Summary
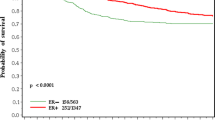
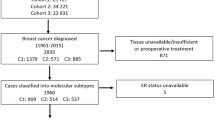
The knowledge of estrogen receptor (ER) status is important in the management of breast cancer patients. More precisely, analytical methods for ER determination have changed over the last two decades from ligand binding assay (LBA) dextran-coated charcoal (DCC) to enzyme immuno-assay (EIA) and more recently immunohistochemistry (IHC). We examined the respective clinical impact of ER determination according to these 3 methods over the period 1983–1999 within a group of 1940 patients, all operated and followed in the single institution Centre Antoine Lacassagne. Validated cut off values were 10 and 15 fmol/mg protein for both LBA-DCC and EIA, respectively and 10% of stained cells for IHC. During the years it was noted that the initial size of the tumor decreased and that the proportion of positive axillary nodes and negative ER tumors was different according to the ER method. ER negativity was 20, 13 and 10% in LBA-DCC, EIA, IHC, respectively. ER was a strong predictor of overall survival in the whole population (Mantel-Cox, p<0.00001); however when stratifying the analysis on ER method groups, ER was still a prognostic indicator in the EIA, LBA-DCC group but not in the IHC group (the follow-up was too short). It is important to keep these data in mind when conducting large retrospective studies evaluating prognostic markers in breast cancer patients.
Similar content being viewed by others
References
Bernoux A, de Cremoux P, Laine-Bidron C, Martin EC, Asselain B, Magdelenat H, 1998 Estrogen receptor negative and progesterone receptor positive primary breast cancer: pathological characteristics and clinical outcome. Institute Curie Breast cancer Study GroupBreast Cancer Res Treat 49(3): 219–225
Kaufmann M, 1996 Review of known prognostic variablesRecent Results Cancer Res 140: 77–87
Zhu K, Bernard LJ, Levine RS, Williams SM, 1997 Estrogen receptor status of breast cancer: a marker of different stages of tumor or different entities of the disease ? Med Hypotheses 49(1): 69–75
Diez-Gibert O, Huguet J, Rosel P, Bonnin MR, Navarro MA, 1998 Immunological quantitation of nuclear steroid receptors to optimize the biological classification of breast tumorsTumor Biol 19(4): 222–228
Dittadi R, Meo S, Amoroso B, Gion M, 1997 Detection of different estrogen receptor forms in breast cancer cytosol by enzyme immunoassayCancer Res 57: 1066–1072
Lacroix M, Querton G, Hennebert P, Larsimont D, Leclercq G, 2001 Estrogen receptor analysis in primary breast tumors by ligand-binding assay, immunocytochemical assay, and northern blot: a comparisonBreast Cancer Res Treat 67: 263–271
Romain S, Formento JL, Guirou O, Francoual M, Milano G, Martin PM, 1994 Determination of oestrogen receptors by enzyme immunoassay. Technical differences between laboratories and their consequencesEur J Cancer 6: 740–746
Jarque F, Lluch A, Vizcarra E, Munoz M, Alberola V, Garcia-Conde J, 1994 Quantitative monoclonal antibody determination of estrogen and progesterone receptors in human breast cancer: correlation with the radioligand methodOncology 51(5): 435–439
Nolan C, Przywara L, Weigand R, 1987 The Abbott ER-AIA monoclonal kitClin Chem 33(6): 1105–1108
Wishart GC, Gaston M, Poultsidis AA, Purushotham AD, 2002 Hormone receptor status in primary breast cancer – time for a consensus ? Eur J Cancer 38: 1201–1203
Allred DC, Harvey JM, Berardo M, Clark GM, 1998 Prognostic and predictive factors in breast cancer by immunohistochemical analysisMod Pathol 11(2): 155–168
Bloom HJG, Richardson WW, 1957 Histological grading and prognosis in breast cancerBr J Cancer 11: 359–377
Mobbs BG, Jonhson IE, 1987 Use of an enzyme immunoassay (EIA) for quantitation of cytosolic and nuclear estrogen receptor, and correlation with progesterone recpetor in human breast cancerJ Steroid Biochem 28: 653–662
Bevitt DJ, Milton ID, Piggot N, Henry L, Carter MJ, Toms GL, Lennard TW, Westley B, Angus B, Horne CH, 1997 New monoclonal antibodies to oestrogen and progesterone receptors effective for paraffin section immunohistochemistryJ Pathol 183(2): 228–232
Kaplan EL, Meier P, 1958 Non-parametric estimation for incomplete observationsJ Am Stat Assoc 53: 457–481
Costa SC, Lange S, Klinga K, Merkle E, Kaufmann M, 2002 Factors influencing the prognostic role of oestrogen and progesterone receptor levels in breast cancer – results of the analysis of 670 patients with 11 years of follow-upEur J Cancer 38: 1329–1334
