Abstract
Background
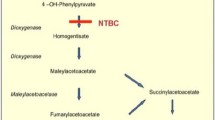
Tyrosinaemia type 1 (HT1) is a rare disorder leading to accumulation of toxic metabolites such as succinylacetone (SA) and a high risk of hepatocellular carcinoma. Children with HT1 traditionally required liver transplantation (OLT) and while the need for this has been reduced by the introduction of nitisinone some still require OLT. SA inhibits the enzyme porphobilinogen (PBG) synthase and its activity can be used as a marker of active SA. Elevated urinary SA post OLT has been reported previously. This study describes a novel finding of elevated plasma SA following OLT for HT1.
Methods
A retrospective analysis was performed of patients treated for HT1 at our institution from 1989-2010.
Results
Thirteen patients had an OLT for HT1. In patients who received nitisinone prior to OLT, mean urinary and plasma SA were elevated prior to treatment but normalised by the time of OLT (p ≤ 0.01). Mean PBG synthase activity increased from 0.032 to 0.99 nkat/gHb (ref range 0.58-1.25) at the time of OLT (p < 0.01). Mean urinary SA in patients not treated with nitisinone was also elevated prior to OLT; plasma levels and PBG synthase activity were not available prior to OLT for this group. Following OLT, mean urinary and plasma SA were elevated in all for the duration of follow-up and associated with low-normal PBG synthase activity.
Conclusion
Urinary and plasma SA levels are elevated following OLT for HT1. Low-normal PBG synthase activity suggests the plasma SA may be active. The clinical significance of this is unclear.


Similar content being viewed by others
References
Arnon R, Kerkar Davis, Anand Yin, Gonzalez-Peralta RP (2010) Liver transplantation in children with metabolic diseases: the studies of pediatric liver transplantation experience. Pediatr Transplant 14(6):796–805, available from: PM:20557477
Arnon R, Annunziato R, Miloh T et al (2011) Liver transplantation for hereditary tyrosinemia type I: analysis of the UNOS database. Pediatr Transplant 15(4):400–405, available from: PM:21504522
Arora-Gupta N, Davies P, McKiernan P, Kelly DA (2004) The effect of long-term calcineurin inhibitor therapy on renal function in children after liver transplantation. Pediatr Transplant 8(2):145–150, available from: PM:15049794
Bartlett DC, Lloyd C, Mirza D, McKiernan P, Newsome P (2010) Nitisinone treatment reduces the need for liver transplantation in children with tyrosinaemia type 1 and is associated with improved post-transplant renal tubular function. Gut 59(Suppl 2) Ref Type: Abstract
Collier HB (1971) A study of the determination of 5-aminolevulinate hydro-lyase (delta-aminolevulinate dehydratase) activity in hemolysates of human erythrocytes. Clin Biochem 4(4):222–232, available from: PM:5005890
El-Karaksy H, Rashed M, El-Sayed R et al (2010) Clinical practice. NTBC therapy for tyrosinemia type 1: how much is enough? Eur J Pediatr 169(6):689–693, available from: PM:19882170
Grenier A, Lescault A, Laberge C, Gagne R, Mamer O (1982) Detection of succinylacetone and the use of its measurement in mass screening for hereditary tyrosinemia. Clin Chim Acta 123(1-2):93–99, available from: PM:7116642
Greter J, Jacobson CE (1987) Urinary organic acids: isolation and quantification for routine metabolic screening. Clin Chem 33(4):473–480, available from: PM:3829377
Grompe M, St-Louis M, Demers SI, Al-Dhalimy M, Leclerc B, Tanguay RM (1994) A single mutation of the fumarylacetoacetate hydrolase gene in French Canadians with hereditary tyrosinemia type I. N Engl J Med 331(6):353–357, available from: PM:8028615
Herzog D, Martin S, Turpin S, Alvarez F (2006) Normal glomerular filtration rate in long-term follow-up of children after orthotopic liver transplantation. Transplantation 81(5):672–677
