Abstract
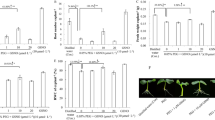
This study aimed to investigate seed germination, seedling growth, and antioxidative responses in two wheat cultivars, Ningchun and Xihan, exposed to different H2O2 concentrations. Ningchun exhibited higher germination rate but lower root and shoot growth than Xihan when exposed to H2O2 treatment. Assays using fluorescent dye H2DCFDA and propidium iodide showed a significantly enhanced H2O2 content and a cell elongation inhibition in H2O2-treated roots. The malondialdehyde content was elevated with increasing exogenous H2O2 concentration. Moreover, treatments of seedlings with H2O2 scavenger, catalase (CAT), and antioxidant, butylated hydroxytoluene, partly abolished H2O2-induced negative effect on root growth. In both untreated and H2O2-treated leaves, SOD activity in Ningchun was higher than that in Xihan, but POD and APX activities in Ningchun were lower than those in Xihan, leading to elevated H2O2 level in Ningchun leaves but decreased H2O2 content in Xihan ones under H2O2 treatment.
Similar content being viewed by others
Abbreviations
- APX:
-
ascorbate peroxidase
- ASA:
-
ascorbate
- BHT:
-
butylated hydroxytoluene
- CAT:
-
catalase
- EDTA:
-
ethylenediaminetetraacetic acid
- GR:
-
glutathione reductase
- H2DCFDA:
-
2’,7’-dichlorodihydrofluorescein diacetate
- MDA:
-
malondialdehyde
- NBT:
-
nitroblue tetrazolium
- PI:
-
propidium iodide
- POD:
-
peroxidase
- SOD:
-
superoxide dismutase
References
Aebi, H.: Catalase. - In: Bergmeyer, H.U. (ed.): Methods of Enzymatic Analysis. Pp. 673–677. Academic Press, New York 1974.
Bradford, M.M.: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. - Anal. Biochem. 72: 248–254, 1976.
Candan, N., Tarhan, L.: Tolerance or sensitivity responses of Mentha pulegium to osmotic and waterlogging stress in terms of antioxidant defense systems and membrane lipid peroxidation. - Environ. exp. Bot. 75: 83–88, 2012.
Cano, A., Artes, F., Arnao, M.B., Sanchez-Bravo, J., Costa, M.A.: Influence of peroxides, ascorbate and glutathione on germination and growth in Lupinus albus L.. - Biol. Plant. 39: 457–461, 1997.
Christophe, B., Hayat, E.M.B., Françoise, C.: From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. - Compt. Rend. Biol. 331: 806–814, 2008.
Cruz de Carvalho, H.M.: Drought stress and reactive oxygen species: production, scavenging and signaling. - Plant Signal Behav. 3: 156–165, 2008.
Dhindsa, R.S., Matowe, W.: Drought tolerance in two mosses: correlated with enzymatic defence against lipid peroxidation. - J. exp. Bot. 32: 79–91, 1981.
Donahue, J.L., Okpodu, C.M., Cramer, C.L., Grabau, E.A., Alscher, R.G.: Responses of antioxidants to paraquat in pea leaves. - Plant. Physiol. 113: 249–257, 1997.
Forman, H.J.: Use and abuse of exogenous H2O2 in studies of signal transduction. - Free Radical Biol. Med. 42: 926–932, 2007.
Foyer, C.H., Lelandais, M., Kunert, K.J.: Photooxidative stress in plants. - Physiol. Plant. 92: 696–717, 1994.
Fridovich, I.: Biological effects of the superoxide radical. - Arch. Biochem. Biophys. 247: 1–11, 1986.
Gechev, T.S., Van Breusegem, F., Stone, J.M., Denev, I., Laloi, C.: Reactive oxygen species as signals that modulate plant stress responses and programmed cell death. - Biol. Essays 28: 1091–1101, 2006.
Gondim, F.A., Gomes-Filho, E., Lacerda, C.F., Prisco, J.T., Azevedo Neto, A.D., Marques, E.C.: Pretreatment with H2O2 in maize seeds: effects on germination and seedling acclimation to salt stress. - Braz. J. Plant Physiol. 22: 103–112, 2010.
Goud, P.B., Kachole, M.S.: Effect of exogenous hydrogen on peroxide and polyphenol oxidase in Cajanus cajan (L.) Millsp. detached leaves. - Int. J. Current Res. 3: 61–65, 2011.
He, L.H., Gao, Z.Q., Li, R.Z.: Pretreatment of seed with H2O2 enhances drought tolerance of wheat (Triticum aestivum L.) seedlings. - African J. Biotechnol. 8: 6151–6157, 2009.
Kabir, M., Zafar Iqbal, M., Shafiq, M., Farooqi, Z.R.: Reduction in germination and seedling growth of Thespesia populnea L., caused by lead and cadmium treatments. - Pakistan J. Bot. 40: 2419–2426, 2008.
Karlsson, A., Nixon, J.B., McPhail, L.C.: Phorbol myristate acetate induces neutrophil NADPH-oxidase activity by two separate signal transduction pathways: dependent or independent of phosphatidylinositol 3-kinase. - J. Leuk. Biol. 67: 396–404, 2000.
