Abstract
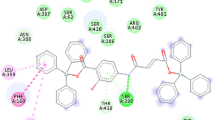
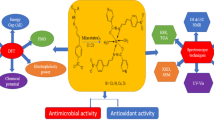
The diorganotin(IV) complexes (5–20) were synthesized in the present research from 4-fluorophenoxyacetic hydrazide and salicylaldehyde derivatives-based hydrazone ligands (1–4) to get an effective biological agent to combat microbial and oxidant deformities. Numerous spectral techniques such as (1H, 13C, 119Sn) NMR, UV–Vis, IR, and mass spectrometry were executed to illuminate the composition of complexes. These techniques ascertained tridentate chelation of hydrazone ligands with tin metal through enolic, phenolic oxygens and imine nitrogen, revealing pentacoordinated geometry of the complexes. The single crystal XRD of complex (5) confirmed distorted trigonal bipyramidal geometry. The TGA studies showed thermal stability up to 180 °C of the complexes, whereas the low conductance observed pointed to the non-electrolytic nature of the compounds. Furthermore, serial dilution assay was implemented to uncover the microbial inhibition efficacy (against six strains) of the compounds using ciprofloxacin and fluconazole. Among the synthesized compounds, (1, 8) exhibited comparable MIC value to standard. The compound (8) was reported as four times more potent than the fluconazole against C. albicans. Using DPPH assay, the antioxidant efficiency was examined which advocates enhanced efficacy of complexes than the ligands. The potency of complex (8) against C. albicans makes it a point of interest for molecular docking investigation, so, complex (8) and its ligand (1) were studied against protein of C. albicans (5TZ1), revealing the more efficacy of complex (binding energy-11.6 kcal/mol) than ligand. Further, the compounds were analysed for ADME prediction which concluded the efficacy of compounds as orally efficient pharmaceuticals.








Similar content being viewed by others
Data availability
The data that has been used is included in supplementary article and manuscript. The datasets generated and/or analysed during the current study are available in the Protein Data Bank (https://www.rcsb.org/) repository. The crystallographic files for complex (5) can be downloaded from the Cambridge Crystallographic Data Centre (CCDC No.-2291927).
References
Abbas Z, Kumar M, Tuli HS et al (2022) Synthesis, structural investigations, and in vitro/in silico bioactivities of flavonoid substituted biguanide: a novel Schiff base and its diorganotin (IV) complexes. Molecules 27:8874. https://doi.org/10.3390/molecules27248874
Abou-Melha KS, Al-Hazmi GA, Althagafi I et al (2021) Synthesis, characterization, DFT calculation, DNA binding and antimicrobial activities of metal complexes of dimedone arylhydrazone. J Mol Liq 334:116498. https://doi.org/10.1016/j.molliq.2021.116498
Ahmed YM, Mohamed GG (2022a) Synthesis, spectral characterization, antimicrobial evaluation and molecular docking studies on new metal complexes of novel Schiff base derived from 4,6-dihydroxy-1,3-phenylenediethanone. J Mol Struct 1256:132496. https://doi.org/10.1016/j.molstruc.2022.132496
Ahmed YM, Mohamed GG (2022b) New Tin(IV) Schiff base complexes: synthesis, characterization and antibacterial investigation, docking and theoretical studies. Inorg Chem Commun 144:109864. https://doi.org/10.1016/j.inoche.2022.109864
Anacona JR, Rincones M (2015) Tridentate hydrazone metal complexes derived from cephalexin and 2-hydrazinopyridine: synthesis, characterization and antibacterial activity. Spectrochim Acta A Mol Biomol Spectrosc 141:169–175. https://doi.org/10.1016/j.saa.2015.01.009
