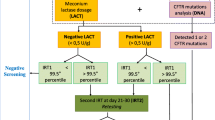
Abstract
Newborn screening (NBS) for cystic fibrosis (CF) offers the opportunity for early diagnosis and improved outcomes in patients with CF and has been universally available in the state of Massachusetts since 1999 using an immunoreactive trypsinogen (IRT)-DNA algorithm. Ideally, CF NBS is incorporated as part of an integrated NBS system that allows for comprehensive and coordinated education, laboratory screening, clinical follow-up, and evaluation so that evidence-based data can be used to maximize quality improvements and optimize the screening algorithm. The New England Newborn Screening Program (NENSP) retrospectively analyzed Massachusetts’s CF newborn screening data that yielded decisions to eliminate a screen-positive category, maintain the IRT cutoff value that prompts the second tier DNA testing, and communicate CF relative risk to primary care providers (PCPs) based on categorization of positive CF NBS results.

Similar content being viewed by others
References
Atkinson K, Zuckerman B, Sharfstein J, Levin D, Blatt RJR, Koh H (2001) A public health response to emerging technology: expansion of the Massachusetts newborn screening program. Public Health Rep 116:122–131
Borowitz D, Parad RB, Sharp JS et al (2009) Cystic Fibrosis Foundation practice guidelines for the management of infants with cystic fibrosis transmembrane conductance regulator-related metabolic syndrome during the first two years of life and beyond. J Pediatr 155(6, Suppl 1):S71–S116
Code of Massachusetts Regulations (2008): 105 CMR 270.000 sections 270.001-011. Blood screening of newborns for treatable diseases and disorders
Comeau AM, Parad RB, Dorkin HL et al (2004) Population-based newborn screening for genetic disorders when multiple mutation DNA testing is incorporated: a cystic fibrosis newborn screening model demonstrating increased sensitivity but more carrier detections. Pediatrics 113:1573–1581
Comeau AM, Accurso FJ, White TB et al (2007) Guidelines for implementation of cystic fibrosis newborn screening programs: Cystic Fibrosis Foundation workshop report. Pediatrics 119:2e495–e518
Cystic Fibrosis Foundation (CFF 2009) Newborn screening. http://www.cff.org/AboutCF/Testing/NewbornScreening/. Accessed November 27, 2009
Farrell PM, Rosenstein BJ, White TB et al (2008) Guidelines for diagnosis of cystic fibrosis in newborns through older adults: Cystic Fibrosis Foundation consensus report. J Pediatr 153(2):S4–S14
Grosse SD, Boyle CA, Botkin JR, Comeau AM, Kharrazi M, Rosenfeld M, Wilfond BS (2004) Newborn screening for cystic fibrosis: evaluation of benefits and risks and recommendations for state newborn screening programs. MMWR Recomm Rep 53(RR-13):1–36
Massachusetts Department of Public Health (2009) Massachusetts births 2007. Division of Research and Epidemiology, Bureau of Health Information, Statistics, Research, and Evaluation, Boston. http://www.mass.gov/Eeohhs2/docs/dph/research_epi/birth_report_2007.pdf . Accessed April 13, 2010
Therrell B (2009) National newborn screening status report (2009). National Newborn Screening and Genetics Resource Center, Austin. http://genes-r-us.uthscsa.edu/nbsdisorders.htm. Accessed December 31, 2009
Watson MS, Desnick RJ, Grody WW, Mennuti MT, Popovich BW, Richards CS (2002) Cystic fibrosis carrier screening: issues in implementation. Genet Med 4:407–409
Watson MS, Mann MY, Lloyd-Puryear, Rinaldo P (2006) Newborn screening: toward a uniform screening panel and system. Genet Med Suppl 1:1S–252S
Acknowledgments
This work was funded in part by The New England Newborn Screening Program of University of Massachusetts Medical School and HRSA grant U22MC03959 subcontract for Priority Focus 2.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Bridget Wilcken
Competing interest: None declared.
Rights and permissions
About this article
Cite this article
Hale, J.E., Parad, R.B., Dorkin, H.L. et al. Cystic fibrosis newborn screening: using experience to optimize the screening algorithm. J Inherit Metab Dis 33 (Suppl 2), 255–261 (2010). https://doi.org/10.1007/s10545-010-9117-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-010-9117-3