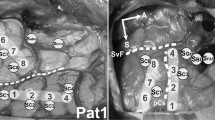
Abstract
During awake brain surgery for tumour resection, in situ EEG recording (ECoG) is used to identify eloquent areas surrounding the tumour. We used the ECoG setup to record the electrical activity of cortical and subcortical tumours and then performed frequency and connectivity analyses in order to identify ECoG impairments and map tumours. We selected 16 patients with cortical (8) and subcortical (8) tumours undergoing awake brain surgery. For each patient, we computed the spectral content of tumoural and healthy areas in each frequency band. We computed connectivity of each electrode using connectivity markers (linear and non-linear correlations, phase-locking and coherence). We performed comparisons between healthy and tumour electrodes. The ECoG alterations were used to implement automated classification of the electrodes using clustering or neural network algorithms. ECoG alterations were used to image cortical tumours.Cortical tumours were found to profoundly alter all frequency contents (normalized and absolute power), with an increase in the δ activity and a decreases for the other bands (P < 0.05). Cortical tumour electrodes showed high level of connectivity compared to surrounding electrodes (all markers, P < 0.05). For subcortical tumours, a relative decrease in the γ1 band and in the alpha band in absolute amplitude (P < 0.05) were the only abnormalities. The neural network algorithm classification had a good performance: 93.6 % of the electrodes were classified adequately on a test subject. We found significant spectral and connectivity ECoG changes for cortical tumours, which allowed tumour recognition. Artificial neural algorithm pattern recognition seems promising for electrode classification in awake tumour surgery.








Similar content being viewed by others
Abbreviations
- Δ:
-
Spectral content in the delta band in percent
- θ:
-
Spectral content in the theta band in percent
- α:
-
Spectral content in the alpha band in percent
- β:
-
Spectral content in the beta band in percent
- γ1 :
-
Spectral content in the gamma1 (<40 Hz) band in percent
- LCorr:
-
Linear Correlation
- H2Corr:
-
H2 Correlation
- CohLF:
-
Coherence in the low frequencies (<4 Hz)
- CohHF:
-
Coherence in high frequencies (4–40 Hz)
- PLVLF:
-
Phase-Locking value in the low frequencies (<4 Hz)
- PLVHF:
-
Phase-Locking value in the high frequencies (4–40 Hz)
- PLI:
-
Phase lag index
References
Ansari-Asl K, Senhadji L, Bellanger J-J, Wendling F (2006) Quantitative evaluation of linear and nonlinear methods characterizing interdependencies between brain signals. Phys Rev E: Stat, Nonlin, Soft Matter Phys 74:031916
Bartolomei F, Bosma I, Klein M et al (2006) Disturbed functional connectivity in brain tumour patients: evaluation by graph analysis of synchronization matrices. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 117:2039–2049. doi:10.1016/j.clinph.2006.05.018
Borchers S, Himmelbach M, Logothetis N, Karnath H-O (2012) Direct electrical stimulation of human cortex - the gold standard for mapping brain functions? Nat Rev Neurosci 13:63–70. doi:10.1038/nrn3140
Bosma I, Douw L, Bartolomei F et al (2008) Synchronized brain activity and neurocognitive function in patients with low-grade glioma: a magnetoencephalography study. Neuro-Oncol 10:734–744. doi:10.1215/15228517-2008-034
Butz M, Gross J, Timmermann L et al (2004) Perilesional pathological oscillatory activity in the magnetoencephalogram of patients with cortical brain lesions. Neurosci Lett 355:93–96
Caudill M (1987) Neural Networks Primer, Part I. AI Expert 2:46–52
Colombet B, Woodman M, Badier JM, Bénar CG (2015) AnyWave: a cross-platform and modular software for visualizing and processing electrophysiological signals. J Neurosci Methods 242:118–126. doi:10.1016/j.jneumeth.2015.01.017
Crone NE, Hao L, Hart J et al (2001) Electrocorticographic gamma activity during word production in spoken and sign language. Neurology 57:2045–2053