Aragón-Muriel A, Liscano Y, Upegui Y et al (2021) In vitro evaluation of the potential pharmacological activity and molecular targets of new benzimidazole-based Schiff base metal complexes. Antibiotics 10:728. https://doi.org/10.3390/antibiotics10060728
Arora T, Devi J, Dubey A et al (2023) Spectroscopic studies, antimicrobial activity, and computational investigations of hydrazone ligands endowed metal chelates. Appl Organomet Chem 37. https://doi.org/10.1002/aoc.7209
Banerjee S, Baidya SK, Ghosh B et al (2023) Quantitative structural assessments of potential meprin β inhibitors by non-linear QSAR approaches and validation by binding mode of interaction analysis. New J Chem 47:7051–7069. https://doi.org/10.1039/D2NJ04753E
Bedia K-K, Elçin O, Seda U et al (2006) Synthesis and characterization of novel hydrazide–hydrazones and the study of their structure–antituberculosis activity. Eur J Med Chem 41:1253–1261. https://doi.org/10.1016/j.ejmech.2006.06.009
Bel Haj Salah S, Hermi S, Alotaibi AA et al (2022) Stabilization of hexachloride net with mixed Sn(IV) metal complex and 2,3-dimethylanilinium organic cation: elaboration, optical, spectroscopic, computational studies and thermal analysis. Chem Pap 76:1861–1873. https://doi.org/10.1007/s11696-021-01974-4
Boora A, Devi J, Rom T, Paul AK (2023) Synthesis, characterization, single crystal structure, biological evaluation of ONO donor hydrazones and their diorganotin(IV) complexes. J Mol Struct 1284:135386. https://doi.org/10.1016/j.molstruc.2023.135386
Boora A, Devi J, Lal K, Rom T, Paul AK (2024). From synthesis to bioactivity: characterization, crystallography, antimalarial and antimicrobial activities of hydrazone-derived organotin (IV) complexes. Inorg Chim Acta 121940. https://doi.org/10.1016/j.ica.2024.121940
Butt SS, Badshah Y, Shabbir M, Rafiq M (2020) Molecular docking using chimera and autodock vina software for nonbioinformaticians. JMIR Bioinform Biotech 1:e14232. https://doi.org/10.2196/14232
Chans GM, Nieto-Camacho A, Ramírez-Apan T, Hernández-Ortega S, Álvarez-Toledano C, Gómez E (2015) Synthetic, spectroscopic, crystallographic, and biological studies of seven-coordinated Diorganotin (IV) complexes derived from Schiff bases and pyridinic carboxylic acids. Aust J Chem 69(3):279–290. https://doi.org/10.1071/CH15344
Chaston TB, Watts RN, Yuan J, Richardson DR (2004) Potent antitumor activity of novel iron chelators derived from di-2-pyridylketone isonicotinoyl hydrazone involves fenton-derived free radical generation. Clin Cancer Res 10:7365–7374. https://doi.org/10.1158/1078-0432.CCR-04-0865
Dawar N, Devi J, Kumar B, Dubey A (2023) Synthesis, Characterization, Pharmacological Screening, Molecular Docking, DFT, MESP, ADMET Studies of Transition Metal(II) Chelates of Bidentate Schiff Base Ligand. Inorg Chem Commun 151:110567. https://doi.org/10.1016/j.inoche.2023.110567
Devi J, Yadav J, Kumar D et al (2020) Synthesis, spectral analysis and in vitro cytotoxicity of diorganotin (IV) complexes derived from indole‐3‐butyric hydrazide. Appl Organomet Chem 34. https://doi.org/10.1002/aoc.5815
Devi J, Yadav J, Lal K et al (2021) Design, synthesis, crystal structure, molecular docking studies of some diorganotin(IV) complexes derived from the piperonylic hydrazide Schiff base ligands as cytotoxic agents. J Mol Struct 1232:129992. https://doi.org/10.1016/j.molstruc.2021.129992
