Abstract
Background
Estrogen receptor (ER) expression is routinely assessed by immunohistochemistry (IHC) in breast carcinoma. Our study compares visual scoring of ER in invasive breast cancer by histopathologists to quantitation of staining using a fully automated system.
Materials and methods
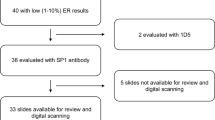
A tissue microarray was constructed from 4,049 cases (3,484 included in analysis) of invasive breast carcinoma linked to treatment and outcome information. Slides were scored independently by two pathologists and scores were dichotomised, with ER positivity recognized at a cut-off of >1% positive nuclei. The slides were scanned and analyzed with an Ariol automated system.
Results
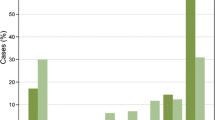
Using data dichotomised as ER positive or negative, both visual and automated scores were highly consistent: there was excellent concordance between two pathologists (kappa = 0.918 (95%CI: 0.903–0.932)) and between two Ariol machines (kappa = 0.913 (95%CI: 0.897–0.928)). The prognostic significance of ER positivity was similar whether determined by pathologist or automated scoring for both the entire patient cohort and subsets of patients treated with tamoxifen alone or receiving no systemic adjuvant therapy. The optimal cut point for the automated scores using breast cancer disease-specific survival as an endpoint was >0.4% positive nuclei. The concordance between dextran-coated charcoal ER biochemical assay data and automated scores (kappa = 0.728 (95%CI: 0.69–0.75); 0.74 (95%CI: 0.71–0.77)) was similar to the concordance between biochemical assay and pathologist scores (kappa = 0.72 (95%CI: 0.70–0.75; 0.70 (95%CI: 0.67–0.72)).
Conclusion
Fully automated quantitation of ER immunostaining yields results that do not differ from human scoring against both biochemical assay and patient outcome gold standards.




Similar content being viewed by others
References
Duffy MJ (2006) Estrogen receptors: role in breast cancer. Crit Rev Clin Lab Sci 43:325–347
Cheang MC, Treaba DO, Speers CH et al (2006) Immunohistochemical detection using the new rabbit monoclonal antibody SP1 of estrogen receptor in breast cancer is superior to mouse monoclonal antibody 1D5 in predicting survival. J Clin Oncol 24:5637–5644
Bolger N, Heffron C, Regan I et al (2006) Implementation and evaluation of a new automated interactive image analysis system. Acta Cytol 50:483–491
Camp RL, Dolled-Filhart M, Rimm DL (2004) X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res 10:7252–7259
Taylor CR, Levenson RM (2006) Quantification of immunohistochemistry-issues concerning methods, utility and semiquantitative assessment II. Histopathology 49:411–424
Walker RA (2006) Quantification of immunohistochemistry-issues concerning methods, utility and semiquantitative assessment I. Histopathology 49:406–410
BVD/FOGRA (1992) Manual for standardization of the offset printing process. Wiesbaden
Insight into images: principles and practices for segmentation, registration, and image analysis (2004). A.K. Peters Ltd., Wellesey, MA
Byrne A, Hilbert DR (2003) Color realism and color science. Behav Brain Sci 26:3–21; discussion 22–63
Gegenfurtner KR (2003) Cortical mechanisms of colour vision. Nat Rev Neurosci 4:563–572
McLelland D, Fuller LU (2005) Photoshop CS2 bible. Wiley Publishing Inc., Hoboken, NJ
Rinner O, Gegenfurtner KR (2000) Time course of chromatic adaptation for color appearance and discrimination. Vision Res 40:1813–1826
Wen C-H, Lee J-J (2000) Design and production of color calibration targets for digital input devices. In: Input/output and imaging technologies II:4080. Taipei, Taiwan, pp 148–158
Greene GL, Nolan C, Engler JP et al (1980) Monoclonal antibodies to human estrogen receptor. Proc Natl Acad Sci USA 77:5115–5119
King WJ, Greene GL (1984) Monoclonal antibodies localize oestrogen receptor in the nuclei of target cells. Nature 307:745–747
Underwood JC (1983) Oestrogen receptors in human breast cancer: review of histopathological correlations and critique of histochemical methods. Diagn Histopathol 6:1–22
Aziz DC, Barathur RB (1994) Quantitation and morphometric analysis of tumors by image analysis. J Cell Biochem Suppl 19:120–125
Esteban JM, Ahn C, Battifora H et al (1994) Quantitative immunohistochemical assay for hormonal receptors: technical aspects and biological significance. J Cell Biochem Suppl 19:138–145