de Jongh A, Baayen JC, de Munck JC et al (2003) The influence of brain tumor treatment on pathological delta activity in MEG. NeuroImage 20:2291–2301
Flinker A, Chang EF, Barbaro NM et al (2011) Sub-centimeter language organization in the human temporal lobe. Brain Lang 117:103–109. doi:10.1016/j.bandl.2010.09.009
Franciotti R, Iacono D, Della Penna S et al (2006) Cortical rhythms reactivity in AD, LBD and normal subjects: a quantitative MEG study. Neurobiol Aging 27:1100–1109. doi:10.1016/j.neurobiolaging.2005.05.027
Ganslandt O, Fahlbusch R, Nimsky C et al (1999) Functional neuronavigation with magnetoencephalography: outcome in 50 patients with lesions around the motor cortex. J Neurosurg 91:73–79. doi:10.3171/jns.1999.91.1.0073
Hagan MT, Demuth HB, Beale MH (1996) Neural network design. Pws Pub, Boston
Harmony T, Fernández-Bouzas A, Marosi E et al (1993) Correlation between computed tomography and voltage and current source density spectral EEG parameters in patients with brain lesions. Electroencephalogr Clin Neurophysiol 87:196–205. doi:10.1016/0013-4694(93)90019-R
Harmony T, Fernández-Bouzas A, Marosi E et al (1995) Frequency source analysis in patients with brain lesions. Brain Topogr 8:109–117
Hassan M, Dufor O, Merlet I et al (2014) EEG Source Connectivity Analysis: from Dense Array Recordings to Brain Networks. PLoS ONE 9:e105041. doi:10.1371/journal.pone.0105041
He B, Yang L, Wilke C, Yuan H (2011) Electrophysiological Imaging of Brain Activity and Connectivity – Challenges and Opportunities. IEEE Trans Biomed Eng 58:1918–1931. doi:10.1109/TBME.2011.2139210
Hill NJ, Gupta D, Brunner P et al (2012) Recording human electrocorticographic (ECoG) signals for neuroscientific research and real-time functional cortical mapping. J Vis Exp JoVE. doi:10.3791/3993
Hirsch JF, Buisson-Ferey J, Sachs M et al (1966) Electrocorticogram and unitary activites with expanding lesions in man. Electroencephalogr Clin Neurophysiol 21:417–428
Jerrett SA, Corsak J (1988) Clinical utility of topographic EEG brain mapping. Clin EEG Electroencephalogr 19:134–143
Kamada K, Möller M, Saguer M et al (2001) A combined study of tumor-related brain lesions using MEG and proton MR spectroscopic imaging. J Neurol Sci 186:13–21
Kuzum D, Takano H, Shim E et al (2014) Transparent and flexible low noise graphene electrodes for simultaneous electrophysiology and neuroimaging. Nat Commun 5:5259. doi:10.1038/ncomms6259
Lachaux JP, Rodriguez E, Martinerie J, Varela FJ (1999) Measuring phase synchrony in brain signals. Hum Brain Mapp 8:194–208
Leuthardt EC, Miller K, Anderson NR et al (2007) Electrocorticographic frequency alteration mapping: a clinical technique for mapping the motor cortex. Neurosurgery 60:260–270. doi:10.1227/01.NEU.0000255413.70807.6E
Logar C, Boswell M (1991) The value of EEG-Mapping in focal cerebral lesions. Brain Topogr 3:441–446. doi:10.1007/BF01129003
Lopes da Silva F, Pijn JP, Boeijinga P (1989) Interdependence of EEG signals: linear vs. nonlinear associations and the significance of time delays and phase shifts. Brain Topogr 2:9–18
Matsumoto R, Nair DR, LaPresto E et al (2007) Functional connectivity in human cortical motor system: a cortico-cortical evoked potential study. Brain J Neurol 130:181–197. doi:10.1093/brain/awl257
Miller KJ, denNijs M, Shenoy P et al (2007) Real-time functional brain mapping using electrocorticography. NeuroImage 37:504–507. doi:10.1016/j.neuroimage.2007.05.029
Miller KJ, Abel TJ, Hebb AO, Ojemann JG (2011) Rapid online language mapping with electrocorticography. J Neurosurg Pediatr 7:482–490. doi:10.3171/2011.2.PEDS1156
Mittal S, Barkmeier D, Hua J et al (2015) Intracranial EEG analysis in tumor-related epilepsy: Evidence of distant epileptic abnormalities. Clin Neurophysiol Off J Int Fed Clin Neurophysiol. doi:10.1016/j.clinph.2015.06.028