Devi J, Kumar B, Taxak B (2022a) Recent advancements of organotin(IV) complexes derived from hydrazone and thiosemicarbazone ligands as potential anticancer agents. Inorg Chem Commun 139:109208. https://doi.org/10.1016/j.inoche.2022.109208
Devi J, Kumar S, Kumar B et al (2022b) Synthesis, structural analysis, in vitro antioxidant, antimicrobial activity and molecular docking studies of transition metal complexes derived from Schiff base ligands of 4-(benzyloxy)-2-hydroxybenzaldehyde. Res Chem Intermed 48:1541–1576. https://doi.org/10.1007/s11164-021-04644-y
Devi J, Sharma S, Kumar S et al (2022c) Synthesis, characterization, in vitro antimicrobial and cytotoxic studies of Co(II), Ni(II), Cu(II), and Zn(II) complexes obtained from Schiff base ligands of 1, 2, 3, 4‐tetrahydro‐naphthalen‐1‐ylamine. Appl Organomet Chem 36. https://doi.org/10.1002/aoc.6760
Divya K, Pinto GM, Pinto AF (2017) Application of metal complexes of Schiff bases as an antimicrobial drug: a review of recent works. Int J Curr Pharm Res 9(3):27–30. https://doi.org/10.22159/ijcpr.2017v9i3.19966
Dolomanov OV, Bourhis LJ, Gildea RJ et al (2009) OLEX2: a complete structure solution, refinement and analysis program. J Appl Crystallogr 42:339–341. https://doi.org/10.1107/S0021889808042726
EL-GammalAlshaterEl-Boraey OA H HA (2019) Schiff base metal complexes of 4-methyl-1H-indol-3-carbaldehyde derivative as a series of potential antioxidants and antimicrobial: synthesis, spectroscopic characterization and 3D molecular modeling. J Mol Struct 1195:220–230. https://doi.org/10.1016/j.molstruc.2019.05.101
Gordon CP, Raynaud C, Andersen RA et al (2019) Carbon-13 NMR chemical shift: a descriptor for electronic structure and reactivity of organometallic compounds. Acc Chem Res 52:2278–2289. https://doi.org/10.1021/acs.accounts.9b00225
Hameed A, al-Rashida M, Uroos M et al (2017) Schiff bases in medicinal chemistry: a patent review (2010–2015). Expert Opin Ther Pat 27:63–79. https://doi.org/10.1080/13543776.2017.1252752
Hashem HE, Nath A, Kumer A (2022) Synthesis, molecular docking, molecular dynamic, quantum calculation, and antibacterial activity of new Schiff base-metal complexes. J Mol Struct 1250:131915. https://doi.org/10.1016/j.molstruc.2021.131915
Hassan AU, Sumrra SH, Raza MA, Zubair M, Zafar MN, Mughal EU, Assiri MA (2021) Design, facile synthesis, spectroscopic characterization, and medicinal probing of metal-based new sulfonamide drugs: a theoretical and spectral study. Appl Organomet Chem 35(1):e6054
Ismail A, Al-Bairmani H, Abbas Z, Rheima A (2020) Nano-synthesis, spectroscopic characterisation and antibacterial activity of some metal complexes derived from Theophylline. Egypt J Chem 0:0–0. https://doi.org/10.21608/ejchem.2020.32582.2690
Jabeen M, Ahmad S, Shahid K et al (2018) Ursolic acid hydrazide based organometallic complexes: synthesis, characterization, antibacterial, antioxidant, and docking studies. Front Chem 6. https://doi.org/10.3389/fchem.2018.00055
Jaiswal N, Kushwaha AK, Singh AP, Dubey RK (2019) Synthesis, spectroscopic characterization and computational studies of Schiff base complexes of tin(IV) chloride. Main Group Met Chem 42:28–36. https://doi.org/10.1515/mgmc-2019-0002
Kostova I, Saso L (2013) Advances in research of Schiff-base metal complexes as potent antioxidants. Curr Med Chem 20(36):4609–4632
