Abstract
14-3-3σ is a tumor suppressor gene induced by p53 in response to DNA damage and reportedly associated with resistance to chemotherapy. The aim of this study was to investigate whether 14-3-3σ expression is also associated with resistance to neoadjuvant chemotherapy consisting of paclitaxel followed by 5-FU/epirubicin/cyclophosphamide (P-FEC) in human breast cancer patients. A total of 123 primary breast cancer patients treated with neoadjuvant chemotherapy (P-FEC) were included in this study. Immunohistochemistry of 14-3-3σ and p53 as well as direct sequencing of TP53 were performed using the tumor biopsy samples obtained prior to neoadjuvant chemotherapy. Thirty-eight of the tumors (31%) were positive for 14-3-3σ. There was no significant association between 14-3-3σ expression and TP53 mutation or p53 expression. However, 14-3-3σ expression showed a significantly (P = 0.009) negative association with pathological complete response (pCR) to P-FEC, and multivariate analysis demonstrated that only 14-3-3σ (P = 0.015) and estrogen receptor (P = 0.021) were significantly and independently associated with pCR. The combination of 14-3-3σ expression and TP53 mutation status had an additive negative effect on pCR, i.e., pCR rates were 45.5% for 14-3-3σ negative/TP53 mutant tumors, 24.6% for 14-3-3σ negative/TP53 wild tumors, 23.1% for 14-3-3σ positive/TP53 mutant tumors, and 0% for 14-3-3σ positive/TP53 wild tumors. These results demonstrate that 14-3-3σ expression is significantly associated with resistance to P-FEC and this association is independent of other biological markers. The combination of 14-3-3σ expression and TP53 mutation status has an additively negative effect on the response to P-FEC.


Similar content being viewed by others
References
Mhawech P (2005) 14-3-3 proteins—an update. Cell Res 15(4):228–236
Horie-Inoue K, Inoue S (2006) Epigenetic and proteolytic inactivation of 14-3-3sigma in breast and prostate cancers. Semin Cancer Biol 16(3):235–239
Lee MH, Lozano G (2006) Regulation of the p53-MDM2 pathway by 14-3-3 sigma and other proteins. Semin Cancer Biol 16(3):225–234
Hermeking H, Benzinger A (2006) 14-3-3 proteins in cell cycle regulation. Semin Cancer Biol 16(3):183–192
Simpson PT, Gale T, Reis-Filho JS, Jones C, Parry S, Steele D, Cossu A, Budroni M, Palmieri G, Lakhani SR (2004) Distribution and significance of 14-3-3sigma, a novel myoepithelial marker, in normal, benign, and malignant breast tissue. J Pathol 202(3):274–285
Mirza S, Sharma G, Parshad R, Srivastava A, Gupta SD, Ralhan R (2010) Clinical significance of Stratifin, ERalpha and PR promoter methylation in tumor and serum DNA in Indian breast cancer patients. Clin Biochem 43(4–5):380–386
Ito K, Suzuki T, Akahira J, Sakuma M, Saitou S, Okamoto S, Niikura H, Okamura K, Yaegashi N, Sasano H, Inoue S (2005) 14-3-3sigma in endometrial cancer—a possible prognostic marker in early-stage cancer. Clin Cancer Res 11(20):7384–7391
Cheng L, Pan CX, Zhang JT, Zhang S, Kinch MS, Li L, Baldridge LA, Wade C, Hu Z, Koch MO, Ulbright TM, Eble JN (2004) Loss of 14-3-3sigma in prostate cancer and its precursors. Clin Cancer Res 10(9):3064–3068
Nakayama H, Sano T, Motegi A, Oyama T, Nakajima T (2005) Increasing 14-3-3 sigma expression with declining estrogen receptor alpha and estrogen-responsive finger protein expression defines malignant progression of endometrial carcinoma. Pathol Int 55(11):707–715
