Abstract
Background
In the primary core analysis of BIG 1–98, a randomized, double-blind trial comparing 5 years of initial adjuvant therapy with letrozole versus tamoxifen in postmenopausal women with hormone receptor-positive (HR+) early breast cancer, letrozole significantly improved disease-free survival by 19% and reduced the risk of breast cancer recurrence by 28% and distant recurrence by 27%.
Methods
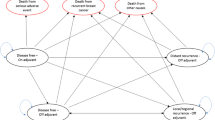
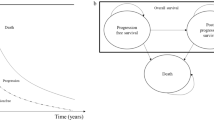
A Markov model was used to estimate the incremental cost per quality-adjusted life year (QALY) gained with 5 years of initial adjuvant therapy with letrozole versus tamoxifen from a Canadian healthcare system perspective. Probabilities of recurrence and side effects for tamoxifen were based on published results of BIG 1–98 and other published population-based studies. Corresponding probabilities for letrozole were calculated by multiplying probabilities for tamoxifen by estimated relative risks for letrozole versus tamoxifen from BIG 1–98. Other probabilities, costs of breast-cancer care and treatment of side effects, and health-state utilities were obtained from published studies. Costs and QALYs were estimated over the lifetime of a cohort of postmenopausal women with HR+ early breast cancer, aged 60 years at initiation of therapy, and discounted at 5% annually.
Results
Compared with tamoxifen, letrozole yields an additional 0.368 life-years (12.453 vs. 12.086) and 0.343 QALYs (11.582 vs. 11.239). These benefits are obtained at an additional cost of $Can 8,110 ($Can 30,819 vs. $Can 22,709). Cost per QALY gained for letrozole versus tamoxifen is $Can 23,662 (95% CI $Can 15,667–$Can 52,014).
Conclusion
In postmenopausal women with HR+ early breast cancer, initial adjuvant treatment with letrozole is cost-effective from the Canadian healthcare system perspective.




Similar content being viewed by others
References
Early Breast Cancer Trialists’ Collaborative Group (1998) Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 351:1451–1467
Mouridsen H, Gershanovich M, Sun Y, Perez-Carrion R, Boni C, Monnier A, Apffelstaedt J, Smith R, Sleeboom HP, Jaenicke F, Pluzanska A, Dank M, Becquart D, Bapsy PP, Salminen E, Snyder R, Chaudri-Ross H, Lang R, Wyld P, Bhatnagar A (2003) Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the International Letrozole Breast Cancer Group. J Clin Oncol 21(11):2101–2109
Goss PE, Ingle JN, Martino S, Robert NJ, Muss HB, Piccart MJ, Castiglione M, Tu D, Shepherd LE, Pritchard KI, Livingston RB, Davidson NE, Norton L, Perez EA, Abrams JS, Cameron DA, Palmer MJ, Pater JL (2005) Randomized trial of letrozole following tamoxifen as extended adjuvant therapy in receptor-positive breast cancer: updated findings from NCIC CTG MA.17. J Natl Cancer Inst 97:1262–1271
Ellis MJ, Coop A, Singh B, Mauriac L, Llombert-Cussac A, Janicke F, Miller WR, Evans DB, Dugan M, Brady C, Quebe-Fehling E, Borgs M (2001) Letrozole is more effective neoadjuvant endocrine therapy than tamoxifen for ErbB-1- and/or ErbB-2-positive, estrogen receptor-positive primary breast cancer: evidence from a phase III randomized trial. J Clin Oncol 19(18):3808–3816
The Breast International Group (BIG) 1–98 Collaborative Group (2005) A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med 353(26):2747–2757
Coates AS, Keshaviah A, Thurlimann B, Mouridsen H, Mauriac L, Forbes JF, Paridaens R, Castiglione-Gertsch M, Gelber RD, Colleoni M, Lang I, Del Mastro L, Smith I, Chirgwin J, Nogaret JM, Pienkowski T, Wardley A, Jakobsen EH, Price KN, Goldhirsch A (2007) Five years of letrozole compared with tamoxifen as initial adjuvant therapy for postmenopausal women with endocrine-responsive early breast cancer: update of study BIG 1–98. J Clin Oncol 25(5):486–492 Epub January 2
Weinstein MC, Toy EL, Sandberg EA, Neumann PJ, Evans JS, Kuntz KM, Graham JD, Hammitt JK (2001) Modeling for health care and other policy decisions: uses, roles, and validity. Value Health 4(5):348–361
Goldie SJ (2003) Chapter 15: Public health policy and cost-effectiveness analysis. J Natl Cancer Inst Monogr (31):102–110
