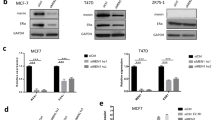
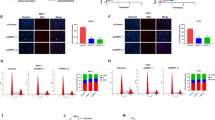
Abstract
E6-associated protein (E6-AP) is a dual function protein. It acts as an E3 ubiquitin-protein ligase enzyme and coactivator of steroid hormone receptors such as estrogen (ERα) and progesterone (PR) receptors. It promotes the degradation of ERα and PR through the ubiquitin–proteasome pathway. Furthermore, it has been shown that the levels of E6-AP are inversely associated with that of ERα in human breast tumors. But the role of wild-type human E6-AP and its ubiquitin-protein ligase activity in mammary tumorigenesis is still unknown. To investigate this role, the authors utilized transgenic mice lines that specifically overexpress either the wild-type human E6-AP (E6-APWT) or the ubiquitin-protein ligase defective E6-AP that contains C833S mutation (E6-APC833S) in the mammary gland. To further substantiate the role of E6-AP in the development of breast tumorigenesis, it was also examined the expression of E6-AP in a large cohort of human breast cancer samples. The transgenic mice that overexpress wild-type E6-AP (E6-APWT) fail to develop mammary tumors. Unlike the E6-APWT mice, the E6-APC833S mice that overexpress ubiquitin-protein ligase defective E6-AP protein develop mammary hyperplasia with a median latency of 18 months. These observations suggest that the inactivation of the ubiquitin-protein ligase function of E6-AP is sufficient to initiate the process of mammary tumor development. Furthermore, the data also suggests that E6-AP exerts its effects on target cells by modulating the protein levels and functions of ERα and PR. In addition, it was found in human breast cancer patients that the level of E6-AP is decreased in invasive breast tumors compared to normal breast tissue. Moreover, the authors also show that the survival patterns for E6-AP negative patients were worse compared to E6-AP positive patients. Taken together, these data suggests that E6-AP may act as a tumor suppressor in breast.






Similar content being viewed by others
Abbreviations
- E6-AP:
-
E6 associated protein
- ER:
-
Estrogen receptor
- PR:
-
Progesterone receptor
- AR:
-
Androgen receptor
- GR:
-
Glucocorticoid receptor
- Hect:
-
Homologus to E6-AP carboxy terminus
- DCIS:
-
Ductal carcinoma in situ
- IC:
-
Invasive carcinoma
- SRA:
-
Steroid receptor RNA activator
- AIB:
-
Amplified in brest cancer
- H&E:
-
Hematoxylin and eosin
- MMTV:
-
Mouse mammary tumor virus
- TAM:
-
Tumor-associated macrophage
References
Scheffner M, Huibregtse JM, Vierstra RD, Howley PM (1993) The HPV-16 E6 and E6-AP complex functions as a ubiquitin-protein ligase in the ubiquitination of p53. Cell 75(3):495–505
Nawaz Z, Lonard DM, Smith CL, Lev-Lehman E, Tsai SY, Tsai MJ, O’Malley BW (1999) The Angelman syndrome-associated protein, E6-AP, is a coactivator for the nuclear hormone receptor superfamily. Mol Cell Biol 19(2):1182–1189
Gao X, Mohsin SK, Gatalica Z, Fu G, Sharma P, Nawaz Z (2005) Decreased expression of e6-associated protein in breast and prostate carcinomas. Endocrinology 146(4):1707–1712
Ramamoorthy S, Dhananjayan SC, Demayo FJ, Nawaz Z (2010) Isoform-specific degradation of PR-B by E6-AP is critical for normal mammary gland development. Mol Endocrinol 24 (11):2099–2113. doi:10.1210/me.2010-0116
Khan OY, Fu G, Ismail A, Srinivasan S, Cao X, Tu Y, Lu S, Nawaz Z (2006) Multifunction steroid receptor coactivator, E6-associated protein, is involved in development of the prostate gland. Mol Endocrinol 20(3):544–559
