Abstract
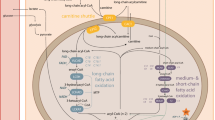
Short-chain acyl-CoA dehydrogenase deficiency (SCADD) is an inherited disorder of mitochondrial fatty acid oxidation that is characterized by the presence of increased butyrylcarnitine and ethylmalonic acid (EMA) concentrations in plasma and urine. Individuals with symptomatic SCADD may show relatively severe phenotype, while the majority of those who are diagnosed through newborn screening by tandem mass spectrometry may remain asymptomatic. As such, the associated clinical symptoms are very diverse, ranging from severe metabolic or neuromuscular disabilities to asymptomatic. Molecular analysis of affected individuals has identified rare gene variants along with two common gene variants, c.511C > T and c.625G > A. In vitro studies have demonstrated that the common variants as well as the great majority of rare variants, which are missense variants, impair folding, that may lead to toxic accumulation of the encoded protein, and/or metabolites, and initiate excessive production of ROS and chronic oxidative stress. It has been suggested that this cell toxicity in combination with yet unknown factors can trigger disease development. This association and the full implications of SCADD are not commonly appreciated. Accordingly, there is a worldwide discussion of the relationship of clinical manifestation to SCADD, and whether SCAD gene variants are disease associated at all. Therefore, SCADD is not part of the newborn screening programs in most countries, and consequently many patients with SCAD gene variants do not get a diagnosis and the possibilities to be followed up during development.



Similar content being viewed by others
Notes
Donor splice site strength was investigated using the Alamut software.
(http://www.interactive-biosoftware.com/doc/alamut-visual/2.6/splicing.html) with access to:
1. Splicesitefinder-like (SSF), 2. MaxEntScan, 3. NNSPLICE, 4. GeneSplicer, 5. Human Splicing Finder (HSF)
References
Amendt BA, Greene C, Sweetman L et al (1987) Short-chain acyl-coenzyme a dehydrogenase deficiency. Clinical and biochemical studies in two patients. J Clin Invest 79(5):1303–1309
Andresen BS, Bross P, Udvari S et al (1997) The molecular basis of medium-chain acyl-CoA dehydrogenase (MCAD) deficiency in compound heterozygous patients: is there correlation between genotype and phenotype? Hum Mol Genet 6(5):695–707
Armstrong DL, Masiowski ML, Wood PA (1993) Pathologic characterization of short-chain acyl-CoA dehydrogenase deficiency in BALB/cByJ mice. Am J Med Genet 47(6):884–892
Ashrafi G, Schwarz TL (2013) The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ 20(1):31–42
Baerlocher KE, Steinmann B, Aguzzi A, Krahenbuhl S, Roe CR, Vianey-Saban C (1997) Short-chain acyl-CoA dehydrogenase deficiency in a 16-year-old girl with severe muscle wasting and scoliosis. J Inherit Metab Dis 20(3):427–431
Barschak AG, Ferreira Gda C, Andre KR et al (2006) Inhibition of the electron transport chain and creatine kinase activity by ethylmalonic acid in human skeletal muscle. Metab Brain Dis 21(1):11–19
Battaile KP, Molin-Case J, Paschke R et al (2002) Crystal structure of rat short chain acyl-CoA dehydrogenase complexed with acetoacetyl-CoA: comparison with other acyl-CoA dehydrogenases. J Biol Chem 277(14):12200–12207
Bennett MJ (2010) Pathophysiology of fatty acid oxidation disorders. J Inherit Metab Dis 33(5):533–537
Bennett MJ, Rinaldo P, Strauss AW (2000) Inborn errors of mitochondrial fatty acid oxidation. Crit Rev Clin Lab Sci 37(1):1–44
Bentinger M, Tekle M, Dallner G (2010) Coenzyme Q--biosynthesis and functions. Biochem Biophys Res Commun 396(1):74–79
