Abstract
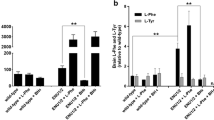
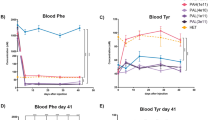
Monoamine neurotransmitter deficiency has been implicated in the etiology of neuropsychiatric symptoms associated with chronic hyperphenylalaninemia in phenylketonuria (PKU). Two proposed explanations for neurotransmitter deficiency in PKU include first, that chronically elevated blood L-phenylalanine (Phe) inhibits the transport of L-tyrosine (Tyr) and L-tryptophan (Trp), the substrates for dopamine and serotonin synthesis respectively, into brain. In the second hypothesis, elevated Phe competitively inhibits brain tyrosine hydroxylase (TH) and tryptophan hydroxylase (TPH) activities, the rate limiting steps in dopamine and serotonin synthesis. Dietary supplementation with large neutral amino acids (LNAA) including Tyr and Trp has been recommended for individuals with chronically elevated blood Phe in an attempt to restore amino acid and monoamine homeostasis in brain. As a potential alternative treatment approach, we demonstrate that pharmacologic inhibition of Tyr degradation through oral administration of nitisinone (NTBC) yielded sustained increases in blood and brain Tyr, decreased blood and brain Phe, and consequently increased dopamine synthesis in a murine model of PKU. Our results suggest that Phe-mediated inhibition of TH activity is the likely mechanism of impaired dopamine synthesis in PKU. Pharmacologic inhibition of Tyr degradation may be a promising adjunct therapy for CNS monoamine neurotransmitter deficiency in hyperphenylalaninemic individuals with PKU.






Similar content being viewed by others
Abbreviations
- PKU:
-
Phenylketonuria
- PAH:
-
Phenylalanine hydroxylase
- NTBC:
-
2-(2-nitro-4-trifluoromethylbenzoyl)-1,3-cyclohexanedione
- Phe:
-
L-phenylalanine
- Tyr:
-
L-tyrosine
- Trp:
-
L-tryptophan
- LNAA:
-
Large neutral amino acids
- L-DOPA:
-
L-3,4-dihydroxyphenylalanine
- DA:
-
Dopamine
- DOPAC:
-
L-3,4-dihydroxyphenylacetic acid
- 3-MT:
-
3-methoxytyramine
- HVA:
-
Homovanillic acid
- 5-HTP:
-
L-5-hydroxytryptophan
- 5-HIAA:
-
5-hydroxyindoleacetic acid
References
Azen CG, Koch R et al (1991) Intellectual development in 12-year-old children treated for phenylketonuria. Am J Dis Child 145(1):35–39
Batshaw ML, Valle D et al (1981) Unsuccessful treatment of phenylketonuria with tyrosine. J Pediatr 99(1):159–160
Choi TB, Pardridge WM (1986) Phenylalanine transport at the human blood–brain barrier. Studies with isolated human brain capillaries. J Biol Chem 261(14):6536–6541
Christ S, Huijbregts S et al (2010) Executive function in early-treated phenylketonuria: profile and underlying mechanisms. Mol Genet Metab 99(Suppl 1):S22–S32
Christensen HN, Streicher JA et al (1948) Effects of feeding individual amino acids upon the distribution of other amino acids between cells and extracellular fluid. J Biol Chem 172(2):515–524
De Groot MJ, Hoeksma M et al (2010) Pathogenesis of cognitive dysfunction in phenylketonuria: review of hypotheses. Mol Genet Metab 99(Suppl 1):S86–S89
Feillet F, Van Spronsen FJ et al (2010) Challenges and pitfalls in the management of phenylketonuria. Pediatrics 126(2):333–341
Harding C (2008) Progress toward cell-directed therapy for phenylketonuria. Clin Genet 74(2):97–104
Harding CO, Wild K et al (1998) Metabolic engineering as therapy for inborn errors of metabolism - development of mice with phenylalanine hydroxylase expression in muscle. Gene Ther 5(5):677–683
Introne WJ, Perry MB et al (2011) A 3-Year randomized therapeutic trial of nitisinone in alkaptonuria. Mol Genet Metab 103(4):307–314
Kanai Y, Segawa H et al (1998) Expression cloning and characterization of a transporter for large neutral amino acids activated by the heavy chain of 4f2 antigen (Cd98). J Biol Chem 273(37):23629–23632