Kumar D, Sharma S, Kalra S et al (2020) Medicinal perspective of indole derivatives: recent developments and structure-activity relationship studies. Curr Drug Targets 21:864–891. https://doi.org/10.2174/1389450121666200310115327
Kumar B, Devi J, Dubey A, Tufail A, Arora T (2023a) Inspecting the anti-tuberculosis, antimicrobial, and anti-inflammatory efficiency of newly synthesized Co (II), Ni (II), Cu (II) and Zn (II) complexes of hydrazone ligands: characterization and computational studies. Appl Organomet Chem 37(12):e7291. https://doi.org/10.1002/aoc.7291
Kumar B, Devi J, Manuja A (2023b) Synthesis, structure elucidation, antioxidant, antimicrobial, anti-inflammatory and molecular docking studies of transition metal(II) complexes derived from heterocyclic Schiff base ligands. Res Chem Intermed 49:2455–2493. https://doi.org/10.1007/s11164-023-04991-y
L. I. G. A. N. D. Design, BIOVIA (2014) California. https://doi.org/10.12688/f1000research.5741.2
Li Y, Yang Z-Y, Wang M-F (2010) Synthesis, Characterization, DNA binding properties, fluorescence studies and antioxidant activity of transition metal complexes with hesperetin-2-hydroxy benzoyl hydrazone. J Fluoresc 20:891–905. https://doi.org/10.1007/s10895-010-0635-z
Manimohan M, Pugalmani S, Sithique MA (2019) Biologically active novel N, N, O donor tridentate water soluble hydrazide based O-carboxymethyl chitosan Schiff base Cu (II) metal complexes: synthesis and characterisation. Int J Biol Macromol 136:738–754. https://doi.org/10.1016/j.ijbiomac.2019.06.115
Mustafa G, Zia-ur-Rehman M, Sumrra SH, Ashfaq M, Zafar W, Ashfaq M (2022) A critical review on recent trends on pharmacological applications of pyrazolone endowed derivatives. J Mol Struct 1262:133044
Pachwania S, Devi J, Taxak B, Boora A (2023) Synthesis, characterization, and biological evaluation of organotin(IV) complexes derived from Schiff bases of 3-methoxybenzohydrazide. Phosphorus Sulfur Silicon Relat Elem 198:102–113. https://doi.org/10.1080/10426507.2022.2116637
Pangborn AB, Giardello MA, Grubbs RH, Rosen RK, Timmers FJ (1996) Safe and convenient procedure for solvent purification. Organometal 15(5):1518–1520. https://doi.org/10.1021/om9503712
Pettersen EF, Goddard TD, Huang CC et al (2004) UCSF Chimera – a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612. https://doi.org/10.1002/jcc.20084
Popiołek Ł (2017) Hydrazide–hydrazones as potential antimicrobial agents: overview of the literature since 2010. Med Chem Res 26:287–301. https://doi.org/10.1007/s00044-016-1756-y
Popiołek Ł, Tuszyńska K, Biernasiuk A (2022) Searching for novel antimicrobial agents among hydrazide-hydrazones of 4-iodosalicylic acid. Biomed Pharmacother 153:113302. https://doi.org/10.1016/j.biopha.2022.113302
Raman N, Dhaveethu Raja J, Sakthivel A (2007) Synthesis, spectral characterization of Schiff base transition metal complexes: DNA cleavage and antimicrobial activity studies. J Chem Sci 119:303–310. https://doi.org/10.1007/s12039-007-0041-5
Ramzan S, Shujah S, Holt KB et al (2023) Structural characterization, DNA binding study, antioxidant potential and antitumor activity of diorganotin(IV) complexes against human breast cancer cell line MDA-MB-231. J Organomet Chem 990:122671. https://doi.org/10.1016/j.jorganchem.2023.122671
Rani M, Devi J, Kumar B (2023) Thiosemicarbazones-based Co(II), Ni(II), Cu(II) and Zn(II) complexes: synthesis, structural elucidation, biological activities and molecular docking. Chem Pap 77:6007–6027. https://doi.org/10.1007/s11696-023-02917-x
