Abstract
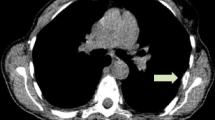
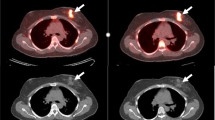
Purpose To determine if response to endocrine therapy of breast cancer can be predicted by either a metabolic “flare reaction” detected by positron emission tomography (PET) with 2-[18F]-fluoro-2-deoxyglucose (FDG), induced by an estradiol challenge, or by estrogen-receptor (ER) status, determined by PET with the estrogen analog 16α-[18F]fluoroestradiol-17β (FES). Methods Fifty-one post-menopausal women with advanced estrogen-receptor positive breast cancer were studied. Patients underwent FES-PET and FDG-PET at baseline and repeat FDG-PET after 30 mg estradiol. Tracer uptakes was measured as the standardized uptake value (SUV). Patients were subsequently treated with either an aromatase inhibitor or fulvestrant. A prospectively defined cut-off SUV ≥ 2 for FES was considered positive for ER expression. A cutoff of ≥12% increase in SUV for FDG, determined by ROC analysis, represented metabolic flare. PET results were correlated with responsiveness to endocrine therapy. Results Seventeen patients responded and 34 patients did not respond to endocrine therapy. Four responders and one non-responder had a clinical flare reaction, while only the responders demonstrated metabolic flare. After estradiol challenge, a significantly higher mean (±SD) percent change in SUV for FDG was noted in responders (20.9 ± 24.2) compared with non-responders (−4.3 ± 11.0, P < 0.0001). On FES-PET, a higher tumor SUV was noted in responders (3.5 ± 2.5) compared with non-responders (2.1 ± 1.8, P = 0.0049). There was significantly longer overall survival in patients with metabolic flare than in those without flare regardless of type of endocrine therapy (P = 0.0062). Conclusion Baseline tumor FES uptake and metabolic flare after an estradiol challenge are both predictive of responsiveness to endocrine therapy in ER+ breast cancer.



Similar content being viewed by others
References
Mouridsen H, Gershanovich M, Sun Y et al (2001) Superior efficacy of letrozole versus tamoxifen as first-line therapy for postmenopausal women with advanced breast cancer: results of a phase III study of the International Letrozole Breast Cancer Group. J Clin Oncol 19:2596–2606
Howell A, Robertson JF, Quaresma Albano J et al (2002) Fulvestrant, formerly ICI 182,780, is as effective as anastrozole in postmenopausal women with advanced breast cancer progressing after prior endocrine treatment. J Clin Oncol 20:3396–3403
Kaufmann M, Bajetta E, Dirix LY et al (2000) Exemestane is superior to megestrol acetate after tamoxifen failure in postmenopausal women with advanced breast cancer: results of a phase III randomized double-blind trial. The Exemestane Study Group. J Clin Oncol 18:1399–1411
Fan C, Oh DS, Wessels L et al (2006) Concordance among gene-expression-based predictors for breast cancer. N Engl J Med 355:560–569
Mintun MA, Welch MJ, Siegel BA et al (1988) Breast cancer: PET imaging of estrogen receptors. Radiology 169:45–48
McGuire AH, Dehdashti F, Siegel BA et al (1991) Positron tomographic assessment of 16 alpha-[18F] fluoro-17 beta-estradiol uptake in metastatic breast carcinoma. J Nucl Med 32:1526–1531
Dehdashti F, Mortimer JE, Siegel BA et al (1995) Positron tomographic assessment of estrogen receptors in breast cancer: comparison with FDG-PET and in vitro receptor assays. J Nucl Med 36:1766–1774
Mortimer JE, Dehdashti F, Siegel BA et al (1996) Positron emission tomography with 2-[18F]Fluoro-2-deoxy-d-glucose and 16alpha-[18F]fluoro-17beta-estradiol in breast cancer: correlation with estrogen receptor status and response to systemic therapy. Clin Cancer Res 2:933–939
Dehdashti F, Flanagan FL, Mortimer JE et al (1999) Positron emission tomographic assessment of “metabolic flare” to predict response of metastatic breast cancer to antiestrogen therapy. Eur J Nucl Med 26:51–56
Mortimer JE, Dehdashti F, Siegel BA et al (2001) Metabolic flare: indicator of hormone responsiveness in advanced breast cancer. J Clin Oncol 19:2797–2803