Huibregtse JM, Scheffner M, Howley PM (1993) Localization of the E6-AP regions that direct human papillomavirus E6 binding, association with p53, and ubiquitination of associated proteins. Mol Cell Biol 13(8):4918–4927
Huibregtse JM, Scheffner M, Beaudenon S, Howley PM (1995) A family of proteins structurally and functionally related to the E6-AP ubiquitin-protein ligase. Proc Natl Acad Sci USA 92(7):2563–2567
Huibregtse JM, Scheffner M, Beaudenon S, Howley PM (1995) A family of proteins structurally and functionally related to the E6-AP ubiquitin-protein ligase. Proc Natl Acad Sci USA 92(11):5249
Kumar S, Kao WH, Howley PM (1997) Physical interaction between specific E2 and Hect E3 enzymes determines functional cooperativity. J Biol Chem 272(21):13548–13554
Conzen SD (2008) Minireview: nuclear receptors and breast cancer. Mol Endocrinol 22(10):2215–2228. doi:10.1210/me.2007-0421
Hewitt SC, Harrell JC, Korach KS (2005) Lessons in estrogen biology from knockout and transgenic animals. Annu Rev Physiol 67:285–308. doi:10.1146/annurev.physiol.67.040403.115914
Green S, Walter P, Greene G, Krust A, Goffin C, Jensen E, Scrace G, Waterfield M, Chambon P (1986) Cloning of the human oestrogen receptor cDNA. J Steroid Biochem 24(1):77–83
Jeltsch JM, Krozowski Z, Quirin-Stricker C, Gronemeyer H, Simpson RJ, Garnier JM, Krust A, Jacob F, Chambon P (1986) Cloning of the chicken progesterone receptor. Proc Natl Acad Sci USA 83(15):5424–5428
Murphy LC, Watson P (2002) Steroid receptors in human breast tumorigenesis and breast cancer progression. Biomed Pharmacother 56(2):65–77
Dimri G, Band H, Band V (2005) Mammary epithelial cell transformation: insights from cell culture and mouse models. Breast Cancer Res 7(4):171–179. doi:10.1186/bcr1275
Fisher ER, Osborne CK, McGuire WL, Redmond C, Knight WA 3rd, Fisher B, Bannayan G, Walder A, Gregory EJ, Jacobsen A, Queen DM, Bennett DE, Ford HC (1981) Correlation of primary breast cancer histopathology and estrogen receptor content. Breast Cancer Res Treat 1(1):37–41
Jones LP, Tilli MT, Assefnia S, Torre K, Halama ED, Parrish A, Rosen EM, Furth PA (2008) Activation of estrogen signaling pathways collaborates with loss of Brca1 to promote development of ERalpha-negative and ERalpha-positive mammary preneoplasia and cancer. Oncogene 27(6):794–802. doi:10.1038/sj.onc.1210674
Herschkowitz JI, Simin K, Weigman VJ, Mikaelian I, Usary J, Hu Z, Rasmussen KE, Jones LP, Assefnia S, Chandrasekharan S, Backlund MG, Yin Y, Khramtsov AI, Bastein R, Quackenbush J, Glazer RI, Brown PH, Green JE, Kopelovich L, Furth PA, Palazzo JP, Olopade OI, Bernard PS, Churchill GA, Van Dyke T, Perou CM (2007) Identification of conserved gene expression features between murine mammary carcinoma models and human breast tumors. Genome Biol 8(5):R76. doi:10.1186/gb-2007-8-5-r76
Knight WA, Livingston RB, Gregory EJ, McGuire WL (1977) Estrogen receptor as an independent prognostic factor for early recurrence in breast cancer. Cancer Res 37(12):4669–4671
Osborne CK, Fisher E, Redmond C, Knight WA, Yochmowitz MG, McGuire WL (1981) Estrogen receptor, a marker for human breast cancer differentiation and patient prognosis. Adv Exp Med Biol 138:377–385
Kleinberg DL, Wood TL, Furth PA, Lee AV (2009) Growth hormone and insulin-like growth factor-I in the transition from normal mammary development to preneoplastic mammary lesions. Endocr Rev 30(1):51–74. doi:10.1210/er.2008-0022
Soyal S, Ismail PM, Li J, Mulac-Jericevic B, Conneely OM, Lydon JP (2002) Progesterone’s role in mammary gland development and tumorigenesis as disclosed by experimental mouse genetics. Breast Cancer Res 4(5):191–196
