Abstract
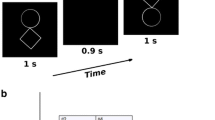
The neuronal activity of the preterm brain is characterized by various endogenous activities whose roles in neurodevelopmental maturation processes have not been fully elucidated. The preterm EEG is characterized by discontinuities composed of short bursts of activity with dominant low frequencies. One of the earliest endogenous activities is the theta temporal activity in coalescence with slow waves (TTA-SW), which appears at 24 to 32 weeks of gestational age (wGA). The present study investigated the influence of TTA-SW on the spatial organization of the early preterm brain network. To achieve this objective, High-Density EEG data were recorded from preterm infants (29–32 wGA) and functional connectivity (FC) was estimated from the scalp EEG. TTA-SW, particularly in the theta band, induced increased FC between left temporal and left frontal areas and between left temporal and parietal areas with TTA-SW at the left temporal region, while FC was limited to the right temporal regions in the case of TTA-SW at the right temporal region. Regardless of the lateralization of TTA-SW, long-range FCs were observed between left frontal to left parietal areas, suggesting that these regions, together with the temporal region, provide a basis for coherent neuronal activation across distal cortical regions. TTA-SW dynamic features showed that brief phases of TTA-SW had an impact on both local and whole brain network organization, supporting the importance of TTA-SW as a biomarker of brain development.






Similar content being viewed by others
References
Acharya JN, Hani AJ, Thirumala PD, Tsuchida TN (2016) American clinical neurophysiology society guideline 3: a proposal for standard montages to be used in clinical EEG. J Clin Neurophysiol 33:312–316. https://doi.org/10.1097/WNP.0000000000000317
André M, Lamblin M-D, d’Allest AM et al (2010) Electroencephalography in premature and full-term infants. Developmental features and glossary. Neurophysiol Clin Clin Neurophysiol 40:59–124. https://doi.org/10.1016/j.neucli.2010.02.002
Ayoub AE, Kostovic I (2009) New horizons for the subplate zone and its pioneering neurons. Cereb Cortex N Y N 19:1705–1707. https://doi.org/10.1093/cercor/bhp025
Ball G, Aljabar P, Zebari S et al (2014) Rich-club organization of the newborn human brain. Proc Natl Acad Sci 111:7456–7461. https://doi.org/10.1073/pnas.1324118111
Blankenship AG, Feller MB (2010) Mechanisms underlying spontaneous patterned activity in developing neural circuits. Nat Rev Neurosci 11:18–29. https://doi.org/10.1038/nrn2759
Dosenbach NUF, Nardos B, Cohen AL et al (2010) Prediction of individual brain maturity using fMRI. Science 329:1358–1361. https://doi.org/10.1126/science.1194144
Fair DA, Cohen AL, Power JD et al (2009) Functional brain networks develop from a “local to distributed” organization. PLoS Comput Biol 5:e1000381. https://doi.org/10.1371/journal.pcbi.1000381
Fransson P, Skiöld B, Horsch S et al (2007) Resting-state networks in the infant brain. Proc Natl Acad Sci USA 104:15531–15536. https://doi.org/10.1073/pnas.0704380104
Fransson P, Aden U, Blennow M, Lagercrantz H (2011) The functional architecture of the infant brain as revealed by resting-state fMRI. Cereb Cortex N Y N 21:145–154. https://doi.org/10.1093/cercor/bhq071
Glasel H, Leroy F, Dubois J et al (2011) A robust cerebral asymmetry in the infant brain: the rightward superior temporal sulcus. NeuroImage 58:716–723. https://doi.org/10.1016/j.neuroimage.2011.06.016
Hipp JF, Hawellek DJ, Corbetta M et al (2012) Large-scale cortical correlation structure of spontaneous oscillatory activity. Nat Neurosci 15:884–890. https://doi.org/10.1038/nn.3101
Iyer KK, Roberts JA, Hellström-Westas L et al (2015) Cortical burst dynamics predict clinical outcome early in extremely preterm infants. Brain J Neurol 138:2206–2218. https://doi.org/10.1093/brain/awv129
Johnson MH (2001) Functional brain development in humans. Nat Rev Neurosci 2:475–483. https://doi.org/10.1038/35081509
Judaš M, Sedmak G, Kostović I (2013) The significance of the subplate for evolution and developmental plasticity of the human brain. Front Hum Neurosci 7:423. https://doi.org/10.3389/fnhum.2013.00423
Kayser J, Tenke CE (2015) Issues and considerations for using the scalp surface Laplacian in EEG/ERP research: a tutorial review. Int J Psychophysiol Off J Int Organ Psychophysiol 97:189–209. https://doi.org/10.1016/j.ijpsycho.2015.04.012
Kilb W, Kirischuk S, Luhmann HJ (2011) Electrical activity patterns and the functional maturation of the neocortex. Eur J Neurosci 34:1677–1686. https://doi.org/10.1111/j.1460-9568.2011.07878.x
Kostović I, Judas M (2010) The development of the subplate and thalamocortical connections in the human foetal brain. Acta Paediatr Oslo Nor 99:1119–1127. https://doi.org/10.1111/j.1651-2227.2010.01811.x
Lamblin MD, André M, Challamel MJ et al (1999) [Electroencephalography of the premature and term newborn. Maturational aspects and glossary]. Neurophysiol Clin 29:123–219
