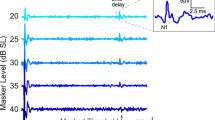
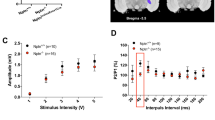
Abstract
Biotinidase deficiency is an autosomal recessively inherited disorder that results in the inability to recycle the vitamin, biotin. If untreated, the disorder can result in a range of neurological and cutaneous symptoms, including sensorineural deficits and deafness. To understand early mechanistic abnormalities that may precede more generalized and nonspecific effects of metabolic deficits such as weight loss and acidosis, we have analyzed auditory brainstem responses (ABRs) in biotinidase-deficient knockout (Btd −/−) mice in the periweaning period with or without dietary biotin supplementation. We find significant increases in the latency of wave V of the ABR elicited by pure tone stimuli at one octave intervals, which precede substantial increases in ABR thresholds. Finer interpeak latency analyses of these changes indicate they are confined to the latter ABR waves associated with the CNS and likely reflect slowed brainstem transmission time. In contrast, peripheral nervous system conduction velocity appears normal. Further, we find that biotin-supplementation after the onset of symptoms reverses the latency shifts, which has significant relevance for early treatment in patients. Finally, ABR latencies in Btd −/− mice fed a biotin-supplemented diet for the first month of life appear refractory to transmission time slowing during a subsequent bout of biotin deficiency. These data suggest a transient vulnerability window for biotin deficiency in the auditory brainstem. Finally, we also observe a developmental vulnerability window involving follicular melanosome production or melanocyte survival. Sensorineural deafness precedes peripheral hearing loss in developmental biotinidase deficiency and is transient if rescued by dietary biotin within a short developmental window.





Similar content being viewed by others
References
Antonelli AR, Bellotto R, Grandori F (1987) Audiologic diagnosis of central versus eighth nerve and cochlear auditory impairment. Audiology 26:209–226
Araki S, Mizuta K, Takeshita T, Morita H, Mineta H, Hoshino T (2002) Degeneration of the stria vascularis during development in melanocyte-deficient mutant rats (Ws/Ws rats). Eur Arch Otorhinolaryngol 259:309–315
Boison D, Stoffel W (1994) Disruption of the compacted myelin sheath of axons of the central nervous system in proteolipid protein-deficient mice. Proc Natl Acad Sci U S A 91:11709–11713
Box GEP, Cox DR (1964) An analysis of transformations. J Royal Stat Soc, Series B 26:211–252
Brumwell CL, Hossain WA, Morest DK, Wolf B (2005) Biotinidase reveals the morphogenetic sequence in cochlea and cochlear nucleus of mice. Hear Res 209:104–121
Denninger AR, Breglio A, Maheras KJ et al (2015) Claudin-11 tight junctions in myelin are a barrier to diffusion and lack strong adhesive properties. Biophys J 109:1387–1397
Devaux JJ, Gow A (2008) Tight junctions potentiate the insulative properties of small CNS myelinated axons. J Cell Biol 183:909–921
El-Hattab AW, Adesina AM, Jones J, Scaglia F (2015) MELAS syndrome: clinical manifestations, pathogenesis, and treatment options. Mol Genet Metab 116:4–12
Fabiani M, Sohmer H, Tait C, Gafni M, Kinarti R (1979) A functional measure of brain activity: brain stem transmission time. Electroencephalogr Clin Neurophysiol 47:483–491
Fox J, Weisberg S (2011) An R companion to applied regression. Sage, Thousand Oaks http://z.umn.edu/carbook
Gow A, Devaux JJ (2008) A model of tight junction function in CNS myelinated axons. Neuron Glia Biol 4:307–317
Gow A, Southwood CM, Li JS et al (1999) CNS myelin and Sertoli cell tight junction strands are absent in Osp/Claudin 11-null mice. Cell 99:649–659
Gow A, Davies C, Southwood CM et al (2004) Deafness in Claudin 11-null mice reveals the critical contribution of basal cell tight junctions to stria vascularis function. J Neurosci 24:7051–7062
Heller AJ, Stanley C, Shaia WT, Sismanis A, Spencer RF, Wolf B (2002) Localization of biotinidase in the brain: implications for its role in hearing loss in biotinidase deficiency. Hear Res 173:62–68
Hernandez-Vazquez A, Wolf B, Pindolia K et al (2013) Biotinidase knockout mice show cellular energy deficit and altered carbon metabolism gene expression similar to that of nutritional biotin deprivation: clues for the pathogenesis in the human inherited disorder. Mol Genet Metab 110:248–254
Kabacoff RI (2015) R in action. Data analysis and graphics with R. Manning, Shelter Island
Kanfer J, Parenty M, Goujet-Zalc C et al (1989) Developmental expression of myelin proteolipid, basic protein, and 2′,3′-cyclic nucleotide 3′-phosphodiesterase transcripts in different rat brain regions. J Mol Neurosci 1:39–46
Karet FE, Finberg KE, Nelson RD et al (1999) Mutations in the gene encoding B1 subunit of H+−ATPase cause renal tubular acidosis with sensorineural deafness. Nat Genet 21:84–90
LeVine SM, Macklin WB (1988) Biotin enrichment in oligodendrocytes in the rat brain. Brain Res 444:199–203