Berg JM, Tymoczko JL, Stryer L, Stryer L (2002) Biochemistry. Freeman, New York
Bhala A, Willi SM, Rinaldo P, Bennett MJ, Schmidt-Sommerfeld E, Hale DE (1995) Clinical and biochemical characterization of short-chain acyl-coenzyme a dehydrogenase deficiency. J Pediatr 126(6):910–915
Birkebaek NH, Simonsen H, Gregersen N (2002) Hypoglycaemia and elevated urine ethylmalonic acid in a child homozygous for the short-chain acyl-CoA dehydrogenase 625G > a gene variation. Acta Paediatr 91(4):480–482
Bok LA, Vreken P, Wijburg FA et al (2003) Short-chain acyl-CoA dehydrogenase deficiency: studies in a large family adding to the complexity of the disorder. Pediatrics 112(5):1152–1155
Bonnefont JP, Bastin J, Behin A, Djouadi F (2009) Bezafibrate for an inborn mitochondrial beta-oxidation defect. N Engl J Med 360(8):838–840
Bonnefont JP, Bastin J, Laforet P et al (2010) Long-term follow-up of bezafibrate treatment in patients with the myopathic form of carnitine palmitoyltransferase 2 deficiency. Clin Pharmacol Ther 88(1):101–108
Brieger K, Schiavone S, Miller FJ Jr, Krause KH (2012) Reactive oxygen species: from health to disease. Swiss Med Wkly 142:w13659
Bross P, Jespersen C, Jensen TG et al (1995) Effects of two mutations detected in medium chain acyl-CoA dehydrogenase (MCAD)-deficient patients on folding, oligomer assembly, and stability of MCAD enzyme. J Biol Chem 270(17):10284–10290
Bross P, Corydon TJ, Andresen BS, Jorgensen MM, Bolund L, Gregersen N (1999) Protein misfolding and degradation in genetic diseases. Hum Mutat 14(3):186–198
Chandel NS (2014) Mitochondria as signaling organelles. BMC Biol 12:34
Chen JS, Faller DV, Spanjaard RA (2003) Short-chain fatty acid inhibitors of histone deacetylases: promising anticancer therapeutics? Curr Cancer Drug Targets 3(3):219–236
Chowdhury I, Mo Y, Gao L, Kazi A, Fisher AB, Feinstein SI (2009) Oxidant stress stimulates expression of the human peroxiredoxin 6 gene by a transcriptional mechanism involving an antioxidant response element. Free Radic Biol Med 46(2):146–153
Circu ML, Aw TY (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 48(6):749–762
Coates PM, Hale DE, Finocchiaro G, Tanaka K, Winter SC (1988) Genetic deficiency of short-chain acyl-coenzyme a dehydrogenase in cultured fibroblasts from a patient with muscle carnitine deficiency and severe skeletal muscle weakness. J Clin Invest 81(1):171–175
Cornelius N, Byron C, Hargreaves I et al (2013) Secondary coenzyme Q10 deficiency and oxidative stress in cultured fibroblasts from patients with riboflavin responsive multiple acyl-CoA dehydrogenation deficiency. Hum Mol Genet 22(19):3819–3827
Cornelius N, Corydon TJ, Gregersen N, Olsen RK (2014) Cellular consequences of oxidative stress in riboflavin responsive multiple acyl-CoA dehydrogenation deficiency patient fibroblasts. Hum Mol Genet 23(16):4285–4301
Corydon MJ, Gregersen N, Lehnert W et al (1996) Ethylmalonic aciduria is associated with an amino acid variant of short chain acyl-coenzyme a dehydrogenase. Pediatr Res 39(6):1059–1066
Corydon MJ, Andresen BS, Bross P et al (1997) Structural organization of the human short-chain acyl-CoA dehydrogenase gene. Mamm Genome 8(12):922–926
Corydon MJ, Vockley J, Rinaldo P et al (2001) Role of common gene variations in the molecular pathogenesis of short-chain acyl-CoA dehydrogenase deficiency. Pediatr Res 49(1):18–23
Davie JR (2003) Inhibition of histone deacetylase activity by butyrate. J Nutr 133(7 Suppl):2485S–2493S
Dawson DB, Waber L, Hale DE, Bennett MJ (1995) Transient organic aciduria and persistent lacticacidemia in a patient with short-chain acyl-coenzyme a dehydrogenase deficiency. J Pediatr 126(1):69–71
