Abstract
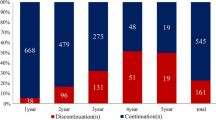
The aim of the study was to analyze the impact of disease management programs (DMPs) on adherence in women with breast cancer (BC) in Germany. Data on 4915 BC patients [1874 DMP and 3041 standard care (SC)] who started hormone therapy between 2008 and 2013 in 234 gynecological practices in Germany were analyzed retrospectively. The primary outcome measure was the rate of discontinuation of hormone therapy within 3 years of the start of treatment. Discontinuation of therapy was defined as a period of at least 90 days without treatment. A multivariate Cox regression model was created to determine the effect of DMPs on the risk of discontinuation. Region (western vs. eastern Germany), patient age, and concomitant diagnoses (depression, osteoporosis, thrombosis, and diabetes) were included as covariates. There was a significant difference between DMPs and SC in terms of age (63 ± 12 years vs. 64 ± 12 years, p value = 0.0012) and region (79.2 % of patients living in western Germany vs. 88.6 %, p value < 0.0001), but not initial therapy (51.8 % vs. 52 %, p value = 0.8696). Depression was also more common in patients in DMPs than those in SC (26.8 % vs. 17.3 %, p value < 0.0001). Within 3 years of therapy initiation, 32.7 % of DMP patients and 39.6 % of SC patients had discontinued their treatment (p < 0.001). Women with BC who were enrolled in a DMP had a lower risk of discontinuing therapy (HR = 0.91, 95 % CI: 0.85–0.98, p value = 0.0092). This risk was also slightly higher in western Germany (HR = 1.13, 95 % CI: 1.02–1.24, p value = 0.0143). Involvement in DMPs has a positive impact on the adherence of BC patients.

Similar content being viewed by others
Abbreviations
- BC:
-
Breast cancer
- DMP:
-
Disease management program
- SC:
-
Standard care
References
World Cancer Research Fund International: Breast cancer statistics (2015). Accessed 13 July 2015
Gemmill M: Research note: Chronic Disease Management in Europe—European Commission [Internet]
Ellrodt G, Cook DJ, Lee J, Cho M, Hunt D, Weingarten S (1997) Evidence-based disease management. JAMA 278:1687–1692
Rupprecht C (2005) First German disease management program for breast cancer. Health Care Financ Rev 27:69–77
World Health Organization: Adherence to long-term therapies: evidence for action [Internet]. 2003
Sackett DL, Haynes RB, Gibson ES, Taylor DW, Roberts RS, Johnson AL (1978) Patient compliance with antihypertensive regimens. Patient Couns Health Educ 1:18–21
Haynes RB, Montague P, Oliver T, McKibbon KA, Brouwers MC, Kanani R (2000) Interventions for helping patients to follow prescriptions for medications. Cochrane Database Syst Rev. doi:10.1002/14651858.CD000011
Balkrishnan R (2005) The importance of medication adherence in improving chronic-disease related outcomes: what we know and what we need to further know. Med Care 43:517–520
Davidson B, Vogel V, Wickerham L (2007) Oncologist-patient discussion of adjuvant hormonal therapy in breast cancer: results of a linguistic study focusing on adherence and persistence to therapy. J Support Oncol 5:139–143
Hadji P, Ziller V, Kyvernitakis J, Bauer M, Haas G, Schmidt N, Kostev K (2013) Persistence in patients with breast cancer treated with tamoxifen or aromatase inhibitors: a retrospective database analysis. Breast Cancer Res Treat 138:185–191. doi:10.1007/s10549-013-2417-1
Becher H, Kostev K, Schröder-Bernhardi D (2009) Validity and representativeness of the “disease analyzer” patient database for use in pharmacoepidemiological and pharmacoeconomic studies. Int J Clin Pharmacol Ther 47:617–626
Masood S (1992) Estrogen and progesterone receptors in cytology: a comprehensive review. Diagn Cytopathol 8:475–491