Nolte G, Bai O, Wheaton L et al (2004) Identifying true brain interaction from EEG data using the imaginary part of coherency. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 115:2292–2307. doi:10.1016/j.clinph.2004.04.029
Oshino S, Kato A, Wakayama A et al (2007) Magnetoencephalographic analysis of cortical oscillatory activity in patients with brain tumors: Synthetic aperture magnetometry (SAM) functional imaging of delta band activity. NeuroImage 34:957–964. doi:10.1016/j.neuroimage.2006.08.054
Pallud J, Capelle L, Huberfeld G (2013) Tumoral epileptogenicity: how does it happen? Epilepsia 54(Suppl 9):30–34. doi:10.1111/epi.12440
Pallud J, Le Van Quyen M, Bielle F et al (2014) Cortical GABAergic excitation contributes to epileptic activities around human glioma. Sci Transl Med 6:244ra89. doi:10.1126/scitranslmed.3008065
Park D-W, Schendel AA, Mikael S et al (2014) Graphene-based carbon-layered electrode array technology for neural imaging and optogenetic applications. Nat Commun 5:5258. doi:10.1038/ncomms6258
Pijn JP, Vijn PC, Lopes da Silva FH et al (1990) Localization of epileptogenic foci using a new signal analytical approach. Neurophysiol Clin Clin Neurophysiol 20:1–11
Roland J, Brunner P, Johnston J et al (2010) Passive real-time identification of speech and motor cortex during an awake craniotomy. Epilepsy Behav EB 18:123–128. doi:10.1016/j.yebeh.2010.02.017
Schnider T, Minto C (2008) Pharmacokinetic models of propofol for TCI: Correspondence. Anaesthesia 63:206–206. doi:10.1111/j.1365-2044.2007.05419_1.x
Schölvinck ML, Leopold DA, Brookes MJ, Khader PH (2013) The contribution of electrophysiology to functional connectivity mapping. NeuroImage 80:297–306. doi:10.1016/j.neuroimage.2013.04.010
Schomer DL, da Silva FHL (2011) Niedermeyer’s Electroencephalography: Basic Principles, Clinical Applications, and Related Fields. Lippincott Williams & Wilkins
Sinai A, Bowers CW, Crainiceanu CM et al (2005) Electrocorticographic high gamma activity versus electrical cortical stimulation mapping of naming. Brain J Neurol 128:1556–1570. doi:10.1093/brain/awh491
Smits A, Zetterling M, Lundin M et al (2015) Neurological Impairment Linked with Cortico-Subcortical Infiltration of Diffuse Low-Grade Gliomas at Initial Diagnosis Supports Early Brain Plasticity. Front Neurol. doi:10.3389/fneur.2015.00137
Stam CJ, Nolte G, Daffertshofer A (2007) Phase lag index: assessment of functional connectivity from multi channel EEG and MEG with diminished bias from common sources. Hum Brain Mapp 28:1178–1193. doi:10.1002/hbm.20346
Tran TA, Spencer SS, Javidan M et al (1997) Significance of spikes recorded on intraoperative electrocorticography in patients with brain tumor and epilepsy. Epilepsia 38:1132–1139
Trebuchon A, Guye M, Tcherniack V et al (2012) Interest of EEG recording during direct electrical stimulation for brain mapping function in surgery. Ann Fr Anesthèsie Rèanimation 31:e87–e90. doi:10.1016/j.annfar.2012.04.010
Walter G (1936) The location of cerebral tumours by electro-encephalography. The Lancet 228(5893):305–308
Walter WG, Dovey VJ (1946) Delimitation of subcortical tumours by direct electrography. Lancet Lond Engl 1:5–9
Wasserman PD (1993) Advanced Methods in Neural Computing, 1st edn. John Wiley & Sons Inc, New York, NY, USA
Wu M, Wisneski K, Schalk G et al (2010) Electrocorticographic frequency alteration mapping for extraoperative localization of speech cortex. Neurosurgery 66:E407–E409. doi:10.1227/01.NEU.0000345352.13696.6F
Funding
this research received institutional funding by Assitance publique Hôpitaux de Marseille and Aix-Marseille University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflict of interest.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Boussen, S., Velly, L., Benar, C. et al. In Vivo Tumour Mapping Using Electrocorticography Alterations During Awake Brain Surgery: A Pilot Study. Brain Topogr 29, 766–782 (2016). https://doi.org/10.1007/s10548-016-0502-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-016-0502-6