Osborne CK, Yochmowitz MG, Knight WA 3rd, McGuire WL (1980) The value of estrogen and progesterone receptors in the treatment of breast cancer. Cancer 46(12 Suppl):2884–2888
Knight WA 3rd, Osborne CK, McGuire WL (1980) Hormone receptors in primary and advanced breast cancer. Clin Endocrinol Metab 9(2):361–368
Tilli MT, Frech MS, Steed ME, Hruska KS, Johnson MD, Flaws JA, Furth PA (2003) Introduction of estrogen receptor-alpha into the tTA/TAg conditional mouse model precipitates the development of estrogen-responsive mammary adenocarcinoma. Am J Pathol 163(5):1713–1719
Howard B, Ashworth A (2006) Signalling pathways implicated in early mammary gland morphogenesis and breast cancer. PLoS Genet 2(8):e112. doi:10.1371/journal.pgen.0020112
Gao X, Loggie BW, Nawaz Z (2002) The roles of sex steroid receptor coregulators in cancer. Mol Cancer 1:7
Sivaraman L, Nawaz Z, Medina D, Conneely OM, O’Malley BW (2000) The dual function steroid receptor coactivator/ubiquitin protein-ligase integrator E6-AP is overexpressed in mouse mammary tumorigenesis. Breast Cancer Res Treat 62(3):185–195
Kerscher O, Felberbaum R, Hochstrasser M (2006) Modification of proteins by ubiquitin and ubiquitin-like proteins. Annu Rev Cell Dev Biol 22:159–180. doi:10.1146/annurev.cellbio.22.010605.093503
Li M, Brooks CL, Wu-Baer F, Chen D, Baer R, Gu W (2003) Mono- versus polyubiquitination: differential control of p53 fate by Mdm2. Science 302(5652):1972–1975
Oda H, Kumar S, Howley PM (1999) Regulation of the Src family tyrosine kinase Blk through E6AP-mediated ubiquitination. Proc Natl Acad Sci USA 96(17):9557–9562
Dornan D, Wertz I, Shimizu H, Arnott D, Frantz GD, Dowd P, O’Rourke K, Koeppen H, Dixit VM (2004) The ubiquitin ligase COP1 is a critical negative regulator of p53. Nature 429(6987):86–92
Cooper B, Schneider S, Bohl J, Jiang Y, Beaudet A, Vande Pol S (2003) Requirement of E6AP and the features of human papillomavirus E6 necessary to support degradation of p53. Virology 306(1):87–99
Lange CA, Shen T, Horwitz KB (2000) Phosphorylation of human progesterone receptors at serine-294 by mitogen-activated protein kinase signals their degradation by the 26S proteasome. Proc Natl Acad Sci USA 97(3):1032–1037
Nawaz Z, Lonard DM, Dennis AP, Smith CL, O’Malley BW (1999) Proteasome-dependent degradation of the human estrogen receptor. Proc Natl Acad Sci USA 96(5):1858–1862
Nawaz Z, O’Malley BW (2004) Urban renewal in the nucleus: is protein turnover by proteasomes absolutely required for nuclear receptor-regulated transcription? Mol Endocrinol 18(3):493–499. doi:10.1210/me.2003-0388
Fang S, Lorick KL, Jensen JP, Weissman AM (2003) RING finger ubiquitin protein ligases: implications for tumorigenesis, metastasis and for molecular targets in cancer. Semin Cancer Biol 13(1):5–14
Ohta T, Fukuda M (2004) Ubiquitin and breast cancer. Oncogene 23(11):2079–2088. doi:10.1038/sj.onc.1207371
Acknowledgments
The authors would like to acknowledge Sarath Dhananjayan and Sathish Srinivasan for critically reading the manuscript. This research was supported by the Braman Family Breast Cancer Institute, Sylvester Comprehensive Cancer Center-University of Miami and Bankhead Coley (BK-Prespore09BR-02).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramamoorthy, S., Tufail, R., Hokayem, J.E. et al. Overexpression of ligase defective E6-associated protein, E6-AP, results in mammary tumorigenesis. Breast Cancer Res Treat 132, 97–108 (2012). https://doi.org/10.1007/s10549-011-1567-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-011-1567-2