Karuppanapandian, T., Moon, J.C., Kim, C., Manoharan, K., Kim, W.: Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. - Aust. J. Crop Sci. 5: 709–725, 2011.
Kennedy, D., Cronin, U.P., Wilkinson, M.G.: Responses of Escherichia coli, Listeria monocytogenes, and Staphylococcus aureus to simulated food processing treatments, determined using fluorescence-activated cell sorting and plate counting. - Appl. environ. Microbiol. 7: 4657–4668, 2011.
Li, J.T., Qiu, Z.B., Zhang, X.W., Wang, L.S.: Exogenous hydrogen peroxide can enhance tolerance of wheat seedlings to salt stress. - Acta Physiol. Plant. 30: 835–842, 2011.
Li, S.W., Xue, L.G., Xu, S.J., An, L.Z.: Hydrogen peroxide acts as a signal molecule in the adventitious root formation of mung bean seedlings. - Environ. exp. Bot. 65: 63–71, 2009.
Lin, C.C., Kao, C.H.: Cell wall peroxidase activity, hydrogen peroxide level and NaCl-inhibited root growth of rice seedlings. - Plant Soil 230: 135–143, 2001.
Liu, Y., Wan, Q., Wu, R., Wang, X., Wang, H., Wang, Z., Shi C., Bi, Y.: Role of hydrogen peroxide in regulating glucose-6-phosphate dehydrogenase activity under salt stress. - Biol. Plant. 56: 313–320, 2012.
Mittler, R.: Oxidative stress, antioxidants and stress tolerance. - Trends Plant Sci. 7: 405–410, 2002.
Nakano, Y., Asada, K.: Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. - Plant Cell Physiol. 22: 867–880, 1981.
Ogawa, K., Iwabuchi, M.: A mechanism for promoting the germination of Zinnia elegans seeds by hydrogen peroxide. - Plant Cell Physiol. 42: 286–291, 2001.
Ozden, M., Demirel, U., Kahraman, A.: Effects of proline on antioxidant system in leaves of grapevine (Vitis vinifera L.) exposed to oxidative stress by H2O2. - Sci. Hort. 119: 163–168, 2009.
Pei, Z.M., Murata, Y., Benning, G., Thomine, S., Klüsener, B., Allen, G., Grill, E., Schroeder, J.I.: Calcium channels activated by hydrogen peroxide mediate abscisic acid signaling in guard cells. - Nature 406: 731–734, 2000.
Peng, L.T., Yang, S.Z., Li, Q., Jiang, Y.M., Joyce, D.C.: Hydrogen peroxide treatments inhibit the browning of freshcut Chinese water chestnut. - Post Biol. Technol. 47: 260–266, 2008.
Rao, M.V., Paliyath, G., Ormrod, D.P.: Ultraviolet-B- and ozone-induced biochemical changes in antioxidant enzymes of Arabidopsis thaliana. - Plant Physiol. 110: 125–136, 1996.
Sergiev, I., Alexieva, V., Karanov, E.: Effect of spermine, atrazine and combination between them on some endogenous protective systems and stress markers in plants. - Compt. Rend. Acad. Bulg. Sci. 51: 121–124, 1997.
Singh, H.P., Batish, D.R., Kohli, R.K., Arora, K.: Arsenic-induced root growth inhibition in mung bean (Phaseolus aureus Roxb.) is due to oxidative stress resulting from enhanced lipid peroxidation. - Plant Growth Regul. 53: 65–73, 2007.
Upadhyay, R.K., Panda, S.K.: Copper-induced growth inhibition, oxidative stress and ultrastructural alterations in freshly grown water lettuce (Pistia stratiotes L.). - Compt. Rend. Biol. 332: 623–32, 2009.
Yamamoto, Y., Kobayashi, Y., Matsumoto, H.: Lipid peroxidation is an early symptom triggered by aluminum, but not the primary cause of elongation inhibition in pea roots. - Plant Physiol. 125: 199–208, 2001.
Yang, Y.L., Zhang, Y.Y., Lu, J., Zhang, H., Liu, Y., Jiang, Y., Shi, R.X.: Exogenous H2O2 treatment induced antioxidative responses and the signal regulation of proline accumulation in halophyte Nitraria tangutorum Bobr. callus. - Biol. Plant. 56: 330–336, 2012.
Zhang, X.L., Jia, X.F., Yu, B., Gao, Y., Bai, J.G.: Exogenous hydrogen peroxide influences antioxidant enzyme activity and lipid peroxidation in cucumber leaves at low light. - Sci. Hort. 129: 656–662, 2011.
Zhou, Q.: The measurement of malondialdehyde in plants. - In: Zhou, Q. (ed): Methods in Plant physiology. Pp. 72–74. China Agricultural Press, Beijing 2001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgement
This work was financially supported by the National Natural Science Foundation of China (No. 31160088) and Program for New Century Excellent Talents at University of China.
Rights and permissions
About this article
Cite this article
Lu, J., Li, X., Yang, Y. et al. Effect of hydrogen peroxide on seedling growth and antioxidants in two wheat cultivars. Biol Plant 57, 487–494 (2013). https://doi.org/10.1007/s10535-013-0312-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-013-0312-6