Sonnenberg FA, Beck JR (1993) Markov models in medical decision making: a practical guide. Med Decis Making 13(4):322–338
Weinstein MC, O’Brien B, Hornberger J, Jackson J, Johannesson M, McCabe C, Luce BR (2003) ISPOR Task Force on Good Research Practices-Modeling Studies: principles of good practice for decision analytic modeling in health-care evaluation: report of the ISPOR Task Force on Good Research Practices-Modeling Studies. Value Health 6:9–17
Statistics US: Table 326–0002—Consumer price index (CPI), 2001 basket content, annual. Available at: http: //www.statcan.ca. Accessed April 4, 2006
Karnon J, Delea T, Johnston SR, Smith R, Brandman J, Sung J, Goss PE (2006) Cost effectiveness of extended adjuvant letrozole in postmenopausal women after adjuvant tamoxifen therapy: the UK perspective. Pharmacoeconomics 24(3):237–250
Howell A, on behalf of the ATAC Trialists’ Group (2004) The ATAC (‘Arimidex’, Tamoxifen, Alone or in Combination) trial in postmenopausal women with early breast cancer—updated efficacy results based on a median follow-up of 5 years. Abstract No. 1, San Antonio Breast Cancer Symposium
Rocchi A, Verma S (2006) Anastrozole is cost-effective vs tamoxifen as initial adjuvant therapy in early breast cancer: Canadian perspectives on the ATAC completed-treatment analysis. Support Care Cancer 14(9):917–927. Epub ahead of print
Locker GY, on behalf of the ATAC Trialists’ Group (2004) Cost-utility analysis of anastrozole versus tamoxifen as primary adjuvant therapy in postmenopausal women with early breast cancer from a US healthcare system perspective: the 5-year completed treatment analysis of the ATAC (‘Arimidex’, Tamoxifen Alone or in Combination) trial. 27th Annual San Antonio Breast Cancer Symposium, December 8–11, 2004. Abstract 2085
Moran MS, Haffty BG (2002) Local-regional breast cancer recurrence: prognostic groups based on patterns of failure. Breast J 8(2):81–87
Doyle T, Schultz DJ, Peters C, Harris E, Solin LJ (2001) Long-term results of local recurrence after breast conservation treatment for invasive breast cancer. Int J Radiat Oncol Biol Phys 51(1):74–80
Haylock BJ, Coppin CML, Jackson J, Basco VE, Wilson KS (2000) Locoregional first recurrence after mastectomy: prospective cohort studies with and without immediate chemotherapy. Int J Radiat Oncol Biol Phys 46(2):355–362
Schmoor C, Sauerbrei W, Bastert G, Schumacher M (2000) Role of isolated 2000 locoregional recurrence of breast cancer: results of four prospective studies. J Clin Oncol 18(8):1696–1708
Kamby C, Sengelov L (1997) Pattern of dissemination and survival following isolated locoregional recurrence of breast cancer. Breast Cancer Res Treat 45:181–192
Borner M, Bacchi M, Goldhirsch A, Greiner R, Harder F, Castiglione M, Jungi WF, Thurlimann B, Cavalli F, Obrecht JP et al (1994) First isolated locoregional recurrence following mastectomy for breast cancer: results of a phase III multicenter study comparing systemic treatment with observation after excision and radiation.J Clin Oncol 12:2071–2077
Toonkel LM, Fix I, Jacobsen LH, Wallach CB (1983) The significance of local recurrence of carcinoma of the breast. Int J Radiat Oncol Biol Phys 9:33–39
Koning C, Hart G (1998) Long-term follow-up of a randomized trial on adjuvant chemotherapy and hormonal therapy in locally advanced breast cancer. Int J Radiat Oncol Biol Phys 41(2):397–400
National Cancer Institute: U.S. National Institutes of Health (2006) SEER Public Use Data, 1973–2002. Available at: http: //www.seer.cancer.gov. Accessed April 4, 2006
Silverstein MD, Heit JA, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ (1998) 3rd: Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med 158(6):585–593
Senni M, Tribouilloy CM, Rodeheffer RJ, Jacobsen SJ, Evans JM, Bailey KR, Redfield MM (1999) Congestive heart failure in the community: trends in incidence and survival in a 10-year period. Arch Intern Med 159(1):29–34
Melton LJ 3rd, Amadio PC, Crowson CS, O’Fallon WM (1998) Long-term trends in the incidence of distal forearm fractures. Osteoporos Int 8(4):341–348
Greenlee RT, Naleway AL, Vidaillet H (2002) Incidence of myocardial infarction in a general population: the Marshfield Epidemiologic Study Area. WMJ 101(7):46–52