Dietzen DJ, Rinaldo P, Whitley RJ et al (2009) National academy of clinical biochemistry laboratory medicine practice guidelines: follow-up testing for metabolic disease identified by expanded newborn screening using tandem mass spectrometry; executive summary. Clin Chem 55(9):1615–1626
Ding WX, Yin XM (2012) Mitophagy: mechanisms, pathophysiological roles, and analysis. Biol Chem 393(7):547–564
Djouadi F, Aubey F, Schlemmer D et al (2005) Bezafibrate increases very-long-chain acyl-CoA dehydrogenase protein and mRNA expression in deficient fibroblasts and is a potential therapy for fatty acid oxidation disorders. Hum Mol Genet 14(18):2695–2703
Djouadi F, Habarou F, Le Bachelier C et al (2016) Mitochondrial trifunctional protein deficiency in human cultured fibroblasts: effects of bezafibrate. J Inherit Metab Dis 39(1):47–58
Duthie SJ, Grant MH (1989) The role of reductive and oxidative metabolism in the toxicity of mitoxantrone, adriamycin and menadione in human liver derived Hep G2 hepatoma cells. Br J Cancer 60(4):566–571
Ebert D, Haller RG, Walton ME (2003) Energy contribution of octanoate to intact rat brain metabolism measured by 13C nuclear magnetic resonance spectroscopy. J Neurosci 23(13):5928–5935
Echtay KS (2007) Mitochondrial uncoupling proteins--what is their physiological role? Free Radic Biol Med 43(10):1351–1371
Edhager AV, Stenbroen V, Nielsen NS et al (2014) Proteomic investigation of cultivated fibroblasts from patients with mitochondrial short-chain acyl-CoA dehydrogenase deficiency. Mol Genet Metab 111(3):360–368
Emenaker NJ, Calaf GM, Cox D, Basson MD, Qureshi N (2001) Short-chain fatty acids inhibit invasive human colon cancer by modulating uPA, TIMP-1, TIMP-2, mutant p53, Bcl-2, Bax, p21 and PCNA protein expression in an in vitro cell culture model. J Nutr 131(11 Suppl):3041S–3046S
Er E, Oliver L, Cartron PF, Juin P, Manon S, Vallette FM (2006) Mitochondria as the target of the pro-apoptotic protein Bax. Biochim Biophys Acta 1757(9–10):1301–1311
Fischer F, Hamann A, Osiewacz HD (2012) Mitochondrial quality control: an integrated network of pathways. Trends Biochem Sci 37(7):284–292
Fogg VC, Lanning NJ, Mackeigan JP (2011) Mitochondria in cancer: at the crossroads of life and death. Chin J Cancer 30(8):526–539
Fukushima A, Lopaschuk GD (2016) Acetylation control of cardiac fatty acid beta-oxidation and energy metabolism in obesity, diabetes, and heart failure. Biochim Biophys Acta 1862(12):2211–2220
Furuta S, Miyazawa S, Hashimoto T (1981) Purification and properties of rat liver acyl-CoA dehydrogenases and electron transfer flavoprotein. J Biochem 90(6):1739–1750
Gallant NM, Leydiker K, Tang H et al (2012) Biochemical, molecular, and clinical characteristics of children with short chain acyl-CoA dehydrogenase deficiency detected by newborn screening in California. Mol Genet Metab 106(1):55–61
Gobin-Limballe S, Djouadi F, Aubey F et al (2007) Genetic basis for correction of very-long-chain acyl-coenzyme a dehydrogenase deficiency by bezafibrate in patient fibroblasts: toward a genotype-based therapy. Am J Hum Genet 81(6):1133–1143
Greer JB, O’Keefe SJ (2011) Microbial induction of immunity, inflammation, and cancer. Front Physiol 1:168
Gregersen N, Bross P (2010) Protein misfolding and cellular stress: an overview. Methods Mol Biol 648:3–23
Gregersen N, Olsen RK (2010) Disease mechanisms and protein structures in fatty acid oxidation defects. J Inherit Metab Dis 33(5):547–553
Gregersen N, Winter VS, Corydon MJ et al (1998) Identification of four new mutations in the short-chain acyl-CoA dehydrogenase (SCAD) gene in two patients: one of the variant alleles, 511C-->T, is present at an unexpectedly high frequency in the general population, as was the case for 625G-->a, together conferring susceptibility to ethylmalonic aciduria. Hum Mol Genet 7(4):619–627