Hutchesson AC, Hall SK, Preece MA, Green A (1996) Screening for tyrosinaemia type I. Arch Dis Child Fetal Neonatal Ed 74(3):F191–F194, available from: PM:8777683
Laberge C, Grenier A, Valet JP, Morissette J (1990) Fumarylacetoacetase measurement as a mass-screening procedure for hereditary tyrosinemia type I. Am J Hum Genet 47(2):325–328, available from: PM:2378358
Laine J, Salo MK, Krogerus L, Karkkainen J, Wahlroos O, Holmberg C (1995) The nephropathy of type I tyrosinemia after liver transplantation. Pediatr Res 37(5):640–645, available from: PM:7603784
Lindstedt S, Holme E, Lock EA, Hjalmarson O, Strandvik B (1992) Treatment of hereditary tyrosinaemia type I by inhibition of 4-hydroxyphenylpyruvate dioxygenase. Lancet 340(8823):813–817, available from: PM:1383656
Mohan N, McKiernan P, Preece MA et al (1999) Indications and outcome of liver transplantation in tyrosinaemia type 1. Eur J Pediatr 158(Suppl 2):S49–S54, available from: PM:10603099
Paradis K (1996) Tyrosinemia: the Quebec experience. Clin Invest Med 19(5):311–316, available from: PM:8889268
Paradis K, D'Angata ID (1997) Nephropathy of tyrosinemia and its long-term outlook. J Pediatr Gastroenterol Nutr 24(1):113–114, available from: PM:9093996
Paradis K, Weber A, Seidman EG et al (1990) Liver transplantation for hereditary tyrosinemia: the Quebec experience. Am J Hum Genet 47(2):338–342, available from: PM:2378360
Pierik LJ, van Spronsen FJ, Bijleveld CM, van Dael CM (2005) Renal function in tyrosinaemia type I after liver transplantation: a long-term follow-up. J Inherit Metab Dis 28(6):871–876, available from: PM:16435179
Russo PA, Mitchell GA, Tanguay RM (2001) Tyrosinemia: a review. Pediatr Dev Pathol 4(3):212–221, available from: PM:11370259
Santra S, Preece MA, Hulton SA, Mckiernan PJ (2008) Renal tubular function in children with tyrosinaemia type I treated with nitisinone. J Inherit Metab Dis 31(3):399–402, available from: PM:18509744
Schwartz GJ, Brion LP, Spitzer A (1987) The use of plasma creatinine concentration for estimating glomerular filtration rate in infants, children, and adolescents. Pediatr Clin North Am 34(3):571–590, available from: PM:3588043
Shoemaker LR, Strife CF, Balistreri WF, Ryckman FC (1992) Rapid improvement in the renal tubular dysfunction associated with tyrosinemia following hepatic replacement. Pediatrics 89(2):251–255, available from: PM:1734392
Tuchman M, Whitley CB, Ramnaraine ML, Bowers LD, Fregien KD, Krivit W (1984) Determination of urinary succinylacetone by capillary gas chromatography. J Chromatogr Sci 22(5):211–215, available from: PM:6725495
Tuchman M, Freese DK, Sharp HL, Ramnaraine ML, Ascher N, Bloomer JR (1987) Contribution of extrahepatic tissues to biochemical abnormalities in hereditary tyrosinemia type I: study of three patients after liver transplantation. J Pediatr 110(3):399–403, available from: PM:3546650
Van Thiel DH, Gartner LM, Thorp FK et al (1986) Resolution of the clinical features of tyrosinemia following orthotopic liver transplantation for hepatoma. J Hepatol 3(1):42–48, available from: PM:3018074
Competing interests
Elisabeth Holme has received a royalty as an inventor of nitisinone. Patrick McKiernan has undertaken consultancy work for Swedish Orphan Biovitrum which has the worldwide rights to Orfadin (nitisinone).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: James V. Leonard
Rights and permissions
About this article
Cite this article
Bartlett, D.C., Preece, M.A., Holme, E. et al. Plasma succinylacetone is persistently raised after liver transplantation in tyrosinaemia type 1. J Inherit Metab Dis 36, 15–20 (2013). https://doi.org/10.1007/s10545-012-9482-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-012-9482-1