Ridout M (2014) Serial dilution assay. Wiley StatsRef: Statistics Reference Online. https://doi.org/10.1002/9781118445112.stat05479
Sathyadevi P, Krishnamoorthy P, Butorac RR et al (2011) Effect of substitution and planarity of the ligand on DNA/BSA interaction, free radical scavenging and cytotoxicity of diamagnetic Ni(ii) complexes: a systematic investigation. Dalton Trans 40:9690. https://doi.org/10.1039/c1dt10767d
Sebbar NK, Ellouz M, Essassi EM et al (2015) Crystal structure of 4-benzyl-2 H -benzo[b][1,4]thiazin-3(4 H)-one. Acta Crystallogr E Crystallogr Commun 71:o999–o999. https://doi.org/10.1107/S2056989015022276
Sedaghat T, Yousefi M, Bruno G et al (2014) Synthesis, spectral characterization, crystal structure and antibacterial studies of diorganotin(IV) complexes with isonicotinoyl hydrazone derivatives. Polyhedron 79:88–96. https://doi.org/10.1016/j.poly.2014.04.061
Sedaghat T, Aminian M, Abaee S et al (2023) New organotin(IV) complexes with a bis-acyl-hydrazone ligand: synthesis, crystal structure and immobilization on magnetic mesoporous silica nanoparticles as a strategy in cancer therapy. J Mol Struct 1275:134622. https://doi.org/10.1016/j.molstruc.2022.134622
Shaaban S, Adam MSS, El-Metwaly NM (2022) Novel organoselenium-based N-mealanilic acid and its zinc (II) chelate: catalytic, anticancer, antimicrobial, antioxidant, and computational assessments. J Mol Liq 363:119907. https://doi.org/10.1016/j.molliq.2022.119907
Shah SS, Shah D, Khan I, Ahmed S, Ali U, Rehman A et al (2020) Synthesis and antioxidant activities of Schiff bases and their complexes: an updated review. Biointerface Res Appl Chem 10:6936–6963. https://doi.org/10.33263/BRIAC106.69366963
Sharfalddin AA, Emwas A, Jaremko M, Hussien MA (2021) Transition metal complexes of 6‐mercaptopurine: characterization, theoretical calculation, DNA‐Binding, molecular docking, and anticancer activity. Appl Organomet Chem 35. https://doi.org/10.1002/aoc.6041
Sharma AK, Chandra S (2013) Synthesis, structural and fungicidal studies of hydrazone based coordination compounds. Spectrochim Acta A Mol Biomol Spectrosc 103:96–100. https://doi.org/10.1016/j.saa.2012.11.012
Shujah S, Ali S, Khalid N et al (2022) Supramolecular diorganotin(IV) complexes of N′-(2-hydroxynaphthalen-1-yl)methylene)formohydrazide: synthesis, spectroscopic characterization, X-ray structure, antibacterial screening, cytotoxicity and docking study. Polyhedron 215:115678. https://doi.org/10.1016/j.poly.2022.115678
Singh J, Singh P (2012) Synthesis, spectroscopic characterization, and in vitro antimicrobial studies of pyridine-2-carboxylic acid N′-(4-Chloro-Benzoyl)-hydrazide and its Co(II), Ni(II), and Cu(II) complexes. Bioinorg Chem Appl 2012:1–7. https://doi.org/10.1155/2012/104549
Sumrra SH, Chohan ZH (2013) Antibacterial and antifungal oxovanadium (IV) complexes of triazole-derived Schiff bases. Med Chem Res 22:3934–3942. https://doi.org/10.1007/s00044-012-0388-0
Sumrra SH, Mushtaq F, Khalid M, Raza MA, Nazar MF, Ali B, Braga AA (2018) Synthesis, spectral characterization and computed optical analysis of potent triazole based compounds. Spectrochim Acta A Mol 190:197–207. https://doi.org/10.1016/j.saa.2017.09.019
Sumrra SH, Mushtaq F, Ahmad F, Hussain R, Zafar W, Imran M, Zafar MN (2022) Coordination behavior, structural, statistical and theoretical investigation of biologically active metal-based isatin compounds. Chem Pap 76(6):3705–3727. https://doi.org/10.1007/s11696-022-02123-1