American Heart Association: Heart Disease and Stroke Statistics (2005) Update. American Heart Association, Dallas, Texas
Eisenstein EL, Shaw LK, Anstrom KJ, Nelson CL, Hakim Z, Hasselblad V, Mark DB (2001) Assessing the clinical and economic burden of coronary artery disease: 1986–1998. Med Care 39(8):824–835
Fisher B, Costantino JP, Wickerham DL, Cecchini RS, Cronin WM, Robidoux A, Bevers TB, Kavanah MT, Atkins JN, Margolese RG, Runowicz CD, James JM, Ford LG, Wolmark N (1998) Tamoxifen for prevention of breast cancer: Report of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Natl Cancer Inst 90:1371–1388
Braithwaite RS, Chlebowski RT, Lau J, George S, Hess R, Col NF (2003) Meta-analysis of vascular and neoplastic events associated with tamoxifen. J Gen Intern Med 18(11):937–947
Statistics Canada: Canadian Vital Statistics, Death Database. Available at: http: //www.statcan.ca. Accessed April 4, 2006
Madison T, Schottenfeld D, James SA, Schwartz AG, Gruber SB (2004) Endometrial cancer: socioeconomic status and racial/ethnic differences in stage at diagnosis, treatment, and survival. Am J Public Health 94(12):2104–2111
Elting LS, Escalante CP, Cooksley C, Avritscher EB, Kurtin D, Hamblin L, Khosla SG, Rivera E (2004) Outcomes and cost of deep venous thrombosis among patients with cancer. Arch Intern Med 164(15):1653–1661
Heit JA, Silverstein MD, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ (1999) 3rd: Predictors of survival after deep vein thrombosis and pulmonary embolism: a population-based, cohort study. Arch Intern Med 159(5):445–453
Roger VL, Jacobsen SJ, Weston SA, Goraya TY, Killian J, Reeder GS, Kottke TE, Yawn BP, Frye RL (2002) Trends in the incidence and survival of patients with hospitalized myocardial infarction, Olmsted County, Minnesota, 1979 to 1994. Ann Int Med 136(5):341–348
Roger VL, Weston SA, Redfield MM, Hellermann-Homan JP, Killian J, Yawn BP, Jacobsen SJ (2004) Trends in heart failure incidence and survival in a community-based population. JAMA 292(3):344–350
Empana J-P, Dargent-Molina P, Bre´art G (2004) Effect of hip fracture on mortality in elderly women: the EPIDOS Prospective Study. J Am Geriatr Soc 52:685–690
Brogan database: Available at: http: //www.broganinc.com. Accessed March 2006
Will BP, Berthelot JM, Le Petit C, Tomiak EM, Verma S, Evans WK (2000) Estimates of the lifetime costs of breast cancer treatment in Canada. Eur J Cancer 36(6):724–735
Pinilla J (1998) for the Gynecology Tumor Group: cost minimization analysis of high-dose-rate versus low-dose-rate brachytherapy in endometrial cancer. Int J Radiat Oncol Biol Phys 42(1):87–90
O’Brien B, Levine M, Willan A, Goeree R, Haley S, Blackhouse G, Gent M (1999) Economic evaluation of outpatient treatment with low-molecular-weight heparin for proximal vein thrombosis. Arch Intern Med 159:2298–2304
Grover SA, Coupal L, Zowall H, Alexander CM, Weiss TW, Gomes DR (2001) How cost-effective is the treatment of dyslipidemia in patients with diabetes but without cardiovascular disease? Diabetes Care 24:45–50
Levy AR, Briggs AH, Demers C, O’Brien BJ (2001) Cost-effectiveness of beta-blocker therapy with metoprolol or with carvedilol for treatment of heart failure in Canada. Am Heart J 142(3):537–543
Wiktorowicz ME, Goeree R, Papaioannou A, Adachi JD, Papadimitropoulos E (2001) Economic implications of hip fracture: health service use, institutional care and cost in Canada. Osteoporos Int 2(4):271–278
Coyle D, Cranney A, Lee KM, Welch V, Tugwell P (2000) Cost-effectiveness research in osteoporosis. Drug Dev Res 49:135–140
Sorensen S, Brown R, Benedict A, Flood E, Revicki D (2004) Patient-rated utilities in postmenopausal early breast cancer: a cross country comparison. Contributed Podium Presentations. Value Health 7(6):637–657
Leibson CL, Tosteson AN, Gabriel SE, Ransom JE, Melton LJ (2002) Mortality, disability, and nursing home use for persons with and without hip fracture: a population-based study. J Am Geriatr Soc 50(10):1644–1650
