Abstract
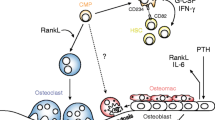
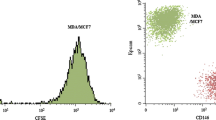
The cellular and humoral mechanisms accounting for tumour osteolysis in metastatic breast cancer are uncertain. Osteoclasts, the specialised multinucleated cells responsible for tumour osteolysis, are derived from monocyte/macrophage precursors. Breast cancer-derived tumour-associated macrophages (TAMs) are capable of osteoclast differentiation but the cellular and humoral mechanisms controlling this activity are uncertain. In this study, TAMs were isolated from primary breast cancers and cultured in the presence and absence of cytokines/growth factors influencing osteoclastogenesis. Extensive TAM-osteoclast differentiation occurred only in the presence of RANKL and M-CSF; this process was inhibited by OPG and RANK:Fc, decoy receptors for RANKL. Breast cancer-derived fibroblasts and human bone stromal cells expressed mRNA for RANKL, OPG and TRAIL, and co-culture of these fibroblasts with human monocytes stimulated osteoclast formation by a RANKL-dependent mechanism. Osteoclast formation and lacunar resorption also occurred by a RANKL-independent mechanism when the conditioned medium from breast cancer cells, MDA-MB-231 and MCF-7, was added (with M-CSF) to monocyte cultures. Our findings indicate that TAMs in breast cancer are capable of osteoclast differentiation and that breast cancer-derived fibroblasts and breast cancer cells contribute to this process by producing soluble factors that influence osteoclast formation by RANKL-dependent and RANKL-independent mechanisms respectively.






Similar content being viewed by others
References
Stoll BA (1983) Natural history, prognosis and staging of bone metastases. In: Stoll BA, Parbhoo S (eds) Bone metastases: monitoring and treatment. Raven Press, New York, pp 1–4
Clohisy DR, Palkert D, Ramnaraine ML, Pekurovsky I, Oursler MJ (1996) Human breast cancer induces osteoclast activation and increases the number of osteoclasts at sites of tumor osteolysis. J Orthop Res 14:396–402
Mundy GR (1991) Mechanisms of osteolytic bone destruction. Bone 12:51–56
Blair HC, Athanasou NA (2004) Recent advances in osteoclast biology. Histol and Histopathol 19:189–199
Fujikawa Y, Quinn JM, Sabokbar A, McGee JO, Athanasou NA (1996) The human osteoclast precursor circulates in the monocyte fraction. Endocrinology 137:4058–4060
Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A, Tsuda E, Morinaga T, Higashio K, Udagawa N, Takahashi N, Suda T (1998) Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclast inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 95:3597–3602
Hofbauer LC, Khosla S, Dunstan C, Lacey DL, Boyle W, Riggs BL (2000) The roles of osteoprotegerin and osteoprotegerin ligand in the paracrine regulation of bone resorption. Bone Min Res 15:2–12
Simonet WS, Lacey DL, Dunstan CR, Kelley M, Chang MS, Luthy R, Nguyen HQ, Wooden S, Bennett L, Boone T, Shimamoto G, DeRose M, Elliott R, Colombero A, Tan HL, Trail G, Sullivan J, Davy E, Bucay N, Renshaw-Gegg L, Hughes TM, Hill D, Pattison W, Campbell P, Boyle WJ, et al (1997) Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89:309–319
Thomas RJ, Guise TA, Yin JJ, Elliott J, Horwood NJ, Martin TJ, Gillespie MT (1999) Breast cancer cells interact with osteoblasts to support osteoclast formation. Endocrinology 140:4451–4458
Itonaga I, Kudo O, Sabokbar A, Danks L, Ferguson D, Fujikawa Y, Athanasou NA (2004) Transforming growth factor β induces human osteoclast formation. Bone 34:57–64
Kobayashi K, Takahashi N, Jimi E, Udagawa N, Takami M, Kotake S, Nakagawa N, Kinosaki M, Yamaguchi K, Shima N, Yasuda H, Morinaga T, Higashio K, Martin TJ, Suda T (2000) Tumor necrosis factor alpha stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL-RANK interaction. J Exp Med 191:275–286
Kudo O, Fujikawa Y, Itonaga I, Sabokbar A, Torisu T, Athanasou NA (2002) Proinflammatory cytokine (TNFalpha/IL-1alpha) induction of human osteoclast formation. J Pathol 198:220–227
Kudo O, Sabokbar A, Pocock A, Itonaga I, Fujikawa Y, Athanasou NA (2003) Interleukin-6 and interleukin-11 support human osteoclast formation by a RANKL-independent mechanism. Bone 32:1–7
Bugelski PJ, Corwin SP, North SM, Kirsh RL, Nicolson GL, Poste G (1987) Macrophage content of spontaneous metastases at different stages of growth. Cancer Res 47:4141–4145
Van Ravenswaay Classen HH, Kluin PM, Fleuren G (1992) Tumour infiltrating cells in human cancer: on the possible role of CD16+ macrophages in anti-tumour cytoxicity. Lab Invest 67:166–174
Quinn JM, Athanasou NA, McGee JO’D (1992) Tumour infiltrating macrophages are capable of bone resorption. J Cell Science 101:681–686
Quinn J, Matsumura Y, Tarin D, McGee JO’D, Athanasou NA (1994) Cellular and hormonal mechanisms associated with malignant bone resorption. Lab Invest 71:465–471
Hunt N, Fujikawa Y, Itonaga I, Harris A, Athanasou NA (2001) Cellular mechanisms of bone resorption in breast cancer. Br J Cancer 85:78–84