Pujol P, Hilsenbeck SG, Chamness GC, Elledge RM, 1994 Rising levels of estrogen receptor in breast cancer over 2 decadesCancer 1: 1601–1606
Early Breast Cancer Trialists Collaborative Group. 1998 Tamoxifen for early breast cancer: overview of the randomized trialsLancet 351: 1451–1467
Chapman JW, Mobbs BG, McCready DR, Lickley HL, Trudeau ME, Hanna W, Kahn HJ, Sawka CA, Fish EB, Pritchard KI, 1996 An investigation of cut-points for primary breast cancer oestrogen and progesterone receptor assaysJ Steroid Biochem Mol Biol 57(5): 323–328
Pichon MF, Broet P, Magdelenat H, Delarue JC, Spyratos F, Basuyau JP, Saez S, Rallet A, Courrire P, Million R, Asselain B, 1996 Prognostic value of steroid receptors after long-term follow-up of 2257 operable breast cancersBr J Cancer 73(12): 1545–1551
Harvey JM, Clark GM, Osborne CK, Allred DC, 1999 Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancerJ Clin Oncol 17(5): 1474–1481
Altman DG, Lausen B, Sauerbrei W, Schumacher M, 1994 Dangers of using “optimal” cutpoints in the evaluation of prognostic factorsJ Natl Cancer Inst 86: 829–835
Botha JL, Bray F, Sankila R, Parkin DM, 2003 Breast cancer incidence and mortality trends in 16 European countriesEur J Cancer 39: 1718–1729
Stierer M, Rosen H, Weber R, Hanak H, Spona J, Tuchler H, 1993 Immunohistochemical and biochemical measurement of estrogen and progesterone receptors in primary breast cancer. Correlation of histopathology and prognostic factorsAnn Surg 218(1): 13–21
Allred DC, Bustamante MA, Daniel CO, Gaskill HV, Cruz AB, Jr, 1990 Immunocytochemical analysis of estrogen receptors in human breast carcinomas. Evaluation of 130 cases and review of the literature regarding concordance with biochemical assay and clinical relevanceArch Surg 125(1): 107–113
Molino A, Micciolo R, Turazza M, Bonetti F, Piubello Q, Corgnati A, Sperotto L, Recaldin E, Spagnolli P, Manfrin E, Bonetti A, Nortilli R, Tomezzoli A, Pollini GP, Modena S, Cetto GL, 1997 Prognostic significance of estrogen receptors in 405 primary breast cancers: a comparison of immunohistochemical and biochemical methodsBreast Cancer Res Treat 45(3): 241–249
Reiner A, Spona J, Reiner G, Schemper M, Kolb R, Kwasny W, Fugger R, Jakesz R, Holzner JH, 1986 Estrogen receptor analysis on biopsies and fine-needle aspirates from human breast carcinoma. Correlation of biochemical and immunohistochemical methods using monoclonal antireceptor anitbodiesAm J Pathol 125(3): 443–449
Charpin C, Martin PM, De Victor B, Lavaut MN, Habib MC, Andrac L, Toga M, 1998 Multiparametric study (SABA 200) of estrogen receptor immunocytochemical assay in 400 human breast carcinomas: analysis of estrogen receptor distribution heterogeneicity in tisues and correlations with dextran charcoal assays and morphological dataCancer Res 15: 1578–1586
Hanna W, Mobbs BG, 1989 Comparative evaluation of ER-ICA and enzyme immunoassay for the quantitation of estrogen receptors in breast cancer Am J Clin Pathol 91(2): 182–186
Elledge RM, Green S, Pugh R, Allred DC, Clark GM, Hill J, Ravdin P, Martino S, Osborne CK, 2000 Estrogen receptor (ER) and progesterone receptor (PgR), by ligand-binding assay compared with ER, PgR and pS2, by immuno-histochemistry in predicting response to tamoxifen in metastasis breast cancer: a Southwest Oncology Group StudyInt J Cancer 20: 111–117
Li CI, Anderson BO, Daking JR, Moe RE, 2003 Trends in incidence rates of invasive lobular and ductal breast carcinomaJAMA 289(11): 1421–1424
Li CI, Daking JR, Malone KE, 2003 Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998J Clin Oncol 21(1): 28–34
Parker RL, Huntsman DG, Lesack DW, Cupples JB, Grant DR, Akbari M, Gilks CB, 2002 Assessment of interlaboratory variation in the immunohistochemical determination of estrogen receptor status using a breast cancer tissue microarrayAm J Clin Pathol 117(5): 723–728
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented in part at the 38th American Society of Clinical Oncology (ASCO) meeting, Orlando, May 18–21, 2002.
Rights and permissions
About this article
Cite this article
Magné, N., Toillon, RA., Castadot, P. et al. Different clinical impact of estradiol receptor determination according to the analytical method: a study on 1940 breast cancer patients over a period of 16 consecutive years. Breast Cancer Res Treat 95, 179–184 (2006). https://doi.org/10.1007/s10549-005-9065-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-005-9065-z