Maheras KJ, Gow A (2013) Increased anesthesia time using 2,2,2-tribromoethanol-chloral hydrate with low impact on mouse psychoacoustics. J Neurosci Methods 219:61–69
Melcher JR, Kiang NY (1996) Generators of the brainstem auditory evoked potential in cat. III: identified cell populations. Hear Res 93:52–71
Parham K, Sun X-M, Kim DO (2001) Noninvasive assessment of auditory function in mice: auditory brainstem response and distortion product otoacoustic emmissions. In: Willott JF (ed) Handbook of mouse auditory Research from behavior to molecular biology. CRC, Boca Raton, pp 37–58
Pena EA, Slate EH (2006) Global validation of linear model assumptions. J Am Stat Assoc 101:341
Peters TA, Kuijpers W, Tonnaer EL, van Muijen GN, Jap PH (1995) Distribution and features of melanocytes during inner ear development in pigmented and albino rats. Hear Res 85:169–180
Pindolia K, Jordan M, Guo C et al (2011) Development and characterization of a mouse with profound biotinidase deficiency: a biotin-responsive neurocutaneous disorder. Mol Genet Metab 102:161–169
Quevedo WC Jr (1956) Effect of biotin deficiency on follicular melanocytes of mice. Proc Soc Exp Biol Med 93:260–263
Rybak LP, Whitworth C, Scott V, Weberg AD, Bhardwaj B (1991) Rat as a potential model for hearing loss in biotinidase deficiency. Ann Otol Rhinol Laryngol 100:294–300
Salamy A, McKean CM, Buda FB (1975) Maturational changes in auditory transmission as reflected in human brain stem potentials. Brain Res 96:361–366
Sharma R, Jiang H, Zhong L, Tseng J, Gow A (2007) Minimal role for activating transcription factor 3 in the oligodendrocyte unfolded protein response in vivo. J Neurochem 102:1703–1712
Sivri HS, Genc GA, Tokatli A et al (2007) Hearing loss in biotinidase deficiency: genotype-phenotype correlation. J Pediatr 150:439–442
Sommerlad SF, Morton JM, Haile-Mariam M, Johnstone I, Seddon JM, O’Leary CA (2012) Prevalence of congenital hereditary sensorineural deafness in Australian cattle dogs and associations with coat characteristics and sex. BMC Vet Res 8:202
Steel KP, Barkway C (1989) Another role for melanocytes: their importance for normal stria vascularis development in the mammalian inner ear. Development (Cambridge, England) 107:453–463
Tachibana M (1999) Sound needs sound melanocytes to be heard. Pigment Cell Res 12:344–354
Talebi H, Yaghini O (2016) Auditory neuropathy/Dyssynchrony in Biotinidase deficiency. J Audiol Otol 20:53–54
Team RC (2014) R: a language and environment for statistical computing. Vienna, Austria. http://www.R-project.org/: R Foundation for Statistical Computing
Team RS (2016) RStudio: integrated development for R. RStudio Inc, Boston. http://www.rstudio.com/
Verity AN, Campagnoni AT (1988) Regional expression of myelin protein genes in the developing mouse brain: in situ hybridization studies. J Neurosci Res 21:238–248
Walsh EJ, McGee J, Javel E (1986) Development of auditory-evoked potentials in the cat. II. Wave latencies. J Acoustic Soc Am 79:725–744
Wolf B (1991) Worldwide survey of neonatal screening for biotinidase deficiency. J Inherit Metab Dis 14:923–927
Wolf B (2012a) Biotinidase deficiency. University of Washington, Seattle, WA
Wolf B (2012b) Biotinidase deficiency: “if you have to have an inherited metabolic disease, this is the one to have”. Genet Med 14:565–575
Wolf B (2015) Biotinidase deficiency should be considered in individuals exhibiting myelopathy with or without and vision loss. Mol Genet Metab 116:113–118
Wolf B, Grier RE, Allen RJ, Goodman SI, Kien CL (1983a) Biotinidase deficiency: the enzymatic defect in late-onset multiple carboxylase deficiency. Clin Chim Acta 131:273–281
Wolf B, Grier RE, Heard GS, Wilcken B, Hammond J (1983b) Hearing loss in biotinidase deficiency. Lancet 322:1365–1366
Wolf B, Spencer R, Gleason T (2002) Hearing loss is a common feature of symptomatic children with profound biotinidase deficiency. J Pediatr 140:242–246
Wu SH, Kelly JB (1991) Physiological properties of neurons in the mouse superior olive: membrane characteristics and postsynaptic responses studied in vitro. J Neurophysiol 65:230–246
Wu SH, Kelly JB (1992) Binaural interaction in the lateral superior olive: time difference sensitivity studied in mouse brain slice. J Neurophysiol 68:1151–1159
Zheng QY, Johnson KR, Erway LC (1999) Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hearing Res 130:94–107
Acknowledgements
We are grateful for technical assistance from Ms. Cisley Cardwell and Mr. Christian Brigolin. This work was supported by grants to A.G. from NIDCD, NIH (DC006262) and the National Multiple Sclerosis Society (RG4078, RG4639 and RG4906) and to B.W from the Safra Research Fund at the Henry Ford Health System.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Responsible editor: Gajja Salomons
Electronic supplementary material
ESM 1
(DOCX 25506 kb)
Rights and permissions
About this article
Cite this article
Maheras, K.J., Pindolia, K., Wolf, B. et al. Developmental window of sensorineural deafness in biotinidase-deficient mice. J Inherit Metab Dis 40, 733–744 (2017). https://doi.org/10.1007/s10545-017-0049-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-017-0049-z