Abstract
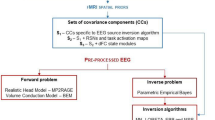
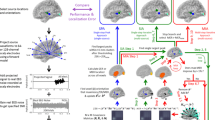
Brain functional networks extracted from fMRI can improve the accuracy of EEG source localization. However, the coupling between EEG and fMRI remains poorly understood, i.e., whether fMRI networks provide information about the magnitude of neural activity, and whether neural sources demonstrate temporal correlations within each network. In this paper, we present an improved version of the NEtwork-based SOurce Imaging method (iNESOI) through Bayesian model comparison. Different models correspond to various matching between EEG and fMRI, and the appropriate one is selected by data with the model evidence. Synthetic and real data tests show that iNESOI has potential to select the appropriate fMRI priors to reach a better source reconstruction than some other typical approaches.






Similar content being viewed by others
References
Ahlfors SP, Simpson GV, Dale AM et al (1999) Spatiotemporal activity of a cortical network for processing visual motion revealed by MEG and fMRI. J Neurophysiol 82(5):2545–2555
Aubert A, Costalat R (2002) A model of the coupling between brain electrical activity, metabolism, and hemodynamics: application to the interpretation of functional neuroimaging. Neuroimage 17(3):1162–1181
Britz J, Van De Ville D, Michel CM (2010) BOLD correlates of EEG topography reveal rapid resting-state network dynamics. Neuroimage 52(4):1162–1170
Calhoun VD, Adali T, McGinty VB, Pekar JJ, Watson TD, Pearlson GD (2001a) fMRI activation in a visual-perception task: network of areas detected using the general linear model and independent components analysis. Neuroimage 14(5):1080–1088
Calhoun VD, Adali T, Pearlson GD, Pekar JJ (2001b) A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp 14(3):140–151
Calhoun VD, Liu J, Adali T (2009) A review of group ICA for fMRI data and ICA for joint inference of imaging, genetic, and ERP data. Neuroimage 45(1 Suppl):S163–S172
Chen H, Yao D, Zhuo Y, Chen L (2003) Analysis of fMRI data by blind separation of data in a tiny spatial domain into independent temporal component. Brain Topogr 15(4):223–232
D’Argembeau A, Collette F, Van der Linden M, Laureys S, Del Fiore G, Degueldre C, Luxen A, Salmon E (2005) Self-referential reflective activity and its relationship with rest: a PET study. Neuroimage 25(2):616–624
Daunizeau J, Grova C, Marrelec G, Mattout J, Jbabdi S, Pelegrini-Issac M, Lina JM, Benali H (2007) Symmetrical event-related EEG/fMRI information fusion in a variational Bayesian framework. Neuroimage 36(1):69–87
Debener S, Ullsperger M, Siegel M, Engel AK (2006) Single-trial EEG-fMRI reveals the dynamics of cognitive function. Trends Cogn Sci 10(12):558–563
Friston K, Mattout J, Trujillo-Barreto N, Ashburner J, Penny W (2007) Variational free energy and the Laplace approximation. Neuroimage 34(1):220–234
Friston K, Harrison L, Daunizeau J, Kiebel S, Phillips C, Trujillo-Barreto N, Henson R, Flandin G, Mattout J (2008) Multiple sparse priors for the M/EEG inverse problem. Neuroimage 39(3):1104–1120
Grova C, Makni S, Flandin G, Ciuciu P, Gotman J, Poline JB (2006) Anatomically informed interpolation of fMRI data on the cortical surface. Neuroimage 31(4):1475–1486
Hampson M, Olson IR, Leung HC, Skudlarski P, Gore JC (2004) Changes in functional connectivity of human MT/V5 with visual motion input. Neuroreport 15(8):1315–1319
Haralick R, Shapiro L (1992) Computer and robot vision, vol I. Addison-Wesley Longman Publishing Co., Boston, pp 28–48
Harrison LM, Penny W, Ashburner J, Trujillo-Barreto N, Friston KJ (2007) Diffusion-based spatial priors for imaging. Neuroimage 38(4):677–695
Heeger DJ, Huk AC, Geisler WS, Albrecht DG (2000) Spikes versus BOLD: what does neuroimaging tell us about neuronal activity? Nat Neurosci 3(7):631–633
Helmholtz H (1853) Ueber einige Gesetze der Vertheilung elektrischer Ströme in körperlichen Leitern mit Anwendung auf die thierisch-elektrischen Versuche. Ann Phys Chem 165(6):211–233
Henson RN, Goshen-Gottstein Y, Ganel T, Otten LJ, Quayle A, Rugg MD (2003) Electrophysiological and haemodynamic correlates of face perception, recognition and priming. Cereb Cortex 13(7):793–805