Ferguson AT, Evron E, Umbricht CB, Pandita TK, Chan TA, Hermeking H, Marks JR, Lambers AR, Futreal PA, Stampfer MR, Sukumar S (2000) High frequency of hypermethylation at the 14-3-3 sigma locus leads to gene silencing in breast cancer. Proc Natl Acad Sci USA 97(11):6049–6054
Umbricht CB, Evron E, Gabrielson E, Ferguson A, Marks J, Sukumar S (2001) Hypermethylation of 14-3-3 sigma (stratifin) is an early event in breast cancer. Oncogene 20(26):3348–3353
Simooka H, Oyama T, Sano T, Horiguchi J, Nakajima T (2004) Immunohistochemical analysis of 14-3-3 sigma and related proteins in hyperplastic and neoplastic breast lesions, with particular reference to early carcinogenesis. Pathol Int 54(8):595–602
Akahira J, Sugihashi Y, Suzuki T, Ito K, Niikura H, Moriya T, Nitta M, Okamura H, Inoue S, Sasano H, Okamura K, Yaegashi N (2004) Decreased expression of 14-3-3 sigma is associated with advanced disease in human epithelial ovarian cancer: its correlation with aberrant DNA methylation. Clin Cancer Res 10(8):2687–2693
Mhawech P, Benz A, Cerato C, Greloz V, Assaly M, Desmond JC, Koeffler HP, Lodygin D, Hermeking H, Herrmann F, Schwaller J (2005) Downregulation of 14-3-3sigma in ovary, prostate and endometrial carcinomas is associated with CpG island methylation. Mod Pathol 18(3):340–348
Perathoner A, Pirkebner D, Brandacher G, Spizzo G, Stadlmann S, Obrist P, Margreiter R, Amberger A (2005) 14-3-3sigma expression is an independent prognostic parameter for poor survival in colorectal carcinoma patients. Clin Cancer Res 11(9):3274–3279
Neupane D, Korc M (2008) 14-3-3sigma Modulates pancreatic cancer cell survival and invasiveness. Clin Cancer Res 14(23):7614–7623
Tanaka K, Hatada T, Kobayashi M, Mohri Y, Tonouchi H, Miki C, Nobori T, Kusunoki M (2004) The clinical implication of 14-3-3 sigma expression in primary gastrointestinal malignancy. Int J Oncol 25(6):1591–1597
Holm R, Ali T, Svendsrud DH, Nesland JM, Kristensen GB, Lyng H (2009) Expression of 14-3-3sigma in cervical squamous cell carcinomas: relationship with clinical outcome. Oncol Rep 22(1):11–15
Nakajima T, Shimooka H, Weixa P, Segawa A, Motegi A, Jian Z, Masuda N, Ide M, Sano T, Oyama T, Tsukagoshi H, Hamanaka K, Maeda M (2003) Immunohistochemical demonstration of 14-3-3 sigma protein in normal human tissues and lung cancers, and the preponderance of its strong expression in epithelial cells of squamous cell lineage. Pathol Int 53(6):353–360
Logsdon CD, Simeone DM, Binkley C, Arumugam T, Greenson JK, Giordano TJ, Misek DE, Kuick R, Hanash S (2003) Molecular profiling of pancreatic adenocarcinoma and chronic pancreatitis identifies multiple genes differentially regulated in pancreatic cancer. Cancer Res 63(10):2649–2657
Guweidhi A, Kleeff J, Giese N, El Fitori J, Ketterer K, Giese T, Buchler MW, Korc M, Friess H (2004) Enhanced expression of 14-3-3sigma in pancreatic cancer and its role in cell cycle regulation and apoptosis. Carcinogenesis 25(9):1575–1585
Liu Y, Liu H, Han B, Zhang JT (2006) Identification of 14-3-3sigma as a contributor to drug resistance in human breast cancer cells using functional proteomic analysis. Cancer Res 66(6):3248–3255
Han B, Xie H, Chen Q, Zhang JT (2006) Sensitizing hormone-refractory prostate cancer cells to drug treatment by targeting 14-3-3sigma. Mol Cancer Ther 5(4):903–912
Sinha P, Hutter G, Kottgen E, Dietel M, Schadendorf D, Lage H (1999) Increased expression of epidermal fatty acid binding protein, cofilin, and 14-3-3-sigma (stratifin) detected by two-dimensional gel electrophoresis, mass spectrometry and microsequencing of drug-resistant human adenocarcinoma of the pancreas. Electrophoresis 20(14):2952–2960
