz J, O’Connell D, Oxman AD, Phillips B, Schünemann HJ, Edejer T, Varonen H, Vist GE, Williams JW, Zaza S, Group GW (2004) Grading quality of evidence and strength of recommendations. BMJ 328(7454):1490. doi:10.1136/bmj.328.7454.1490
Boekhout AH, Vincent AD, Dalesio OB, Bosch J, Foekema-Töns JH, Adriaansz S, Sprangers S, Nuijen B, Beijnen JH, Schellens JH (2011) Management of hot flashes in patients who have breast cancer with venlafaxine and clonidine: a randomized, double-blind, placebo-controlled trial. J Clin Oncol 29(29):3862–3868. doi:10.1200/jco.2010.33.1298
Bordeleau L, Pritchard KI, Loprinzi CL, Ennis M, Jugovic O, Warr D, Haq R, Goodwin PJ (2010) Multicenter, randomized, cross-over clinical trial of venlafaxine versus gabapentin for the management of hot flashes in breast cancer survivors. J Clin Oncol 28(35):5147–5152. doi:10.1200/jco.2010.29.9230
Buijs C, Mom CH, Willemse PH, Marike Boezen H, Maurer JM, Wymenga AN, Jong RS, Nieboer P, Vries EG, Mourits MJ (2009) Venlafaxine versus clonidine for the treatment of hot flashes in breast cancer patients: a double-blind, randomized cross-over study. Breast Cancer Res Treat 115(3):573–580. doi:10.1007/s10549-008-0138-7
Loibl S, Schwedler K, von Minckwitz G, Strohmeier R, Mehta KM, Kaufmann M (2007) Venlafaxine is superior to clonidine as treatment of hot flashes in breast cancer patients–a double-blind, randomized study. Ann Oncol 18(4):689–693. doi:10.1093/annonc/mdl478
Biglia N, Sgandurra P, Peano E, Marenco D, Moggio G, Bounous V, Tomasi Cont N, Ponzone R, Sismondi P (2009) Non-hormonal treatment of hot flushes in breast cancer survivors: gabapentin vs. vitamin E. Climacteric 12(4):310–318. doi:10.1080/13697130902736921
Maclaughlan David S, Salzillo S, Bowe P, Scuncio S, Malit B, Raker C, Gass JS, Granai CO, Dizon DS (2013) Randomised controlled trial comparing hypnotherapy versus gabapentin for the treatment of hot flashes in breast cancer survivors: a pilot study. BMJ Open 3(9):e003138. doi:10.1136/bmjopen-2013-003138
Othman AH, Zaky AH (2014) Management of hot flushes in breast cancer survivors: comparison between stellate ganglion block and pregabalin. Pain Med 15(3):410–417. doi:10.1111/pme.12331
Walker EM, Rodriguez AI, Kohn B, Ball RM, Pegg J, Pocock JR, Nunez R, Peterson E, Jakary S, Levine RA (2010) Acupuncture versus venlafaxine for the management of vasomotor symptoms in patients with hormone receptor-positive breast cancer: a randomized controlled trial. J Clin Oncol 28(4):634–640. doi:10.1200/jco.2009.23.5150
American Congress of Obstetrics and Gynecology (2012) ACOG practice bulletin no. 126: management of gynecologic issues in women with breast cancer. Obstet Gynecol 119(3):666–682. doi:10.1097/AOG.0b013e31824e12ce
Desmarais JE, Looper KJ (2009) Interactions between tamoxifen and antidepressants via cytochrome P450 2D6. J Clin Psychiatry 70(12):1688–1697. doi:10.4088/JCP.08r04856blu
Bardia A, Novotny P, Sloan J, Barton D, Loprinzi C (2009) Efficacy of nonestrogenic hot flash therapies among women stratified by breast cancer history and tamoxifen use: a pooled analysis. Menopause 16(3):477–483. doi:10.1097/gme.0b013e31818c91ca
Sloan JA, Loprinzi CL, Novotny PJ, Barton DL, Lavasseur BI, Windschitl H (2001) Methodologic lessons learned from hot flash studies. J Clin Oncol 19(23):4280–4290
Curran S, Andrykowski M, Stadts J (1995) Short form of the profile of mood states (POMS-SF): Psychometric information. Psychol Assess 7(1):80–83
National Cancer Institute (2016) Protocol Development: Common Terminology Criteria for Adverse Events (CTCAE). http://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm. Accessed Feb 12 2016
Beck AT, Steer RA, Garbin MG (1988) Psychometric properties of the Beck Depression Inventory: 25 years of evaluation. Clin Psychol Rev 8(1):77–100
Cleeland CS, Mendoza TR, Wang XS, Chou C, Harle MT, Morrissey M, Engstrom MC (2000) Assessing symptom distress in cancer patients: the M.D. Anderson Symptom Inventory. Cancer 89(7):1634–1646
Radloff LS (1977) The CES-D Scale: a self-report depression scale for research in the general population. Appl Psychol Measurement 1:385–401
Zigmond AS, Snaith RP (1983) The Hospital Anxiety and Depression scale. Acta Psychiatr Scand 67(6):361–370
Stewart AL, Ware JE (1992) Measuring Function and Well Being: The Medical Outcomes Study Approach. Duke University Press, Durham
Brooks R (2013) The EuroQOL Group after 25 Years. Springer, Netherlands
Meijman TF, de Vries-Griever AH, de Vries G (1988) The evaluation of the Groningen sleep quality scale. Heymans Bulletin (HB 88-13-EX), Groningen
Thirlaway K, Fallowfield L, Cuzick J (1996) The sexual activity questionnaire: a measure of women’s sexual functioning. Qual Life Res 5(1):81–90
Ware JE, Snow KK, Konsinski M, Gandek B (1993) SF-36 health survey: manual and interpretation guide. The Health Institute, Boston
Zung WW (1965) A self-rating depression scale. Arch Gen Psychiatry 12:63–70
Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ (1989) The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res 28(2):193–213
Heinemann K, Ruebig A, Potthoff P, Schneider HP, Strelow F, Heinemann LA, Do MT (2004) The Menopause Rating Scale (MRS) scale: a methodological review. Health Qual Life Outcomes 2:45. doi:10.1186/1477-7525-2-45
Hilditch JR, Lewis J, Peter A, van Maris B, Ross A, Franssen E, Guyatt GH, Norton PG, Dunn E (1996) A menopause-specific quality of life questionnaire: development and psychometric properties. Maturitas 24(3):161–175
Ware JE, Kosinski M, Keller SD (1996) A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care 34(3):220–233
Acknowledgments
The authors wish to thank Jennifer Ehren, PhD, Diana T. Chingos, MS, MFA, and Michael Krychman, MD, MPH for their contributions to the study. This study was financially supported by the California Breast Cancer Research Program Translational Award 200B-0 144. The funding organization was not involved in the study design, data collection, data analyses, or writing of the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The study complies with the current laws of the United States. The authors declare no conflicts of interest.
Informed consent
This type of study is exempt from institutional review board review, and human consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10549_2016_3765_MOESM2_ESM.png
Supplementary material 2 Risk of bias summaries for trials on A pharmacologic treatment dose comparisons, B two different pharmacological treatments, C pharmacological versus non-pharmacological treatments (PNG 134 kb)
Rights and permissions
About this article
Cite this article
Johns, C., Seav, S.M., Dominick, S.A. et al. Informing hot flash treatment decisions for breast cancer survivors: a systematic review of randomized trials comparing active interventions. Breast Cancer Res Treat 156, 415–426 (2016). https://doi.org/10.1007/s10549-016-3765-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-016-3765-4