Henson RN, Flandin G, Friston KJ, Mattout J (2010) A parametric empirical bayesian framework for fMRI-constrained MEG/EEG source reconstruction. Hum Brain Mapp 31(10):1512–1531
Hu D, Yan L, Liu Y, Zhou Z, Friston KJ, Tan C, Wu D (2005) Unified SPM-ICA for fMRI analysis. Neuroimage 25(3):746–755
Jacobs J, Hawco C, Kobayashi E, Boor R, LeVan P, Stephani U, Siniatchkin M, Gotman J (2008) Variability of the hemodynamic response as a function of age and frequency of epileptic discharge in children with epilepsy. Neuroimage 40(2):601–614
Lei X, Xu P, Chen A, Yao D (2009) Gaussian source model based iterative algorithm for EEG source imaging. Comput Biol Med 39(11):978–988
Lei X, Qiu C, Xu P, Yao D (2010) A parallel framework for simultaneous EEG/fMRI analysis: Methodology and simulation. Neuroimage 52(3):1123–1134
Lei X, Xu P, Luo C, Zhao J, Zhou D, Yao D (2011) fMRI functional networks for EEG source imaging. Hum Brain Mapp. doi:10.1002/hbm.21098
Li YO, Adali T, Calhoun VD (2007) Estimating the number of independent components for functional magnetic resonance imaging data. Hum Brain Mapp 28(11):1251–1266
Liu AK, Belliveau JW, Dale AM (1998) Spatiotemporal imaging of human brain activity using functional MRI constrained magnetoencephalography data: Monte Carlo simulations. Proc Natl Acad Sci USA 95(15):8945–8950
Logothetis NK (2008) What we can do and what we cannot do with fMRI. Nature 453(7197):869–878
Mattout J, Phillips C, Penny WD, Rugg MD, Friston KJ (2006) MEG source localization under multiple constraints: an extended Bayesian framework. Neuroimage 30(3):753–767
Mattout J, Henson RN, Friston KJ (2007) Canonical source reconstruction for MEG. Comput Intell Neurosci:Article 67613
McKeown MJ, Makeig S, Brown GG, Jung TP, Kindermann SS, Bell AJ, Sejnowski TJ (1998) Analysis of fMRI data by blind separation into independent spatial components. Hum Brain Mapp 6(3):160–188
Michel CM, Murray MM, Lantz G, Gonzalez S, Spinelli L, Grave de Peralta R (2004) EEG source imaging. Clin Neurophysiol 115(10):2195–2222
Nunez PL, Silberstein RB (2000) On the relationship of synaptic activity to macroscopic measurements: does co-registration of EEG with fMRI make sense? Brain Topogr 13(2):79–96
Operto G, Bulot R, Anton JL, Coulon O (2008) Projection of fMRI data onto the cortical surface using anatomically-informed convolution kernels. Neuroimage 39(1):127–135
Phillips C, Rugg MD, Fristont KJ (2002) Systematic regularization of linear inverse solutions of the EEG source localization problem. Neuroimage 17(1):287–301
Phillips C, Mattout J, Rugg MD, Maquet P, Friston KJ (2005) An empirical Bayesian solution to the source reconstruction problem in EEG. Neuroimage 24(4):997–1011
Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL (2001) A default mode of brain function. Proc Natl Acad Sci USA 98(2):676–682
Shmuel A, Augath M, Oeltermann A, Logothetis NK (2006) Negative functional MRI response correlates with decreases in neuronal activity in monkey visual area V1. Nat Neurosci 9(4):569–577
Trujillo-Barreto N, Martinez-Montes E, Melie-Garcia L, Valdes-Sosa P (2001) A symmetrical Bayesian model for fMRI and EEG/MEG neuroimage fusion. Int J Bioelectromagn 3(1):1
Wipf D, Nagarajan S (2009) A unified Bayesian framework for MEG/EEG source imaging. Neuroimage 44(3):947–966
Yao D (2001) A method to standardize a reference of scalp EEG recordings to a point at infinity. Physiol Meas 22(4):693–711
Acknowledgments
This project was funded by grants from the National Nature Science Foundation of China #60736029, the 863 Project 2009AA02Z301 and the 973 project 2011CB707803. The authors are grateful to the FIL methods group (http://www.fil.ion.ucl.ac.uk) for providing the data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lei, X., Hu, J. & Yao, D. Incorporating fMRI Functional Networks in EEG Source Imaging: A Bayesian Model Comparison Approach. Brain Topogr 25, 27–38 (2012). https://doi.org/10.1007/s10548-011-0187-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-011-0187-9