Gregersen N, Andresen BS, Corydon MJ et al (2001a) Mutation analysis in mitochondrial fatty acid oxidation defects: exemplified by acyl-CoA dehydrogenase deficiencies, with special focus on genotype-phenotype relationship. Hum Mutat 18(3):169–189
Gregersen N, Bross P, Andrese BS, Pedersen CB, Corydon TJ, Bolund L (2001b) The role of chaperone-assisted folding and quality control in inborn errors of metabolism: protein folding disorders. J Inherit Metab Dis 24(2):189–212
Gregersen N, Bross P, Andresen BS (2004) Genetic defects in fatty acid beta-oxidation and acyl-CoA dehydrogenases. Molecular pathogenesis and genotype-phenotype relationships. European journal of biochemistry / FEBS 271(3):470–482
Gregersen N, Bross P, Vang S, Christensen JH (2006) Protein misfolding and human disease. Annu Rev Genomics Hum Genet 7:103–124
Gregersen N, Andresen BS, Pedersen CB, Olsen RK, Corydon TJ, Bross P (2008) Mitochondrial fatty acid oxidation defects--remaining challenges. J Inherit Metab Dis 31(5):643–657
Hartl FU, Hayer-Hartl M (2002) Molecular chaperones in the cytosol: from nascent chain to folded protein. Science 295(5561):1852–1858
Henriques BJ, Rodrigues JV, Olsen RK, Bross P, Gomes CM (2009) Role of flavinylation in a mild variant of multiple acyl-CoA dehydrogenation deficiency: a molecular rationale for the effects of riboflavin supplementation. J Biol Chem 284(7):4222–4229
Hinnebusch BF, Meng S, Wu JT, Archer SY, Hodin RA (2002) The effects of short-chain fatty acids on human colon cancer cell phenotype are associated with histone hyperacetylation. J Nutr 132(5):1012–1017
Hinsdale ME, Kelly CL, Wood PA (1993) Null allele at Bcd-1 locus in BALB/cByJ mice is due to a deletion in the short-chain acyl-CoA dehydrogenase gene and results in missplicing of mRNA. Genomics 16(3):605–611
Hockenbery DM, Oltvai ZN, Yin XM, Milliman CL, Korsmeyer SJ (1993) Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 75(2):241–251
Holley AK, Bakthavatchalu V, Velez-Roman JM, St Clair DK (2011) Manganese superoxide dismutase: guardian of the powerhouse. Int J Mol Sci 12(10):7114–7162
Izai K, Uchida Y, Orii T, Yamamoto S, Hashimoto T (1992) Novel fatty acid beta-oxidation enzymes in rat liver mitochondria. I Purification and properties of very-long-chain acyl-coenzyme A dehydrogenase J Biol Chem 267(2):1027–1033
Jethva R, Bennett MJ, Vockley J (2008) Short-chain acyl-coenzyme a dehydrogenase deficiency. Mol Genet Metab 95(4):195–200
Kasser TR, Deutch A, Martin RJ (1986) Uptake and utilization of metabolites in specific brain sites relative to feeding status. Physiol Behav 36(6):1161–1165
Kasubuchi M, Hasegawa S, Hiramatsu T, Ichimura A, Kimura I (2015) Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients 7(4):2839–2849
Kelly CL, Wood PA (1996) Cloning and characterization of the mouse short-chain acyl-CoA dehydrogenase gene. Mamm Genome 7(4):262–264
Kelly CL, Hinsdale ME, Wood PA (1993) Cloning and characterization of the mouse short-chain acyl-CoA dehydrogenase cDNA. Genomics 18(1):137–140
Kelso GF, Porteous CM, Coulter CV et al (2001) Selective targeting of a redox-active ubiquinone to mitochondria within cells: antioxidant and antiapoptotic properties. J Biol Chem 276(7):4588–4596
Kim JJP, Wang M, Djordjevic S, Paschke R, Bennett DW (1994) Three dimensional structures of acyl-CoA dehydrogenases: structural basis of substrate specificity. In: Yagi K (ed) Flavins and flavoproteins. W. de Gruyter, Berlin; New York, pp 273–282