Quinn JMW, McGee JO’D, Athanasou NA (1998) Human tumour-associated macrophages differentiate into osteoclastic bone-resorbing cells. J Pathol 184:31–36
Athanasou NA, Quinn J (1990) Immunophenotypic differences between osteoclasts and macrophage polykaryons: immunohistological distinction and implications for osteoclast ontogeny and function. J Clin Pathol 43:997–1003
Minkin C (1982) Bone acid phosphatase: tartrate-resistant acid phosphatase as a marker of osteoclast function. Calcif Tissue Int 34:285–290
Davies J, Warwick J, Totty N, Philp R, Helfrich M, Horton M (1989) The osteoclast functional antigen, implicated in the regulation of bone resorption, is biochemically related to the vitronectin receptor. J Cell Biol 109:1817–1826
Gundle R, Beresford JN (1995) The isolation and culture of cells from explants of human trabecular bone. Calcif Tissue Int 56:8–10
Sun SG, Lau YS, Itonaga I, Sabokbar A, Athanasou NA (2006) Bone stromal cells in pagetic bone and Paget’s sarcoma express RANKL and support human osteoclast formation. J Pathol 209(1):114–120
Bugelski PJ, Kirsh RL, Sowinski JM, Poste GL (1985) Changes in the macrophage content of lung metastases at different stages in tumor growth. Am J Pathol 118:419–424
Normann SJ (1985) Macrophage infiltration and tumour progression. Cancer Metastasis Rev 4:227–291
Galasko CBS (1976) Mechanisms of bone destruction in the development of skeletal metastasis. Nature 276:726–728
Mantovani A (1994) Tumor-associated macrophages in neoplastic progression: a paradigm for the in vivo function of chemokines. Lab Invest 71:5–16
Yamashiro S, Takaya M, Nishi T, Kuratsu J, Yashimura T, Usho Y, Takahashi K (1994) Tumor-derived monocyte chemoattractant protein induces intratumoral infiltration of monocyte-derived macrophage subpopulation in transplanted rat tumour. Am J Pathol 145:856–867
Yoshimura T, Takeya M, Takahashi K (1991) Molecular cloning of rat monocyte chemoattractant protein-1 (MCP-1) and its expression in rat spleen cells and tumour cell lines. Biochem Biophys Res Commun 174:504–509
Flanagan AM, Lader CS (1998) Update on the biologic effects of macrophage-colony stimulating factor. Curr Opinion Hematol 5:181–185
Faust J, Lacey DL, Hunt P, Burgess TL, Scully S, Van G, Eli A, Qian Y, Shalhoub V (1999) Osteoclast markers accumulate on cells developing from human peripheral blood mononuclear precursors. J Cell Biochem 72:67–80
Quinn JWM, Elliott J, Gillespie MT, Martin TJ (1998) A combination of osteoclast differentiation factor and macrophage-colony stimulating factor is sufficient for both human and mouse osteoclast formation. Endocrinology 139:4424–4427
Massey HM, Flanagan AM (1999) Human osteoclasts derive from CD14-positive monocytes. Br J Haematol 106:167–170
Quinn JM, Horwood NJ, Elliott J, Gillespie MT, Martin TJ (2000) Fibroblastic stromal cells express receptor activator of NF-kappa B ligand and support osteoclast differentiation. J Bone Miner Res 15:1459–1466
Sabokbar A, Itonaga I, Sun SG, Kudo O, Athanasou NA (2005) Arthroplasty membrane-derived fibroblasts directly induce osteoclast formation and osteolysis in aseptic loosening. J Orthop Res 23:511–519
Takayanagi H, Iizuka H, Juji T, Nakagawa T, Yamamoto A, Miyazaki T, Koshihara Y, Oda H, Nakamura K, Tanaka S (2000) Involvement of receptor activator of nuclear factor kappaB ligand/osteoclast differentiation factor in osteoclastogenesis from synoviocytes in rheumatoid arthritis. Arthritis Rheum 43:259–269
Emery JG, McDonnell P, Burke MB, Deen KC, Lyn S, Silverman C, Dul E, Appelbaum ER, Eichman C, Diprinzio R, Dodds RA, James IE, Rosenberg M, Lee JC, Young PR (1998) Osteoprotegerin is a receptor for the cytotoxic ligand TRAIL. J Biol Chem 273:14363–14367
Van Poznak C, Cross SS, Saggese M, Hudis C, Panageas KS, Norton L, Coleman RE, Holen I (2006) Espression of osteoprotegerin (OPG), TNF related apoptosis inducing ligand (TRAIL), and receptor activator of nuclear factor kappaB ligand (RANKL) in human breast tumours. J Clin Pathol 59:56–63
Pederson L, Winding B, Foged NT (1999) Identification of breast cancer cell line-derived paracrine factors that stimulate osteoclast activity. Cancer Res 59:5849–5855
Lau YS, Sabokbar A, Giele H, Cerundolo V, Hofstetter W, Athanasou NA (2006) Malignant melanoma and bone resorption. Br J Cancer 94:1496–503
Acknowledgements
The authors wish to thank the Frances and Augustus Newman Foundation, Jenny Mays-Smith Skin Cancer Research Fund, Oxfordshire Health Service Research Committee and the Rosetrees Charitable Trust.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lau, Y.S., Danks, L., Sun, S.G. et al. RANKL-dependent and RANKL-independent mechanisms of macrophage-osteoclast differentiation in breast cancer. Breast Cancer Res Treat 105, 7–16 (2007). https://doi.org/10.1007/s10549-006-9438-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9438-y