Schultz DS, Katz RL, Patel S et al (1992) Comparison of visual and CAS-200 quantitation of immunocytochemical staining in breast carcinoma samples. Anal Quant Cytol Histol 14:35–40
Kononen J, Bubendorf L, Kallioniemi A et al (1998) Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 4:844–847
Makretsov N, Gilks CB, Coldman AJ et al (2003) Tissue microarray analysis of neuroendocrine differentiation and its prognostic significance in breast cancer. Hum Pathol 34:1001–1008
Turbin DA, Cheang MC, Bajdik CD et al (2006) MDM2 protein expression is a negative prognostic marker in breast carcinoma. Mod Pathol 19:69–74
Liu CL, Prapong W, Natkunam Y et al (2002) Software tools for high-throughput analysis and archiving of immunohistochemistry staining data obtained with tissue microarrays. Am J Pathol 161:1557–1565
Liu CL, Montgomery KD, Natkunam Y et al (2005) TMA-Combiner, a simple software tool to permit analysis of replicate cores on tissue microarrays. Mod Pathol 18:1641–1648
Ng TL, Gown AM, Barry TS et al (2005) Nuclear beta-catenin in mesenchymal tumors. Mod Pathol 18:68–74
de las Mulas JM, van Niel M, Millan Y et al (2000) Immunohistochemical analysis of estrogen receptors in feline mammary gland benign and malignant lesions: comparison with biochemical assay. Domest Anim Endocrinol 18:111–125
Magne N, Toillon RA, Castadot P et al (2006) Different clinical impact of estradiol receptor determination according to the analytical method: A study on 1940 breast cancer patients over a period of 16 consecutive years. Breast Cancer Res Treat 95:179–184
Costa SD, Lange S, Klinga K et al (2002) Factors influencing the prognostic role of oestrogen and progesterone receptor levels in breast cancer – results of the analysis of 670 patients with 11 years of follow-up. Eur J Cancer 38:1329–1334
Franklin WA, Bibbo M, Doria MI et al (1987) Quantitation of estrogen receptor content and Ki-67 staining in breast carcinoma by the microTICAS image analysis system. Anal Quant Cytol Histol 9:279–286
Gil J, Wu HS (2003) Applications of image analysis to anatomic pathology: realities and promises. Cancer Invest 21:950–959
Rojo MG, Garcia GB, Mateos CP et al (2006) Critical comparison of 31 commercially available digital slide systems in pathology. Int J Surg Pathol 14:285–305
Cregger M, Berger AJ, Rimm DL (2006) Immunohistochemistry and quantitative analysis of protein expression. Arch Pathol Lab Med 130:1026–1030
McCabe A, Dolled-Filhart M, Camp RL et al (2005) Automated quantitative analysis (AQUA) of in situ protein expression, antibody concentration, and prognosis. J Natl Cancer Inst 97:1808–1815
Rimm DL (2006) What brown cannot do for you. Nat Biotechnol 24:914–916
Harvey JM, Clark GM, Osborne CK et al (1999) Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol 17:1474–1481
Diaz LK, Sahin A, Sneige N (2004) Interobserver agreement for estrogen receptor immunohistochemical analysis in breast cancer: a comparison of manual and computer-assisted scoring methods. Ann Diagn Pathol 8:23–27
Hasegawa T, Yamamoto S, Matsuno Y (2002) Quantitative immunohistochemical evaluation of MIB-1 labeling index in adult soft-tissue sarcomas by computer-assisted image analysis. Pathol Int 52:433–437
Kirkegaard T, Edwards J, Tovey S et al (2006) Observer variation in immunohistochemical analysis of protein expression, time for a change? Histopathology 48:787–794
Lorinc E, Jakobsson B, Landberg G et al (2005) Ki67 and p53 immunohistochemistry reduces interobserver variation in assessment of Barrett’s oesophagus. Histopathology 46:642–648
Acknowledgements
The study was supported in part by an unrestricted educational grant from Sanofi-Aventis, Canada and a Translational Acceleration grant from the Canadian Breast Cancer Research Alliance. TON and DGH are scholars of Michael Smith Foundation for Health Research.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary figure
(PDF 118 kb)
Rights and permissions
About this article
Cite this article
Turbin, D.A., Leung, S., Cheang, M.C.U. et al. Automated quantitative analysis of estrogen receptor expression in breast carcinoma does not differ from expert pathologist scoring: a tissue microarray study of 3,484 cases. Breast Cancer Res Treat 110, 417–426 (2008). https://doi.org/10.1007/s10549-007-9736-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9736-z