Abstract
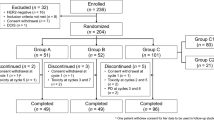
The aim of this study is to estimate the frequency of pathologic complete response (pCR) after neoadjuvant treatment with cisplatin chemotherapy in women with breast cancer and a BRCA1 mutation. One hundred and seven women with breast cancer and a BRCA1 mutation, who were diagnosed with stage I to III breast cancer between December 2006 and June 2014, were treated with cisplatin 75 mg/m2 every 3 weeks for four cycles, followed by mastectomy and conventional chemotherapy. Information was collected on clinical stage, grade, hormone receptor status, and Her2neu status prior to treatment. pCR was determined by review of surgical specimens. One hundred and seven patients were enrolled in the study, including 93 patients who were treated for first primary breast cancer and 14 patients who had previously received treatment for a prior cancer. A pCR was observed in 65 of the 107 patients (61 %). Platinum-based chemotherapy is effective in a high proportion of patients with BRCA1-associated breast cancer.
Similar content being viewed by others
References
Liedtke C, Mazouni C, Hess KR et al (2008) Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol 10:1275–1281
Scully R, Ganesan S, Vlasakova K et al (1999) Genetic analysis of BRCA1 function in a defined tumor cell line. Mol Cell 4:1093–1099
Tassone P, Tagliaferri P, Perricelli A et al (2003) BRCA1 expression modulates chemosensitivity of BRCA1-defective HCC1937 human breast cancer cells. Br J Cancer 88:1285–1291
Lakhani SR, Jacquemier J, Sloane JP et al (1998) Multifactorial analysis of differences between sporadic breast cancers and cancers involving BRCA1 and BRCA2 mutations. J Natl Cancer Inst 90:1138–1145
Foulkes WD, Metcalfe K, Sun P et al (2004) Estrogen receptor status in BRCA1- and BRCA2-related breast cancer: the influence of age, grade, and histological type. Clin Cancer Res 10:2029–2034
Tilanus-Linthorst MM, Obdeijn IM, Hop WC et al (2007) BRCA1 mutation and young age predict fast breast cancer growth in the Dutch, United Kingdom, and Canadian magnetic resonance imaging screening trials. Clin Cancer Res 13:7357–7362
Robson ME, Chappuis PO, Satagopan J et al (2004) A combined analysis of outcome following breast cancer: differences in survival based on BRCA1/BRCA2 mutation status and administration of adjuvant treatment. Breast Cancer Res 6:R8–R17
Rennert G, Bisland-Naggan S, Barnett-Griness O et al (2007) Clinical outcomes of breast cancer in carriers of BRCA1 and BRCA2 mutations. N Engl J Med 357:115–123
Narod SA, Metcalfe K, Lynch HT et al (2013) Should all BRCA1 mutation carriers with stage I breast cancer receive chemotherapy? Breast Cancer Res Treat 138:273–279
Kennedy RD, Quinn JE, Mullan PB, Johnston PG, Harkin DP (2004) The role of BRCA1 in the cellular response to chemotherapy. J Natl Cancer Inst 96:1659–1668
Quinn JE, Kennedy RD, Mullan PB et al (2003) BRCA1 functions as a differential modulator of chemotherapy-induced apoptosis. Cancer Res 6319:6221–6228
Bhattacharyya A, Ear US, Koller BH, Weichselbaum RR, Bishop DK (2000) The breast cancer susceptibility gene BRCA1 is required for subnuclear assembly of Rad51 and survival following treatment with the DNA cross-linking agent cisplatin. J Biol Chem 275:23899–23903
Chabalier C, Lamare C, Racca C, Privat M, Valette A, Larminat F (2003) BRCA1 down-regulation leads to premature inactivation of spindle checkpoint and confers paclitaxel resistance. Cell Cycle 5:1001–1007
Byrski T, Gronwald J, Huzarski T et al (2010) Pathologic complete response rates in young women with BRCA1-positive breast cancers after neoadjuvant chemotherapy. J Clin Oncol 28:375–379
Górski B, Jakubowska A, Huzarski T et al (2004) A high proportion of founder BRCA1 mutations in Polish breast cancer families. Int J Cancer 110:683–686
NCI (2003) National Cancer Institute: Common Terminology Criteria for Adverse Events (CTCAE), version 3, June 10, 2004 update. Bethesda, MD, National Cancer Institute, National Institutes of Health
von Minckwitz G, Schneeweiss A, Loibl S (2014) Neoadjuvant carboplatin in patients with triple-negative and HER2-positive early breast cancer (GeparSixto; GBG 66): a randomised phase 2 trial. Lancet Oncol 15:747–756
Sikov WM, Berry DA, Perou CM, Singh B, et al (2013) Impact of the addition of carboplatin (Cb) and/or bevacizumab (B) to neoadjuvant weekly paclitaxel (P) followed by dose-dense AC on pathologic complete response (pCR) rates in triple-negative breast cancer (TNBC): CALGB 40603 (Alliance). In: San Antoniou Breast Cancer Conference 2013 (abstracts S-501)
Silver DP, Richardson AL, Eklund AC et al (2010) Efficacy of neoadjuvant Cisplatin in triple-negative breast cancer. J Clin Oncol 28:1145–1153
Mazouni C, Kau SW, Frye D et al (2007) Inclusion of taxanes, particularly weekly paclitaxel, in preoperative chemotherapy improves pathologic complete response rate in estrogen receptor-positive breast cancers. Ann Oncol 18:874–880
Huzarski T, Byrski T, Gronwald J et al (2013) Ten-year survival in patients with BRCA1-negative and BRCA1-positive breast cancer. J Clin Oncol 31:3191–3196. doi:10.1200/JCO.2012.45.3571
Acknowledgments
We would like to thank M. Siolek, M. Szwiec, H. Symonowicz, D. Surdyka, O. Ashuryk, B. Gorski, T. Debniak, D. Zuziak, and D. Sawka for helping to recruit patients to this study. We thank W. Domagala for the pathology review. We would also like to thank the Estee Lauder companies for their generous support on this project. The study is registered in the Clinical Trails gov, study number NCT01630226 and was by funded by Polish Ministry of Science; Grant No. PBZ-KBN-122 /po5/ 2004.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Byrski, T., Huzarski, T., Dent, R. et al. Pathologic complete response to neoadjuvant cisplatin in BRCA1-positive breast cancer patients. Breast Cancer Res Treat 147, 401–405 (2014). https://doi.org/10.1007/s10549-014-3100-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-3100-x