Gehrmann ML, Fenselau C, Hathout Y (2004) Highly altered protein expression profile in the adriamycin resistant MCF-7 cell line. J Proteome Res 3(3):403–409
Gehrmann ML, Hathout Y, Fenselau C (2004) Evaluation of metabolic labeling for comparative proteomics in breast cancer cells. J Proteome Res 3(5):1063–1068
Moreira JM, Ohlsson G, Rank FE, Celis JE (2005) Down-regulation of the tumor suppressor protein 14-3-3sigma is a sporadic event in cancer of the breast. Mol Cell Proteomics 4(4):555–569
Iwamoto T, Yamamoto N, Taguchi T, Tamaki Y, Noguchi S (2011) BRCA1 promoter methylation in peripheral blood cells is associated with increased risk of breast cancer with BRCA1 promoter methylation. Breast Cancer Res Treat 129(1):69–77
Shimomura A, Miyoshi Y, Taguchi T, Tamaki Y, Noguchi S (2009) Association of loss of BRCA1 expression with centrosome aberration in human breast cancer. J Cancer Res Clin Oncol 135(3):421–430
Tanei T, Morimoto K, Shimazu K, Kim SJ, Tanji Y, Taguchi T, Tamaki Y, Noguchi S (2009) Association of breast cancer stem cells identified by aldehyde dehydrogenase 1 expression with resistance to sequential Paclitaxel and epirubicin-based chemotherapy for breast cancers. Clin Cancer Res 15(12):4234–4241
Bloom HJ, Richardson WW (1957) Histological grading and prognosis in breast cancer; a study of 1409 cases of which 359 have been followed for 15 years. Br J Cancer 11(3):359–377
Oshima K, Naoi Y, Kishi K, Nakamura Y, Iwamoto T, Shimazu K, Nakayama T, Kim SJ, Baba Y, Tamaki Y, Noguchi S (2011) Gene expression signature of TP53 but not its mutation status predicts response to sequential paclitaxel and 5-FU/epirubicin/cyclophosphamide in human breast cancer. Cancer Lett 307(2):149–157
Urano T, Saito T, Tsukui T, Fujita M, Hosoi T, Muramatsu M, Ouchi Y, Inoue S (2002) Efp targets 14-3-3 sigma for proteolysis and promotes breast tumour growth. Nature 417(6891):871–875
Suzuki H, Itoh F, Toyota M, Kikuchi T, Kakiuchi H, Imai K (2000) Inactivation of the 14-3-3 sigma gene is associated with 5′ CpG island hypermethylation in human cancers. Cancer Res 60(16):4353–4357
Wang Z, Trope CG, Suo Z, Troen G, Yang G, Nesland JM, Holm R (2008) The clinicopathological and prognostic impact of 14-3-3 sigma expression on vulvar squamous cell carcinomas. BMC Cancer 8:308
Acknowledgments
This work was supported in part by a grant for the Comprehensive 10-Year Strategy for Cancer Control from the Ministry of Health, Labour and Welfare, Japan.
Conflict of interest
Research grants: S. Noguchi from Pfizer and Bristol-Myers Squibb., Honoraria: S. Noguchi from Pfizer and Bristol-Myers Squibb, S.J. Kim from Bristol-Myers, and T. Nakayama from Pfizer and Bristol-Myers.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10549_2012_1976_MOESM2_ESM.jpg
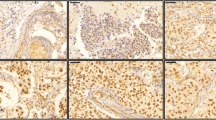
Supplementary Fig. 1 . 14-3-3σ promoter hypermethylation and 14-3-3σ protein expression. Immunohistochemical staining results for six tumors with 14-3-3σ promoter hypermethylation. M, amplification with methylated primers; U, amplification with unmethylated primers. Scale bars: 200 μm. Marker: 200 bp DNA ladder marker. (JPG 147 kb)
Rights and permissions
About this article
Cite this article
Nakamura, Y., Oshima, K., Naoi, Y. et al. 14-3-3σ expression is associated with poor pathological complete response to neoadjuvant chemotherapy in human breast cancers. Breast Cancer Res Treat 134, 229–236 (2012). https://doi.org/10.1007/s10549-012-1976-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-1976-x