Koshimura K, Miwa S et al (1990) Enhancement of dopamine release in vivo from the rat striatum by dialytic perfusion of 6r-L-erythro-5,6,7,8-tetrahydrobiopterin. J Neurochem 54(4):1391–1397
Lindstedt S, Holme E et al (1992) Treatment of hereditary tyrosinaemia type I by inhibition of 4-hydroxyphenylpyruvate dioxygenase. Lancet 340(8823):813–817
Lou HC, Lykkelund C et al (1987) Increased vigilance and dopamine synthesis by large doses of tyrosine or phenylalanine restriction in phenylketonuria. Acta Paediatr Scand 76(4):560–565
Lykkelund C, Nielsen JB et al (1988) Increased neurotransmitter biosynthesis in phenylketonuria induced by phenylalanine restriction or by supplementation of unrestricted diet with large amounts of tyrosine. Eur J Pediatr 148(3):238–245
Mastroberardino L, Spindler B et al (1998) Amino-acid transport by heterodimers of 4f2hc/Cd98 and members of a Permease family. Nature 395(6699):288–291
Matalon R, Michals-Matalon K et al (2006) Large neutral amino acids in the treatment of phenylketonuria (Pku). J Inherit Metab Dis 29(6):732–738
McDonald JD, Bode VC et al (1990) Pahhph-5: a mouse mutant deficient in phenylalanine hydroxylase. Proc Natl Acad Sci U S A 87(5):1965–1967
Miyamoto M, Fitzpatrick T (1957) Competitive inhibition of mammalian tyrosinase by phenylalanine and its relationship to hair pigmentation in phenylketonuria. Nature 179(4552):199–200
Ogburn KD, Bottiglieri T et al (2006) Prostaglandin J2 reduces Catechol-O-Methyltransferase activity and enhances dopamine toxicity in neuronal cells. Neurobiol Dis 22(2):294–301
Pascucci T, Ventura R et al (2002) Deficits in brain serotonin synthesis in a genetic mouse model of phenylketonuria. Neuroreport 13(18):2561–2564
Pascucci T, Andolina D et al (2009) 5-hydroxytryptophan rescues serotonin response to stress in prefrontal cortex of hyperphenylalaninaemic mice. Int J Neuropsychopharmacol 12(8):1067–1079
Pietz J, Kreis R et al (1999) Large neutral amino acids block phenylalanine transport into brain tissue in patients with phenylketonuria. J Clin Invest 103(8):1169–1178
Puglisi-Allegra S, Cabib S et al (2000) Dramatic brain aminergic deficit in a genetic mouse model of phenylketonuria. Neuroreport 11(6):1361–1364
Slocum RH, Cummings JG (1991) Amino acid analysis of physiological samples. In: Hommes FA (ed) Techniques in diagnostic human biochemical genetics: A laboratory manual. Wiley-Liss, New York, pp 87–126
Thompson AJ, Smith I et al (1990) Neurological deterioration in young adults with phenylketonuria. Lancet 336:602–605
Vanzutphen KH, Packman W et al (2007) Executive functioning in children and adolescents with phenylketonuria. Clin Genet 72(1):13–18
Vogel KR, Arning E et al (2013) Non-physiological amino acid (Npaa) therapy targeting brain phenylalanine reduction: pilot studies in Pahenu2 mice. J Inherit Metab Dis 36(3):513–523
Walter JH, White FJ (2004) Blood phenylalanine control in adolescents with phenylketonuria. Int J Adolesc Med Health 16(1):41–45
Acknowledgments
This work was supported in part by NIH Grants R01 DK059371 and R01 NS080866 and a grant from the National PKU Alliance (NPKUA, www.npkua.org).
We thank Gloria Baca, Baoyu Lin, Lindsey Stetson, and Katie Cobb for able technical assistance and Dr. Melanie Gillingham for critical review of the manuscript.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Nenad Blau
Rights and permissions
About this article
Cite this article
Harding, C.O., Winn, S.R., Gibson, K.M. et al. Pharmacologic inhibition of L-tyrosine degradation ameliorates cerebral dopamine deficiency in murine phenylketonuria (PKU). J Inherit Metab Dis 37, 735–743 (2014). https://doi.org/10.1007/s10545-013-9675-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-013-9675-2