Kmoch S, Zeman J, Hrebicek M, Ryba L, Kristensen MJ, Gregersen N (1995) Riboflavin-responsive epilepsy in a patient with SER209 variant form of short-chain acyl-CoA dehydrogenase. J Inherit Metab Dis 18(2):227–229
Koeberl DD, Young SP, Gregersen NS et al (2003) Rare disorders of metabolism with elevated butyryl- and isobutyryl-carnitine detected by tandem mass spectrometry newborn screening. Pediatr Res 54(2):219–223
Kolvraa S, Gregersen N (1986) Acyl-CoA:glycine N-acyltransferase: organelle localization and affinity toward straight- and branched-chained acyl-CoA esters in rat liver. Biochem Med Metab Biol 36(1):98–105
Kowaltowski AJ, de Souza-Pinto NC, Castilho RF, Vercesi AE (2009) Mitochondria and reactive oxygen species. Free Radic Biol Med 47(4):333–343
Kristensen MJ, Kmoch S, Bross P, Andresen BS, Gregersen N (1994) Amino acid polymorphism (Gly209Ser) in the ACADS gene. Hum Mol Genet 3(9):1711
Kurian MA, Hartley L, Zolkipli Z et al (2004) Short-chain acyl-CoA dehydrogenase deficiency associated with early onset severe axonal neuropathy. Neuropediatrics 35(5):312–316
Lampret BR, Murko S, Debeljak M, Tansek MZ, Fister P, Battelino T (2015) A case report of short-chain acyl-CoA dehydrogenase deficiency (SCADD). Biochem Med (Zagreb) 25(2):279–284
Lane MD, Halenz DR, Kosow DP, Hegre CS (1960) Further studies on mitochondrial propionyl carboxylase. J Biol Chem 235:3082–3086
Lee BJ, Huang YC, Chen SJ, Lin PT (2012) Coenzyme Q10 supplementation reduces oxidative stress and increases antioxidant enzyme activity in patients with coronary artery disease. Nutrition 28(3):250–255
Li S, Li J, Ning L et al (2015) In silico Identification of protein S-Palmitoylation sites and their involvement in human inherited disease. J Chem Inf Model 55(9):2015–2025
Lindner M, Hoffmann GF, Matern D (2010) Newborn screening for disorders of fatty-acid oxidation: experience and recommendations from an expert meeting. J Inherit Metab Dis 33(5):521–526
Loor G, Kondapalli J, Schriewer JM, Chandel NS, Vanden Hoek TL, Schumacker PT (2010) Menadione triggers cell death through ROS-dependent mechanisms involving PARP activation without requiring apoptosis. Free Radic Biol Med 49(12):1925–1936
Lucas TG, Henriques BJ, Rodrigues JV, Bross P, Gregersen N, Gomes CM (2011) Cofactors and metabolites as potential stabilizers of mitochondrial acyl-CoA dehydrogenases. Biochim Biophys Acta 1812(12):1658–1663
Lupton JR (2004) Microbial degradation products influence colon cancer risk: the butyrate controversy. J Nutr 134(2):479–482
Matern D, Hart P, Murtha AP et al (2001) Acute fatty liver of pregnancy associated with short-chain acyl-coenzyme a dehydrogenase deficiency. J Pediatr 138(4):585–588
McGee DJ, George AE, Trainor EA, Horton KE, Hildebrandt E, Testerman TL (2011) Cholesterol enhances helicobacter pylori resistance to antibiotics and LL-37. Antimicrob Agents Chemother 55(6):2897–2904
Miao L, St Clair DK (2009) Regulation of superoxide dismutase genes: implications in disease. Free Radic Biol Med 47(4):344–356
Nagan N, Kruckeberg KE, Tauscher AL, Bailey KS, Rinaldo P, Matern D (2003) The frequency of short-chain acyl-CoA dehydrogenase gene variants in the US population and correlation with the C(4)-acylcarnitine concentration in newborn blood spots. Mol Genet Metab 78(4):239–246
Nagao M, Tanaka K (1992) FAD-dependent regulation of transcription, translation, post-translational processing, and post-processing stability of various mitochondrial acyl-CoA dehydrogenases and of electron transfer flavoprotein and the site of holoenzyme formation. J Biol Chem 267(25):17925–17932
Naito E, Ozasa H, Ikeda Y, Tanaka K (1989) Molecular cloning and nucleotide sequence of complementary DNAs encoding human short chain acyl-coenzyme a dehydrogenase and the study of the molecular basis of human short chain acyl-coenzyme a dehydrogenase deficiency. J Clin Invest 83(5):1605–1613