Fryback DG, Dasbach EJ, Klein R, Klein BE, Dorn N, Peterson K, Martin PA (1993) The Beaver Dam Health Outcomes Study: initial catalog of health-state quality factors. Med Decis Making 13(2):89–102
Briggs AH (1999) A Bayesian approach to stochastic cost-effectiveness analysis. Health Econ 8:257–261
Doubilet P, Begg CB, Weinstein MC, Braun P, McNeil BJ (1985) Probabilistic sensitivity analysis using Monte Carlo simulation. A practical approach. Med Decis Making 5(2):157–177
Bank of Canada. Rates and Statistics (2007) Daily Currency Converter. Availalbe at: http: //www.bankofcanada.ca/en/rates/converter.html. Accessed April 12, 2007
Eichler HG, Kong SX, Gerth WC, Mavros P, Jonsson B (2004) Use of cost-effectiveness analysis in health-care resource allocation decision-making: how are cost-effectiveness thresholds expected to emerge? Value Health 7(5):518–528
Laupacis A, Feeny D, Detsky AS, Tugwell PX (1992) How attractive does a new technology have to be to warrant adoption and utilization? Tentative guidelines for using clinical and economic evaluations. Can Med Assoc J 146:473–481
Laupacis A, Feeny D, Detsky AS, Tugwell PX (1993) Tentative guidelines for using clinical and economic evaluations revisited. Can Med Assoc J 148:927–929
Hirth RA, Chernew ME, Miller E, Fendrick AM, Weissert WG (2000) Willingness to pay for a quality-adjusted life year: in search of a standard. Med Decis Making 20(3):332–342
Ubel PA, Hirth RA, Chernew ME, Fendrick AM (2003) What is the price of life and why doesn’t it increase at the rate of inflation? Arch Intern Med 163:1637–1641
Delea T, Smith R, Karnon J (2002) Cost-effectiveness of letrozole vs tamoxifen as 1st line hormonal therapy for postmenopausal women with advanced breast cancer: the US perspective. 25th Annual San Antonio Breast Cancer Symposium; December 11–14, 2002. Abstract 542
Karnon J, Delea T, Johnston SR, Smith R, Brandman J, Sung J, Goss PE (2006) Cost effectiveness of extended adjuvant letrozole in postmenopausal women after adjuvant tamoxifen therapy: the UK perspective. Pharmacoeconomics 24(3):237–250
Delea TE, Karnon J, Smith RE, Johnston SR, Brandman J, Sung JC, Gross PE (2006) Cost-effectiveness of extended adjuvant letrozole therapy after 5 years of adjuvant tamoxifen therapy in postmenopausal women with early-stage breast cancer. Am J Manag Care 12(7):374–386
El Ouagari K, Karnon J, Delea T, Talbot W, Brandman J (2006) Cost-effectiveness of letrozole in the extended adjuvant treatment of women with early breast cancer. Breast Cancer Res Treat 101(1):37–49
Hillner BE (2004) Benefit and projected cost-effectiveness of anastrozole versus tamoxifen as initial adjuvant therapy for patients with early-stage estrogen receptor-positive breast cancer. Cancer 101:1311–1322
National Institute for Health and Clinical Excellenc (2006) Final appraisal determination—Hormonal therapies for the adjuvant treatment of early oestrogen receptor. Positive breast cancer. Issue date: August 2006. Available at: http: //www.nice.org.uk/page.aspx?o=63296. Accessed October 4, 2006
Cuzick J, Sasieni P, Howell A (2006) Should aromatase inhibitors be used as initial adjuvant treatment or sequenced after tamoxifen? Br J Cancer 94:460–464. doi:10.1038/sj.bjc.6602964 Published online January 24, 2006
Acknowledgements
Financial Support: This study was funded by Novartis Pharmaceuticals Corporation.
Potential Conflicts of Interest: TD and OS are employees of PAI, an independent contract research organization, which has received research support and consulting fees from Novartis and other pharmaceutical companies. JK has received research support from Novartis. K E-O is an employee of Novartis.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was conducted independently of the BIG 1-98 Steering Committee.
Rights and permissions
About this article
Cite this article
Delea, T.E., El-Ouagari, K., Karnon, J. et al. Cost-effectiveness of letrozole versus tamoxifen as initial adjuvant therapy in postmenopausal women with hormone-receptor positive early breast cancer from a Canadian perspective. Breast Cancer Res Treat 108, 375–387 (2008). https://doi.org/10.1007/s10549-007-9607-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9607-7