Wilson RE, Jessiman AG, Moore FD (1958) Severe exacerbation of cancer of the breast after oophorectomy and adrenalectomy: report of four cases. N Engl J Med 258:312–317
Plotkin D, Lechner JJ, Jung WE et al (1978) Tamoxifen flare in advanced breast cancer. JAMA 240:2644–2646
Vogel CL, Schoenfelder J, Shemano I et al (1995) Worsening bone scan in the evaluation of antitumor response during hormonal therapy of breast cancer. J Clin Oncol 13:1123–1128
Coleman RE, Mashiter G, Whitaker KB et al (1988) Bone scan flare predicts successful systemic therapy for bone metastases. J Nucl Med 29:1354–1359
Coleman RE, Whitaker KB, Moss DW et al (1988) Biochemical prediction of response of bone metastases to treatment. Br J Cancer 58:205–210
Legha SS, Powell K, Buzdar AU et al (1981) Tamoxifen-induced hypercalcemia in breast cancer. Cancer 47:2803–2806
Goss PE, Strasser K (2001) Aromatase inhibitors in the treatment and prevention of breast cancer. J Clin Oncol 19:881–894
Brodack JW, Kilbourn MR, Welch MJ et al (1986) Application of robotics to radiopharmaceutical preparation: controlled synthesis of fluorine-18 16 alpha-fluoroestradiol-17 beta. J Nucl Med 27:714–721
Toorongian SA, Mulholland GK, Jewett DM et al (1990) Routine production of 2-deoxy-2-[18F]fluoro-d-glucose by direct nucleophilic exchange on a quaternary 4-aminopyridinium resin. Int J Rad Appl Instrum B 17:273–279
Wienhard K, Dahlbom M, Eriksson L et al (1994) The ECAT EXACT HR: performance of a new high resolution positron scanner. J Comput Assist Tomogr 18:110–118
Wienhard K, Eriksson L, Grootoonk S et al (1992) Performance evaluation of the positron scanner ECAT EXACT. J Comput Assist Tomogr 16:804–813
Adam LE, Zaers J, Ostertag H et al (1997) Performance evaluation of the whole-body PET scanner ECAT EXAT HR+ following the IEC standard. IEEE Trans Nucl Sci 44:1172–1179
Xu M, Luk WK, Cutler PD et al (1994) Local threshold for segmented attenuation correction of PET imaging of the thorax. IEEE Trans Nucl Sci 41:1532–1537
Furman E, Rushkin E, Margalit R et al (1992) Tamoxifen induced changes in MCF7 human breast cancer: in vitro and in vivo studies using nuclear magnetic resonance spectroscopy and imaging. J Steroid Biochem Mol Biol 43:189–195
Shinkarenko L, Kaye AM, Degani H (1994) 13C NMR kinetic studies of the rapid stimulation of glucose metabolism by estrogen in immature rat uterus. NMR Biomed 7:209–217
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Kubota K, Matsuzawa T, Ito M et al (1985) Lung tumor imaging by positron emission tomography using C-11 l-methionine. J Nucl Med 26:37–42
Linden HM, Stekhova SA, Link JM et al (2006) Quantitative fluoroestradiol positron emission tomography imaging predicts response to endocrine treatment in breast cancer. J Clin Oncol 24:2793–2799
Jordan VC, Lewis JS, Osipo C et al (2005) The apoptotic action of estrogen following exhaustive antihormonal therapy: a new clinical treatment strategy. Breast 14:624–630
Nathanson IT, Kelley RM (1952) Hormonal treatment of cancer. N Engl J Med 246:135–145
Kimmick G, Muss HB (1995) Current status of endocrine therapy for metastatic breast cancer. Oncology (Williston Park) 9:877–886, 889–890; discussion 892–874
Schiff R, Massarweh S, Shou J et al (2003) Breast cancer endocrine resistance: how growth factor signaling and estrogen receptor coregulators modulate response. Clin Cancer Res 9:447S–454S
Acknowledgment
This work was supported by Komen Foundation, AstraZeneca, Department of Energy DE FG02 84ER60218 and PHS R01 CA25836.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dehdashti, F., Mortimer, J.E., Trinkaus, K. et al. PET-based estradiol challenge as a predictive biomarker of response to endocrine therapy in women with estrogen-receptor-positive breast cancer. Breast Cancer Res Treat 113, 509–517 (2009). https://doi.org/10.1007/s10549-008-9953-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-008-9953-0