Naito E, Indo Y, Tanaka K (1990) Identification of two variant short chain acyl-coenzyme a dehydrogenase alleles, each containing a different point mutation in a patient with short chain acyl-coenzyme a dehydrogenase deficiency. J Clin Invest 85(5):1575–1582
Nalecz KA, Miecz D, Berezowski V, Cecchelli R (2004) Carnitine: transport and physiological functions in the brain. Mol Asp Med 25(5–6):551–567
Nelson DL, Cox MM, Lehninger AL (2005) Lehninger principles of biochemistry. Freeman, New York
Olsen RK, Cornelius N, Gregersen N (2013) Genetic and cellular modifiers of oxidative stress: what can we learn from fatty acid oxidation defects? Mol Genet Metab 110(Suppl):S31–S39
Olsen RK, Cornelius N, Gregersen N (2015) Redox signalling and mitochondrial stress responses; lessons from inborn errors of metabolism. J Inherit Metab Dis 38(4):703–719
Palmfeldt J, Vang S, Stenbroen V et al (2011) Proteomics reveals that redox regulation is disrupted in patients with ethylmalonic encephalopathy. J Proteome Res 10(5):2389–2396
Panov A, Orynbayeva Z, Vavilin V, Lyakhovich V (2014) Fatty acids in energy metabolism of the central nervous system. Biomed Res Int 2014:472459
Pedersen CB, Bross P, Winter VS et al (2003) Misfolding, degradation, and aggregation of variant proteins. The molecular pathogenesis of short chain acyl-CoA dehydrogenase (SCAD) deficiency. J Biol Chem 278(48):47449–47458
Pedersen CB, Kolvraa S, Kolvraa A et al (2008) The ACADS gene variation spectrum in 114 patients with short-chain acyl-CoA dehydrogenase (SCAD) deficiency is dominated by missense variations leading to protein misfolding at the cellular level. Hum Genet 124(1):43–56
Pedersen CB, Zolkipli Z, Vang S et al (2010) Antioxidant dysfunction: potential risk for neurotoxicity in ethylmalonic aciduria. J Inherit Metab Dis 33(3):211–222
Quinzii CM, Hirano M (2010) Coenzyme Q and mitochondrial disease. Dev Disabil Res Rev 16(2):183–188
Quinzii CM, Hirano M (2011) Primary and secondary CoQ(10) deficiencies in humans. Biofactors 37(5):361–365
Qureshi IA, Ratnakumari L, Michalak A, Giguere R, Cyr D, Butterworth RF (1993) A profile of cerebral and hepatic carnitine, ammonia, and energy metabolism in a model of organic aciduria: BALB/cByJ mouse with short-chain acyl-CoA dehydrogenase deficiency. Biochem Med Metab Biol 50(2):145–158
Ribes A, Riudor E, Garavaglia B et al (1998) Mild or absent clinical signs in twin sisters with short-chain acyl-CoA dehydrogenase deficiency. Eur J Pediatr 157(4):317–320
Saijo T, Tanaka K (1995) Isoalloxazine ring of FAD is required for the formation of the core in the Hsp60-assisted folding of medium chain acyl-CoA dehydrogenase subunit into the assembly competent conformation in mitochondria. J Biol Chem 270(4):1899–1907
Saijo T, Welch WJ, Tanaka K (1994) Intramitochondrial folding and assembly of medium-chain acyl-CoA dehydrogenase (MCAD). Demonstration of impaired transfer of K304E-variant MCAD from its complex with hsp60 to the native tetramer. J Biol Chem 269(6):4401–4408
Sauer SW, Okun JG, Hoffmann GF, Koelker S, Morath MA (2008) Impact of short- and medium-chain organic acids, acylcarnitines, and acyl-CoAs on mitochondrial energy metabolism. Biochim Biophys Acta 1777(10):1276–1282
Scharlau D, Borowicki A, Habermann N et al (2009) Mechanisms of primary cancer prevention by butyrate and other products formed during gut flora-mediated fermentation of dietary fibre. Mutat Res 682(1):39–53
Schmidt SP, Corydon TJ, Pedersen CB, Bross P, Gregersen N (2010) Misfolding of short-chain acyl-CoA dehydrogenase leads to mitochondrial fission and oxidative stress. Mol Genet Metab 100(2):155–162