Taxak B, Devi J, Kumar S, Asija S (2023a) Exploring the antimalarial, antioxidant and antimicrobial properties of newly synthesized diorganotin(IV) complexes with ONO-donor hydrazone ligands. Inorg Chem Commun 158:111473. https://doi.org/10.1016/j.inoche.2023.111473
Taxak B, Devi J, Dubey A, Kumar B, Tufail A, Pachwania S, Boora A (2023b) Investigation of anti-inflammatory and antimicrobial activities of hydrazone-based diorganotin (IV) complexes: synthesis, spectroscopic characterization, and computational studies. Appl Organomet Chem. https://doi.org/10.1002/aoc.7323
Toh-Boyo GM, Njong RN, Babette EM, Nfor EN (2021) Synthesis, spectroscopic, molecular modeling and anti-fungal studies of some divalent metal complexes of 4-hydroxyacetophenone isonicotinoyl hydrazone. Open J Inorg Chem 11:95–109. https://doi.org/10.4236/ojic.2021.113007
Uddin MN, Ahmed SS, Alam SMR (2020) Review: Biomedical applications of Schiff base metal complexes. J Coord Chem 73:3109–3149. https://doi.org/10.1080/00958972.2020.1854745
Ullah H, Previtali V, Mihigo HB et al (2019) Structure-activity relationships of new Organotin(IV) anticancer agents and their cytotoxicity profile on HL-60, MCF-7 and HeLa human cancer cell lines. Eur J Med Chem 181:111544. https://doi.org/10.1016/j.ejmech.2019.07.047
Vinusha HM, Kollur SP, Revanasiddappa HD et al (2019) Preparation, spectral characterization and biological applications of Schiff base ligand and its transition metal complexes. Results Chem 1:100012. https://doi.org/10.1016/j.rechem.2019.100012
Weng J-Q, Tan C-X, Liu X-H (2012) Synthesis and fungicidal activity of hydrazones containing 4-methylbenzo[d]thiazole moiety. J Pestic Sci 37:164–168. https://doi.org/10.1584/jpestics.G11-41
Yadav M, Yadav D, Pal Singh D, Kumar Kapoor J (2023) Pharmaceutical properties of macrocyclic Schiff base transition metal complexes: urgent need in today’s world. Inorganica Chim Acta 546:121300. https://doi.org/10.1016/j.ica.2022.121300
Yang Z, Li P, Gan X (2018) Novel pyrazole-hydrazone derivatives containing an isoxazole moiety: design, synthesis, and antiviral activity. Molecules 23:1798. https://doi.org/10.3390/molecules23071798
Yasmeen S, Sumrra SH, Akram MS, Chohan ZH (2017) Antimicrobial metal-based thiophene derived compounds. J Enzyme Inhib Med Chem 32(1):106–112. https://doi.org/10.1080/14756366.2016.1238363
Acknowledgements
Ms. Bharti Taxak (SRF) extends her heartfelt appreciation to the CSIR, New Delhi, India, the Deptt. of Chemistry, Guru Jambheshwar University of Science & Technology, Hisar, India and Dr. APJ Abdul Kalam Central Instrumentation Laboratory for providing research facilities.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
BT: writing the original draft, data curation, investigation, methodology. JD: supervision, validation, writing, review and editing. BK: review, editing and formal analysis. TA: formal analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
No animal/human studies were carried out in the present work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Taxak, B., Devi, J., Kumar, B. et al. Hydrazone-containing organotin(IV) complexes: synthesis, characterization, antimicrobial, antioxidant activity and molecular-docking studies. Biometals (2024). https://doi.org/10.1007/s10534-024-00593-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10534-024-00593-2