Leroy F, Cai Q, Bogart SL et al (2015) New human-specific brain landmark: the depth asymmetry of superior temporal sulcus. Proc Natl Acad Sci 112:1208–1213. https://doi.org/10.1073/pnas.1412389112
Mahmoudzadeh M, Dehaene-Lambertz G, Fournier M et al (2013) Syllabic discrimination in premature human infants prior to complete formation of cortical layers. Proc Natl Acad Sci 110:4846–4851. https://doi.org/10.1073/pnas.1212220110
Maris E (2012) Statistical testing in electrophysiological studies. Psychophysiology 49:549–565. https://doi.org/10.1111/j.1469-8986.2011.01320.x
O’Leary DDM, Chou S-J, Sahara S (2007) Area patterning of the mammalian cortex. Neuron 56:252–269. https://doi.org/10.1016/j.neuron.2007.10.010
Omidvarnia A, Fransson P, Metsäranta M, Vanhatalo S (2014) Functional bimodality in the brain networks of preterm and term human newborns. Cereb Cortex N Y N 24:2657–2668. https://doi.org/10.1093/cercor/bht120
Omidvarnia A, Metsäranta M, Lano A, Vanhatalo S (2015) Structural damage in early preterm brain changes the electric resting state networks. NeuroImage 120:266–273. https://doi.org/10.1016/j.neuroimage.2015.06.091
Pallas SL (2001) Intrinsic and extrinsic factors that shape neocortical specification. Trends Neurosci 24:417–423
Penn AA, Shatz CJ (1999) Brain waves and brain wiring: the role of endogenous and sensory-driven neural activity in development. Pediatr Res 45:447–458. https://doi.org/10.1203/00006450-199904010-00001
Perrin F, Pernier J, Bertrand O, Echallier JF (1989) Spherical splines for scalp potential and current density mapping. Electroencephalogr Clin Neurophysiol 72:184–187. https://doi.org/10.1016/0013-4694(89)90180-6
Routier L, Mahmoudzadeh M, Panzani M et al (2017) Plasticity of neonatal neuronal networks in very premature infants: source localization of temporal theta activity, the first endogenous neural biomarker, in temporoparietal areas. Hum Brain Mapp. https://doi.org/10.1002/hbm.23521
Rubinov M, Sporns O (2010) Complex network measures of brain connectivity: uses and interpretations. NeuroImage 52:1059–1069. https://doi.org/10.1016/j.neuroimage.2009.10.003
Selton D, Andre M, Hascoët JM (2000) Normal EEG in very premature infants: reference criteria. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 111:2116–2124
Srinivasan R, Winter WR, Ding J, Nunez PL (2007) EEG and MEG coherence: measures of functional connectivity at distinct spatial scales of neocortical dynamics. J Neurosci Methods 166:41–52. https://doi.org/10.1016/j.jneumeth.2007.06.026
Suppiej A, Cainelli E, Cappellari A et al (2015) Neonatal cortical auditory evoked potentials are affected by clinical conditions occurring in early prematurity. J Clin Neurophysiol 32:419–423. https://doi.org/10.1097/WNP.0000000000000182
Tolonen M, Palva JM, Andersson S, Vanhatalo S (2007) Development of the spontaneous activity transients and ongoing cortical activity in human preterm babies. Neuroscience 145:997–1006. https://doi.org/10.1016/j.neuroscience.2006.12.070
Tononi G, Sporns O, Edelman GM (1994) A measure for brain complexity: relating functional segregation and integration in the nervous system. Proc Natl Acad Sci USA 91:5033–5037
Tritsch NX, Rodríguez-Contreras A, Crins TTH et al (2010) Calcium action potentials in hair cells pattern auditory neuron activity before hearing onset. Nat Neurosci 13:1050–1052. https://doi.org/10.1038/nn.2604
van den Heuvel MP, Sporns O (2013) Network hubs in the human brain. Trends Cogn Sci 17:683–696. https://doi.org/10.1016/j.tics.2013.09.012
van den Heuvel MP, Kersbergen KJ, de Reus MA et al (2015) The neonatal connectome during preterm brain development. Cereb Cortex 25:3000–3013. https://doi.org/10.1093/cercor/bhu095
Vanhatalo S, Kaila K (2006) Development of neonatal EEG activity: from phenomenology to physiology. Semin Fetal Neonatal Med 11:471–478. https://doi.org/10.1016/j.siny.2006.07.008
Vanhatalo S, Palva JM, Holmes MD et al (2004) Infraslow oscillations modulate excitability and interictal epileptic activity in the human cortex during sleep. Proc Natl Acad Sci USA 101:5053–5057. https://doi.org/10.1073/pnas.0305375101
Vecchierini M-F, d’Allest A-M, Verpillat P (2003) EEG patterns in 10 extreme premature neonates with normal neurological outcome: qualitative and quantitative data. Brain Dev 25:330–337. https://doi.org/10.1016/S0387-7604(03)00007-X
Vecchierini M-F, André M, d’Allest AM (2007) Normal EEG of premature infants born between 24 and 30 weeks gestational age: terminology, definitions and maturation aspects. Neurophysiol Clin Clin Neurophysiol 37:311–323. https://doi.org/10.1016/j.neucli.2007.10.008
Wallois F (2010) Synopsis of maturation of specific features in EEG of premature neonates. Neurophysiol Clin Clin Neurophysiol 40:125–126. https://doi.org/10.1016/j.neucli.2010.02.001
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Micah M. Murray.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Adebimpe, A., Routier, L. & Wallois, F. Preterm Modulation of Connectivity by Endogenous Generators: The Theta Temporal Activities in Coalescence with Slow Waves. Brain Topogr 32, 762–772 (2019). https://doi.org/10.1007/s10548-019-00713-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-019-00713-z