Schmidt SP, Corydon TJ, Pedersen CB et al (2011) Toxic response caused by a misfolding variant of the mitochondrial protein short-chain acyl-CoA dehydrogenase. J Inherit Metab Dis 34(2):465–475
Schuck PF, Busanello EN, Moura AP et al (2010) Promotion of lipid and protein oxidative damage in rat brain by ethylmalonic acid. Neurochem Res 35(2):298–305
Schuck PF, Milanez AP, Felisberto F et al (2015) Brain and muscle redox imbalance elicited by acute ethylmalonic acid administration. PLoS One 10(5):e0126606
Seidel J, Streck S, Bellstedt K et al (2003) Recurrent vomiting and ethylmalonic aciduria associated with rare mutations of the short-chain acyl-CoA dehydrogenase gene. J Inherit Metab Dis 26(1):37–42
Seppet E, Gruno M, Peetsalu A et al (2009) Mitochondria and energetic depression in cell pathophysiology. Int J Mol Sci 10(5):2252–2303
Sewell AC, Herwig J, Bohles H, Rinaldo P, Bhala A, Hale DE (1993) A new case of short-chain acyl-CoA dehydrogenase deficiency with isolated ethylmalonic aciduria. Eur J Pediatr 152(11):922–924
Shirao K, Okada S, Tajima G et al (2010) Molecular pathogenesis of a novel mutation, G108D, in short-chain acyl-CoA dehydrogenase identified in subjects with short-chain acyl-CoA dehydrogenase deficiency. Hum Genet 127(6):619–628
Tein I, Haslam RH, Rhead WJ, Bennett MJ, Becker LE, Vockley J (1999) Short-chain acyl-CoA dehydrogenase deficiency: a cause of ophthalmoplegia and multicore myopathy. Neurology 52(2):366–372
Tein I, Elpeleg O, Ben-Zeev B et al (2008) Short-chain acyl-CoA dehydrogenase gene mutation (c.319C>T) presents with clinical heterogeneity and is candidate founder mutation in individuals of Ashkenazi Jewish origin. Mol Genet Metab 93(2):179–189
Tilg H, Moschen AR (2014) Microbiota and diabetes: an evolving relationship. Gut 63(9):1513–1521
Tonin R, Caciotti A, Funghini S et al (2016) Clinical relevance of short-chain acyl-CoA dehydrogenase (SCAD) deficiency: exploring the role of new variants including the first SCAD-disease-causing allele carrying a synonymous mutation. BBA Clin 5:114–119
Toscani A, Soprano DR, Soprano KJ (1988) Molecular analysis of sodium butyrate-induced growth arrest. Oncogene Res 3(3):223–238
Turpin B, Tobias JD (2005) Perioperative management of a child with short-chain acyl-CoA dehydrogenase deficiency. Paediatr Anaesth 15(9):771–777
Turunen M, Olsson J, Dallner G (2004) Metabolism and function of coenzyme Q. Biochim Biophys Acta 1660(1–2):171–199
Vanhoutvin SA, Troost FJ, Hamer HM et al (2009) Butyrate-induced transcriptional changes in human colonic mucosa. PLoS One 4(8):e6759
van Maldegem BT, Waterham HR, Duran M et al (2005) The 625G>a SCAD gene variant is common but not associated with increased C4-carnitine in newborn blood spots. J Inherit Metab Dis 28(4):557–562
van Maldegem BT, Duran M, Wanders RJ et al (2006) Clinical, biochemical, and genetic heterogeneity in short-chain acyl-coenzyme a dehydrogenase deficiency. JAMA 296(8):943–952
van Maldegem BT, Duran M, Wanders RJ et al (2010a) Fasting and fat-loading tests provide pathophysiological insight into short-chain acyl-coenzyme a dehydrogenase deficiency. J Pediatr 156(1):121–127
van Maldegem BT, Duran M, Wanders RJ, Waterham HR, Wijburg FA (2010b) Flavin adenine dinucleotide status and the effects of high-dose riboflavin treatment in short-chain acyl-CoA dehydrogenase deficiency. Pediatr Res 67(3):304–308
van Maldegem BT, Wanders RJ, Wijburg FA (2010c) Clinical aspects of short-chain acyl-CoA dehydrogenase deficiency. J Inherit Metab Dis 33(5):507–511