Burstein HJ, Temin S, Anderson H, Buchholz TA, Davidson NE, Gelmon KE, Giordano SH, Hudis CA, Rowden D, Solky AJ et al (2014) Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: american society of clinical oncology clinical practice guideline focused update. J Clin Oncol Off J Am Soc Clin Oncol 32:2255–2269. doi:10.1200/JCO.2013.54.2258
Matsen CB, Neumayer LA (2013) Breast cancer: a review for the general surgeon. JAMA Surg 148:971–979. doi:10.1001/jamasurg.2013.3393
O’Malley BW, Khan S (2013) Elwood V. Jensen (1920–2012): father of the nuclear receptors. Proc Natl Acad Sci U. S. A. 110:3707–3708. doi:10.1073/pnas.1301566110
Early Breast Cancer Trialists’ Collaborative Group (1998) Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet Lond Engl 351:1451–1467
Jordan VC (2008) Tamoxifen: catalyst for the change to targeted therapy. Eur J Cancer Oxf Engl 44:30–38. doi:10.1016/j.ejca.2007.11.002
Mourits MJE, De Vries EGE, Willemse PHB, Ten Hoor KA, Hollema H, Van der Zee AGJ (2001) Tamoxifen treatment and gynecologic side effects: a review. Obstet Gynecol 97:855–866. doi:10.1016/S0029-7844(00)01196-0
Curtis RE, Boice JD, Shriner DA, Hankey BF, Fraumeni JF (1996) Second cancers after adjuvant tamoxifen therapy for breast cancer. J Natl Cancer Inst 88:832–834
Fisher B, Costantino JP, Redmond CK, Fisher ER, Wickerham DL, Cronin WM (1994) Endometrial cancer in tamoxifen-treated breast cancer patients: findings from the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-14. J Natl Cancer Inst 86:527–537
van Leeuwen FE, Benraadt J, Coebergh JW, Kiemeney LA, Gimbrère CH, Otter R, Schouten LJ, Damhuis RA, Bontenbal M, Diepenhorst FW (1994) Risk of endometrial cancer after tamoxifen treatment of breast cancer. Lancet Lond Engl 343:448–452
Sasco AJ, Chaplain G, Amoros E, Saez S (1996) Endometrial cancer following breast cancer: effect of tamoxifen and castration by radiotherapy. Epidemiol Camb Mass 7:9–13
Partridge AH, Avorn J, Wang PS, Winer EP (2002) Adherence to therapy with oral antineoplastic agents. J Natl Cancer Inst 94:652–661
Fisher B, Dignam J, Bryant J, Wolmark N (2001) Five versus more than five years of tamoxifen for lymph node-negative breast cancer: updated findings from the National Surgical Adjuvant Breast and Bowel Project B-14 randomized trial. J Natl Cancer Inst 93:684–690
Altundag K, Ibrahim NK (2006) Aromatase inhibitors in breast cancer: an overview. Oncologist 11:553–562. doi:10.1634/theoncologist.11-6-553
Evans CT, Ledesma DB, Schulz TZ, Simpson ER, Mendelson CR (1986) Isolation and characterization of a complementary DNA specific for human aromatase-system cytochrome P-450 mRNA. Proc Natl Acad Sci U S. A 83:6387–6391
Nelson LR, Bulun SE (2001) Estrogen production and action. J Am Acad Dermatol 45:S116–S124
Buzdar AU, Coombes RC, Goss PE, Winer EP (2008) Summary of aromatase inhibitor clinical trials in postmenopausal women with early breast cancer. Cancer 112:700–709. doi:10.1002/cncr.23193
Burgess C, Cornelius V, Love S, Graham J, Richards M, Ramirez A (2005) Depression and anxiety in women with early breast cancer: five year observational cohort study. BMJ 330:702. doi:10.1136/bmj.38343.670868.D3
Acknowledgments
Professional English language editing services were provided by Claudia Jones, MA, Radford, Virginia, United States.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Jacob, L., Hadji, P., Albert, US. et al. Impact of disease management programs on women with breast cancer in Germany. Breast Cancer Res Treat 153, 391–395 (2015). https://doi.org/10.1007/s10549-015-3530-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-015-3530-0