Viscomi C, Burlina AB, Dweikat I et al (2010) Combined treatment with oral metronidazole and N-acetylcysteine is effective in ethylmalonic encephalopathy. Nat Med 16(8):869–871
Waisbren SE, Levy HL, Noble M et al (2008) Short-chain acyl-CoA dehydrogenase (SCAD) deficiency: an examination of the medical and neurodevelopmental characteristics of 14 cases identified through newborn screening or clinical symptoms. Mol Genet Metab 95(1–2):39–45
Wang G (2014) Human antimicrobial peptides and proteins. Pharmaceuticals (Basel) 7(5):545–594
Wang W, Mohsen AW, Uechi G et al (2014) Complex changes in the liver mitochondrial proteome of short chain acyl-CoA dehydrogenase deficient mice. Mol Genet Metab 112(1):30–39
Waterham HR, Koster J, van Roermund CW, Mooyer PA, Wanders RJ, Leonard JV (2007) A lethal defect of mitochondrial and peroxisomal fission. N Engl J Med 356(17):1736–1741
Wickner S, Maurizi MR, Gottesman S (1999) Posttranslational quality control: folding, refolding, and degrading proteins. Science 286(5446):1888–1893
Wilcken B, Wiley V, Hammond J, Carpenter K (2003) Screening newborns for inborn errors of metabolism by tandem mass spectrometry. N Engl J Med 348(23):2304–2312
Wong JM, de Souza R, Kendall CW, Emam A, Jenkins DJ (2006) Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol 40(3):235–243
Wood PA, Amendt BA, Rhead WJ, Millington DS, Inoue F, Armstrong D (1989) Short-chain acyl-coenzyme a dehydrogenase deficiency in mice. Pediatr Res 25(1):38–43
Wood PA, Amendt BA, Rhead WJ, Armstrong D, Millington DS, Inoue F (1990) A murine model for short-chain acyl-CoA dehydrogenase deficiency. Prog Clin Biol Res 321:427–434
Wu YT, Wu SB, Wei YH (2014) Metabolic reprogramming of human cells in response to oxidative stress: implications in the pathophysiology and therapy of mitochondrial diseases. Curr Pharm Des 20(35):5510–5526
Yamaguchi S, Li H, Purevsuren J et al (2012) Bezafibrate can be a new treatment option for mitochondrial fatty acid oxidation disorders: evaluation by in vitro probe acylcarnitine assay. Mol Genet Metab 107(1–2):87–91
Yonezawa H, Osaki T, Hanawa T et al (2012) Destructive effects of butyrate on the cell envelope of helicobacter pylori. J Med Microbiol 61(Pt 4):582–589
Young SP, Matern D, Gregersen N et al (2003) A comparison of in vitro acylcarnitine profiling methods for the diagnosis of classical and variant short chain acyl-CoA dehydrogenase deficiency. Clin Chim Acta 337(1–2):103–113
Zolkipli Z, Pedersen CB, Lamhonwah AM, Gregersen N, Tein I (2011) Vulnerability to oxidative stress in vitro in pathophysiology of mitochondrial short-chain acyl-CoA dehydrogenase deficiency: response to antioxidants. PLoS One 6(4):e17534
Acknowledgments
We thank our colleagues at the Research Unit for Molecular Medicine, who have contributed to the ideas and concepts discussed in the present review. The investigations of this work have been supported by the Danish Council of Medical Research (#4004-00548), Aarhus County Research Initiative, the John and Birthe Meyer Foundation, Department for Clinical Medicine, and the faculty of Health, Aarhus University.
Details of the contributions of individual authors
Zahra Nochi: Drafting the review article.
Rikke Katrine Jentoft Olsen: Critically revising the review article.
Niels Gregersen: Critically revising the review article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Responsible editor: Piero Rinaldo
Rights and permissions
About this article
Cite this article
Nochi, Z., Olsen, R.K.J. & Gregersen, N. Short-chain acyl-CoA dehydrogenase deficiency: from gene to cell pathology and possible disease mechanisms. J Inherit Metab Dis 40, 641–655 (2017). https://doi.org/10.1007/s10545-017-0047-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-017-0047-1